ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Naveen Bharat1, Md. Irshad2, Md. Moshahid Alam Rizvi3, and Tasneem Fatma4
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Present study screened ten Cyanobacterial extracts for antimicrobial activity, cytotoxicity (against human cervical carcinoma cells Hela and SiHa) & GC-MS for chemical composition. Cyanobacterial extracts were subjected to Agar Well Diffusion Assay at concentration of 100μg. well-1 and incubated at 37 + 2oC. Inhibition zone was measured in millimetres (mm) after 18-24 hour. Minimum inhibitory concentration (MIC) was determined by using the broth microdilution method in 96-well microtitre plates. Drug dilutions were performed using cation adjusted Mueller Hinton Broth (MHB) in a concentration range of 128-0.25 μg.ml-1. Cytotoxicities were assessed using MTT assay. GCMS analysis was carried out on a GCMS-QP 2010 Plus Shimadzu system having RTX-5 column (Restek, USA) (60 m, ID 0.25mm, film thickness 0.25 μm). Cyanobacterial extracts exhibited significant antibacterial effect on clinical isolates of S.aureus MRSA & MRSE whereas selectively inhibited Gram-negative. Minimum inhibitory concentrations (MIC) were in the range between 64 to 128 μg/ml. Activity Index of active extracts ranged from 0.33 to 1.50. Activity Index and Zone of Inhibition were significantly correlated with p>0.03.GC-MS detected distinct groups of active compounds with pronounced presence of saturated and unsaturated fatty acids. Pharmaceutically important compounds like sesquiterpenoids (Farnesol), dicarboxylic acids, imidazole, indolinones, α-tocopherol, phenolics, phytosterols, heptadecane, tetradecane and 9-octadecenal were moderately present. Extracts also exhibited cytotoxic effect on human cervical carcinoma cells lines with LD50 values ranging from 34 to 146μg/ml. Cyanobacterial species have distinct active group metabolites which are promising sources of antiproliferative and antimicrobial compounds
Keywords |
| Cyanobacteria; GC-MS; Antibacterial; cytotoxicity. |
INTRODUCTION |
