ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| JAMPALA BALAJI, BANDI RAMACHANDRA, N.V.S.NAIDU* Department of Chemistry, Sri Venkateswara University, Tirupathi, India |
| Corresponding Author: N.V.S.NAIDU, |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
This work is concerned with application of simple, accurate, precise and selective reverse phase high performance liquid chromatographic (RP-HPLC) method for determination of pregabalin in bulk and determination of pregabalin capsule dosage form. Chromatographic separation was achieved gradiently using waters alliance 2695 separation module, C18 column ( 250 x 4.6 mm, 3.5 μm)at temperature 25o C . Flow rate selected was 0.8 ml/min using PDA detector at 210 nm. Mobile phase was prepared using A-pump 10 mM NH4OAC in water (pH-6.8) and B-pump acetonitrile and methanol in the ratio of 80:20 which gave better resolution and sensitivity. The developed method was validated with respect to linearity, specificity, precession robustness and accuracy.
INTRODUCTION |
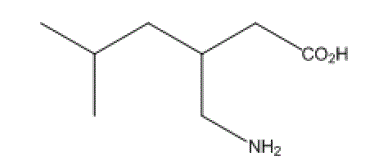
| Pregabalin[1] chemically known as (S) –2(amino methyl)-5-methyl hexanoicacid, is a white crystalline solid , which is soluble in water and in both basic and acidicaqueous solutionsand used as anticonvulsant , analgesic medication and neurotransmitter. It binds with high affinity and specificity to voltage-gated calcium channel alpha (2)-delta proteins [2,3].It received U.S.FDA approval for use in treating neuropathy pain and post herpetic neuralgia in 2004,appeared on the U.S market in fall 2005[4].Recent studies have shown that pregabalin is effective at treating chronic pain in disorders such as fibromyalgia[5] and spinal card injury[6].It is consider to have a low potential for abuse, and a limited dependence liability if missed, and is thus classified as a schedule V drug in the U.S[7]. Although various bio analytical methods for estimation of pregabalin in human serum [8] and spectrophotometric method for estimation of pregabalin in dosage form [9,10] have been reported in the literature. Recently a new validated HPLC method [11] was developed for determination of pregabalin in bulk drug and capsule dosage forms.Allof these methods are very expensive because these methods require long and tedious pre-treatment of thesamples, laborious clean up procedures andderivatization for the analysisof pregabalin.It requires simple new HPLC method for analysis of pregabalin in bulk and the determination of pregabalin in capsules. So the attempt was made to develop a simple, efficient and selective method for the analysis of pregabalin in bulk and the determination of pregabalin in capsules. The UV detection, which is readily available in most analytical and pharmaceutical laboratories was used for HPLC instrumentation. The run time of the method was less than 10 minutes (6minutes). |
 |
EXPERIMENTAL |
| Materials |
| i. Pregabalin (Lee Pharma, Hyderabad, AP, India) . |
| ii. Pharmaceutical formulation of Pregabalin such as Pregabalin 75 (Torrent pharmaceutical ltd. Baddi, India) |
| iii. Ammonium acetate was purchased from Merck, Mumbai, India. |
| iv. Acetonitrile was HPLC grade purchased from Qualigens fine chemicals,Mumbai, India. |
| v. Methanol was HPLC grade purchased from Qualigens fine chemicals,Mumbai, India. |
| vi. All other chemicals were of analytical grade and used without any further purification. |
INSTRUMENTATION |
| i. The HPLC used was Waters alliance 2695 separation module, waters 2996 photodiode array detector, Empower software. |
| ii. Micro mass, Quattro micro TM API, ESCI MULTI-MODE IONISATION ( make Waters ) , Masslynx software. |
| iii. Digital pH meter, model-LP-1393 (makePOLMAN). |
| iv. The HPLC column used was a X-Bridge C18, 3.5 μm (150mm x 4.6mm) , make waters |
| Chromatographic conditions |
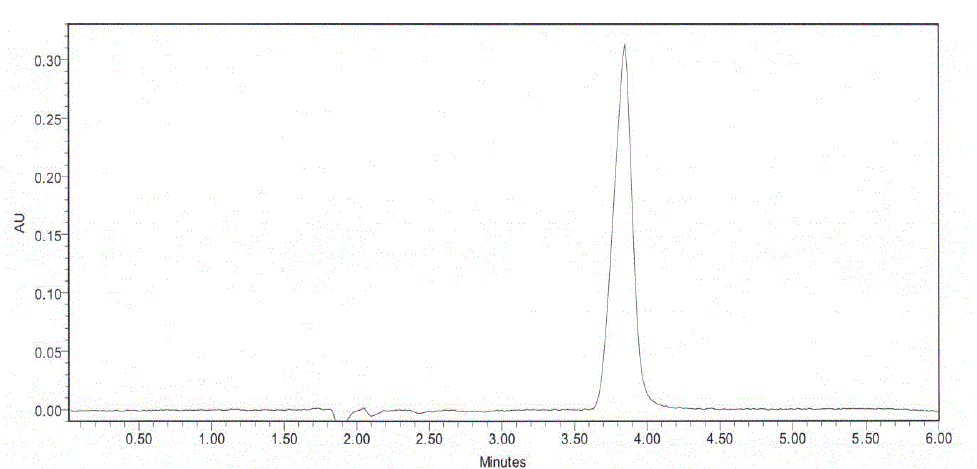