| Cyanobacteria are morphologically diverse group of Gram-negative eubacteria leading in the development of natural biopharmaceuticals due to presence of structurally diverse groups of compound [1a,b]. Antimicrobial property of natural products have gained importance in recent years due to growing antibiotic resistance and further increase in the infection by antibiotic resistant microorganisms [2a,b]. Present study found chemically diverse groups of compounds among 10 fresh water Cyanobacteria sp. Thin layer chromatographic study showed qualitative relationship as pair affinity (PA) value 50% between H. fontinalis and L. officinalis, 49.2% between A. ambigua and G. gelatinosa, 37.5% between Chrococcus sp., A. fertillisma, and Anabaena sp., 33.3% between A. variabilis, W. prolifica, and Scytonema sp., 28.6% between G. gelatinosa, A. variabilis, W. prolifica, and Scytonema sp. and 16.7% between A. variabilis, W. prolifica, and Scytonema sp. (Fig. 1). |
 |
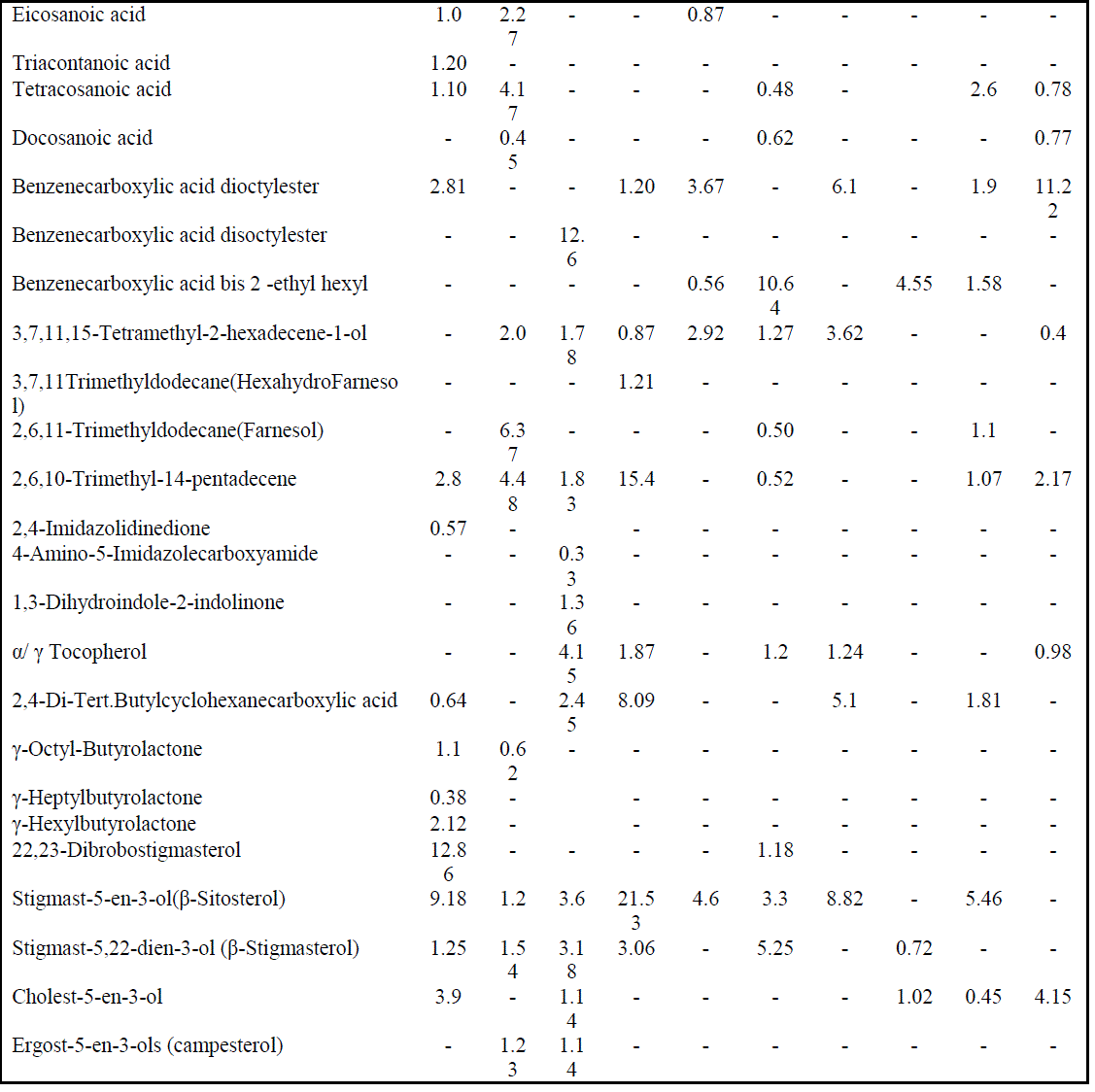
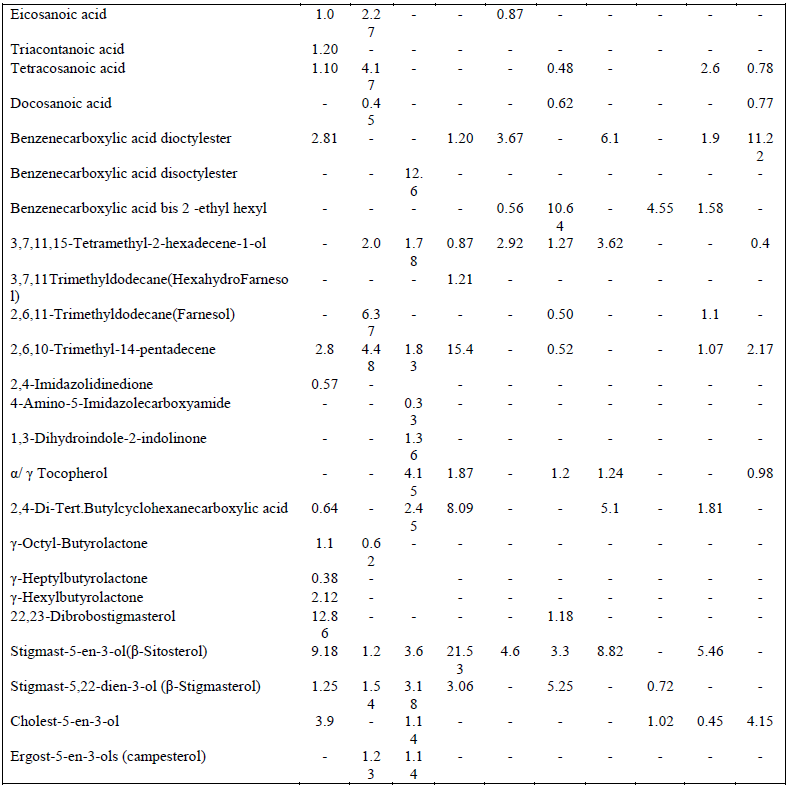
| Highest PA values predict the existence of some common compounds within these species [3]. These relationships were also correlated with the grouped affinity and isolation values of these species. The GC-MS total ion chromatogram of these extracts resolved into several peaks indicating an equally corresponding compounds; 23 in H. fontinalis, 21 in A. ambigua, 51 in L. officinalis,25 in G. gelatinosa, 18 in Chrococcus Sp., 34 in A. fertillisma, 27 in Anabaena Sp., 19 in A. variabilis, 19 in W. prolifica & 25 in Scytonema Sp.(Data not shown here). Principal and pharmacologically active components identified in these extracts included several fatty acids (hydroxy, saturated, branched, unsaturated ), tertiary butyl group containing compounds, phenols, terpenoids, phytols, farnesol , hexahydro farnesol, dicarboxylic acids, lactones, heterocyclic compounds, several polyhydroxysterols, nucleoside & complex sugars and natural antioxidant alpha- tocopherol. These components may act synergistically and induce antimicrobial as well as cytotoxic activities. Among fatty acids hexadecanoic acids (C17H34O2:Mw-270), 9,12-octadecadienoic acid (C19H34O2:Mw-294 ), 9-Octadecenoic acids (C19H36O2:Mw-296), 10 octadecenoic(C19H36O2:Mw-296), eicosanoic(C21H42O2: Mw-326), triacontanoic (C31H62O2:Mw-466) and tetracosanoic acid(C25H50O2:Mw-382) were abundantly present. Benzenedicarboxylic acids such as 1,2-benzenedicarboxylic acid –dioctyl ester (C24H38O4: Mw- 390), disooctyl ester (C24H38O4:Mw-390), ethyl hexyl ester (C24H38O4:Mw-278) were also present which are potent antibacterial, antifungal and antialgal agents [4a,b,c] ; phytols like 3,7,11(tri); 3,7,11,15(tetra) ; & 2,6,10; 2,6,11- Trimethyldodecane (C15H32 :Mw-212) were detected which are known as hexahydrofarnesol and farnesol reportedly possess strong antibacterial activities [5] Imidazole containing alkaloids like 2,4-Imidazolidinedione(C8H6N4O5:Mw- 238),4 amino-5-imidazole carboxymide(C4H6N4O: Mw-126) &1,3dihydroindol-2indolinone(C8H7NO: Mw-133) were present which are reported to possess antimicrobial, cytotoxic, antitumor and antiviral activities [6a,b]. Other pharmacologically active compounds detected like 9,12, octadecadienoic acid(C19H34O2:Mw-294), α- tocopherol(C29H50O2:Mw-430), and 2,4, di-, tert-butyl phenol(C14H22O:Mw-206) have been reported as anti-oxidant [7]; 9,12,15, octadecatrienoic acid (C20H34O2:Mw-306) have been reported as anti-inflammatory activity [8]; γ-octyl butyrolactones(C12H22O2: Mw-198) are known as pro-drugs [9]. Several steroidal compounds like dibromostigmasterol acetate (C31H50Br2O2 : Mw-612); Stigmast-5-En-3-ol(C29H50O : Mw-414); Stigmasta-5,22-Dien-3.beta-ol (C31H50O2 :Mw-454); cholest 5-en-3-ol (C28H45ClO2 : Mw-448 ); Ergost-5-En-3-ol (C28H48O : Mw-400); Stigmast-5-En-3-Yl 9- octadecenoate (C47H82O2 : Mw-678) were predominantly present in these extracts (Table 4). These typical sterols are known to posses antibacterial, antifungal and cytotoxic properties. This study was designed to investigate antibacterial activity of Cyanobacterial extracts against 12 Gram-positive and 12 Gram-negative bacteria strains as listed in table I. They were evaluated for their antibacterial potential by agar-well diffusion assay. MICs values were determined further to check the concentration ranges for significant inhibition. Chromatographic separation of the extracts was done by employing TLC. The study was extended to identify the chemical composition of the extracts by Gas Chromatography coupled with Mass Spectrometer (GC-MS) and their cytotoxicity against cervical cancer cell line Hela and SiHa using MTT assay. |