| The proposed gradient method was performed using a liquid chromatography of model Waters alliance, 2695 separation model. The chromatographic separation was achieved on a Waters X-Bridge C18, 3.5μm (150 mm X 4.6 mm)column. The gradient LC method employs solution A and B as mobile phase. The solution A contains 0.01 M Ammonium acetate in water (pH – 6.8). The solution B contains a mixture of acetonitrile and methanol in the ratio of 80: 20. The flow rate was 0.8 ml /min and the detection wavelength was 210 nm. For LCMS the above conditions HPLC connected to mass instrument (Micro mass, Quattro microTMAPI , ESCI Multi mode Ionization, ESI source , triple quad analyser ) and detect the mass . |
 |
 |
| Standard solution |
| Stock standard solution of pregabalin was prepared by dissolving an appropriate amount of the compound in mobile phase to give a final concentration of 0.5 mg/1ml. (A: 5mM NH4OAC in water ( pH 6.8 ) , B: 20%methanol + 80% acetonitrile , diluent – A : B :: 80 : 20 ) |
METHODS |
| System suitability |
| System suitability test is an integral part of liquid chromatographic method. It is used to verification of the resolution of the chromatographic system or adequate for the analysis to be done. The tests are based on the concept that the equipment, electronic analytical operations and samples to be analysed constitute an integral system that can be evaluated as such. |
| Procedure for determination of pregabalin in pharmaceuticals formulations |
| Pregalin75mg capsule was weighed accurately and finally powdered. A quantity of the powder equivalent to 100 mg of pregabalin was extracted by shaking with 25ml of the mobile phase (A : 5mM NH4OAC in water ( pH 6.8 ) , B : 20%methanol + 80% acetonitrile , diluent –A : B :: 80 : 20).It was filtered through a 0.45 μm Millipore filter. The solution was diluted with mobile phase to obtain a concentration of about 0.5mg/ml and then analysed by the following proposed procedures and determine the pregabalin in capsule. In the determination of pregabalin in capsule formulations, the result obtained showed that the recoveries were high and RSD value is low. So this method is suitable for routine determination of pregabalin in its pharmaceutical preparation. The results are shown in table4. |
METHOD VALIDATION |
| Specificity and selectivity |
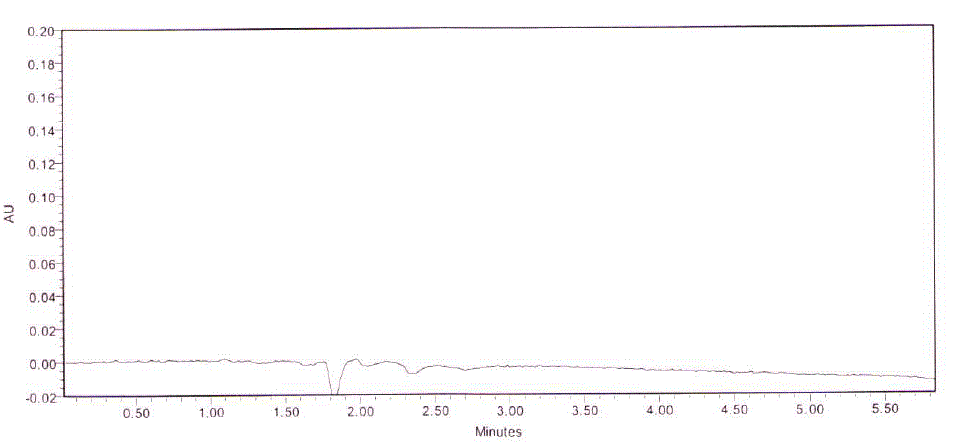
| The specificity of the method was checked by injecting blank solution and sample solution. There was no interference from blank and excipients at the retention time of analyte peak. Based on spectral data, the peak purity checked there is no interference from relative impurities and excipients. |
| Linearity |
| Pregabalin showed linearity in the concentration range of 150μg/mlto 850μg/ml. The correlation coefficient was 0.999.The results are shown in table3. |
| Accuracy |
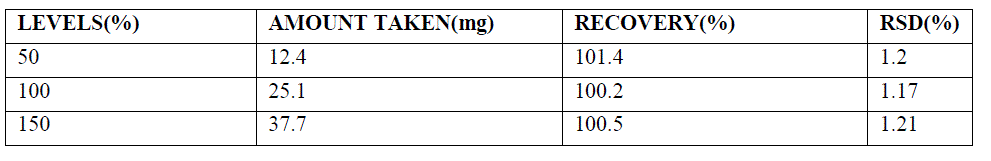
| The accuracy of the method was determined by adding known amount of drug substance corresponding to three concentration levels of 50%, 100%, and 150% of target analyte concentration along with the excipients in triplicate. The accuracy was calculated by the percentage of analyte recovered by the assay method. Based on our results that the method is highly accurate. The accuracy results are shown in table1. |
| Precision |
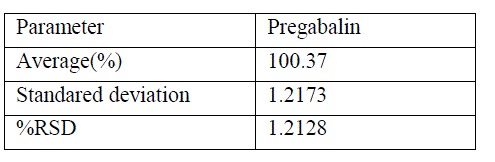
| The precision of the method was calculated within the limits based on analysis of pregabalin.The results of the precision were shown in table2. |
| Robustness of the method |
| Changing with the flow rate, pH, mobile phase ratio and temperature, no change in retention time of pregabalin.It indicates robustness of the method. |
RESULTS AND DISCUSSIONS |
| The reverse phase HPLC method was developed and validated of pregabalin and determination of pregabalin in capsule dosage form. It was validated for linearity, specificity, precision and accuracy. As per my best of knowledge most of the methodsare developed in phosphate buffers. But the phosphate buffers are not suitable for LCMS analysis and ammonium acetate buffer was suitable for LCMS analysis. So the developed HPLC method was suitable for LCMS analysis. |
 ' ' |
 |
CONCLUSION |
| The developed HPLC method was simple, accurate and reproducible method for routine analysis of pregabalin in bulk and pharmaceutical formulations. The major advantage of this method was short retention time and same method was suitable for LCMS analysis. |
References |
|