MATERIALS AND METHODS |
| A. Collection of cyanobacterial culture |
| Fresh water cyanobacteria (Lyngbya officinalis NCCU-102, Gleocapsa gelatinosa NCCU-430, Chrococcus sp. NCCU- 207, Aulosira fertillisma NCCU-444, Anabaena ambigua NCCU-160, Hapalosiphon fontinalis NCCU-339, Anabaena sp. NCCU-09, Anabaena variabilis NCCU-441, Westiellopsis prolifica NCCU-331, and Scytonema sp. NCCU-126) were procured from the National Centre for Culture Collection and Utilization of Blue Green Algae, Indian Agriculture Research Institute, New Delhi, India. Cyanobacteria were grown phototrophically in BG-11 medium [10]. |
| B. Collection of microorganisms |
| Bacterial strains (Table I) were obtained generously from the culture collections of Ranbaxy Research Laboratories (RRL), Gurgaon, All India Institute of Medical Sciences (AIIMS), New Delhi and National Facility for Marine Cyanobacteria, Department of Marine Biotechnology, Bharathidasan University, India. The test bacterial strains were inoculated on Tryptic Soy Agar (TSA, Difco) and incubated overnight at 37 oC ± 2 oC. |
 |
 |
| C. Cyanobacterial biomass harvesting and extraction |
| Cyanobacterial late exponential growth phase culture were harvested by centrifugation at 5000 x g for 10 min. Biomass of Cyanobacteria were lyophilized (Labconco, USA) and crushed to fine powder. Fine powder of biomass and diethyl ether solvent were mixed in 100 ml flask maintained in ice bath and sonicated using small probe (Sonics Vibracell, USA), for 2 min, time interval of 5 sec, at 10% of total amplitude and left for 30 min. Extracts were filtered through Whatman No.1 filter paper and solvent was dried in rotary evaporator (Buchi, USA). Dry residue was transferred to a freeze drying vessel and used for different assays by the clearance of bio-safety and ethical committee of the institute. |
| D. Thin layer chromatography |
| Chromatography was performed on silica gel 60 F254 TLC aluminium sheet 20x20 cm (Merck). A 50μl sample (1mg/ml) was spotted manually on silica gel and run in mobile phase, hexane: ethyl acetate (8.5:1.5). Numbers of spots were counted and retention factors (RF) of each spot was calculated. Qualitative relationships of the each spots were calculated as values of paired affinity, group affinity and isolation value. |
| E. Gas chromatography – Mass spectrometry |
| Cyanobacterial extracts were subjected to GC-MS analysis on a GCMS-QP 2010 Plus Shimadzu system (All India Research Facility, JNU, Campus, New Delhi)comprising an auto injector and RTX-5 column (Restek, USA) (60 m, ID 0.25mm, film thickness 0.25 μm). Helium was used as the carrier gas (1ml/min). The column temperature was programmed at 60°C for 1 min and then raised to 180°C for the total analysis time of 50 min. Sample (0.1mL) was injected in the splitless mode. The chemical components of the extracts were identified by comparing the retention time of the chromatographic peaks with that of authentic compound using the WILEY8.LIB and NIST05s.LIB. |
| F. Agar well diffusion assay |
| Antimicrobial assay of dried Cyanobacterial extracts (reconstituted in 1% aq. DMSO) against the bacteria were assessed using the National Committee for Clinical Laboratory Standards guidelines (NCCLS) for agar well diffusion assay [11]. Inhibition zone was measured in millimetres (mm) after 24 hour. 1% DMSO used as negative control and standard antibiotic, Vancomycin (Eli Lily), Ciprofloxacin and Kanamycin (HiMedia) were used as positive control. Antimicrobial index was calculated as: Inhibition zone of test sample /Inhibition zone of standard. |
| G. Minimum Inhibitory Concentration (MIC) by broth microdilution assay |
| MIC of Cyanobacterial extracts was determined by using the broth microdilution method as recommended by NCCLS [12]. The tests were performed in sterile 96-well microtitre plates. Drug dilutions were performed using cation adjusted Mueller Hinton Broth (MHB, Difco) to get a concentration range of 0.25 -128 μg.ml-1. Inoculum was prepared by direct colony suspension in normal saline to get an innoculum size 1-2 x 106 CFU.ml-1. 100 μl of the innoculum was added in each wells containing 100μl extracts dilution (final innoculum was approx. 3-7 x105 cfu/ml). For positive control extracts free inoculated broth and for negative controls sterile broth were used. The final volume in each well was 200 μl. Plates were incubated for 24 hours at 37 + 2oC and observed for presence or absence of growth. The lowest concentration of the extracts that prevented the growth of bacteria was considered to be the MIC. |
| H. Cytotoxicity Assay |
| Human cervical cancer cell lines SiHa and HeLa were obtained from National Centre for Cell Sciences (NCCS) Pune, India. Cell lines were grown as monolayer cultures in Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum and antibiotics (100 units/ml penicillin and 100 mg L-1 streptomycin) in a humidified atmosphere of 5% CO2 at 37°C. Cytotoxicity of Cyanobacterial extracts were carried in overnight seeded cells (2x104/well) for 48 h. At the end of the treatment, the medium was removed and cells were incubated with 20μl of MTT (5 mg/ml in PBS) in fresh medium for 4 hrs at 37oC. After four hours, formazan crystals, formed by mitochondrial reduction of MTT, were solubilized in DMSO (200μl/well) and the absorbance was read at 570 nm after 10 min incubation on the iMark Microplate Reader (Bio-Rad, USA) [13a,b]. Cytotoxicity of Cyanobacterial species was expressed as LC50. |
| I. Statistical analysis |
| The mean value ± standard error of mean (SEM) was determined from triplicate samples for each experimental group using using Graph Pad prism 2.01 (2) version for Windows 2001 (Graph Pad Software, San Diego). Statistical significance was examined with Student’s t test. |
RESULTS AND DISCUSSIONS |
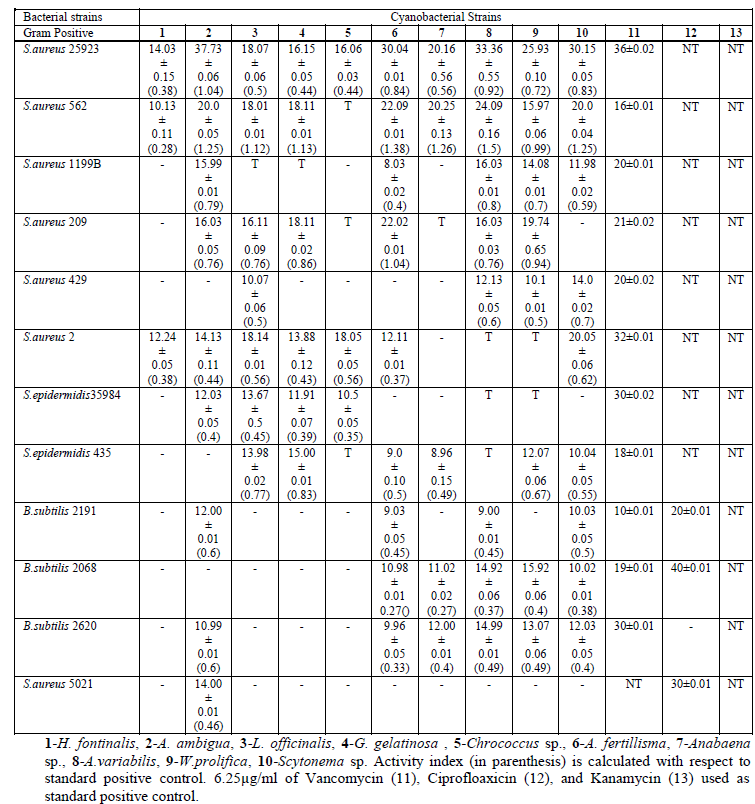
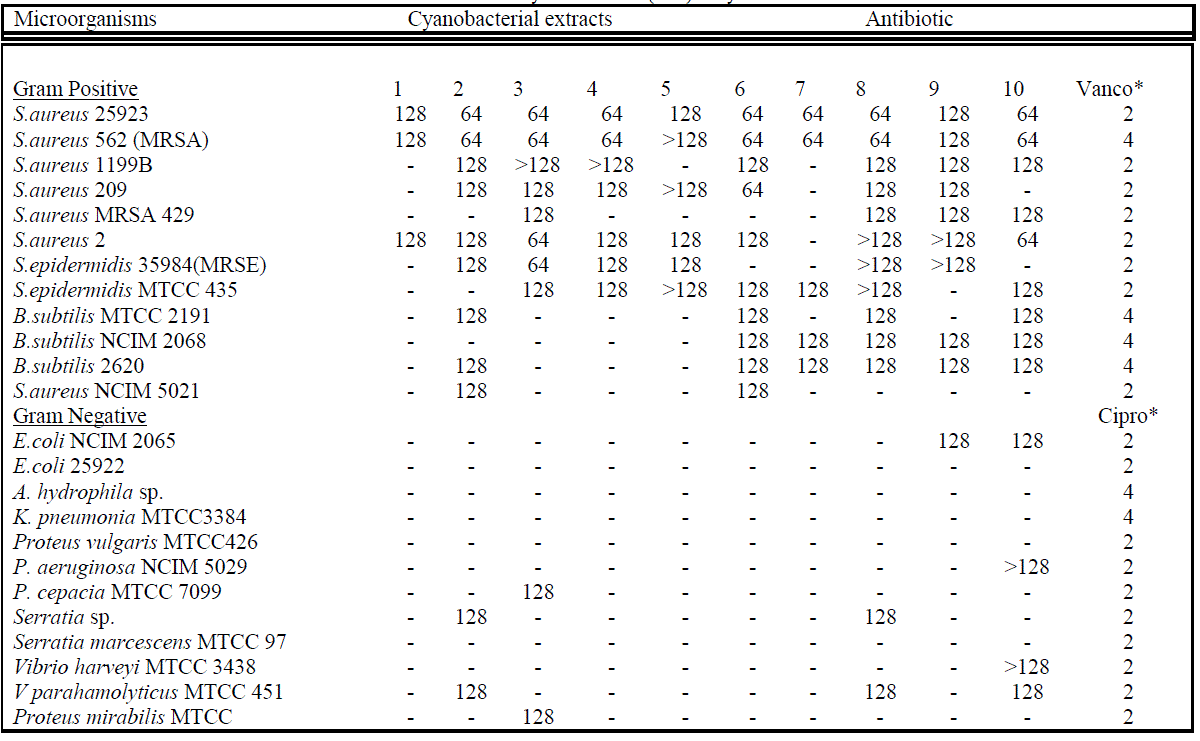
| The antibacterial activities (zone of inhibition; respective activity index and MIC values) of each extracts against Gram-positive and Gram-negative bacteria including MRSA & MRSE are summarized in tables II a,b & III. Cyanobacterial extracts significantly inhibited Gram-positive bacteria, whereas selectively inhibited Gram-negative bacteria. It was noted that the cyanobacterial extracts inhibited all tested Staphylococcal isolates. Maximum inhibition zone in millimetre was observed in case of A. ambigua (37.73), A.variabilis (33.36), Scytonema Sp. (30.15) and A. fertillisma (30.01) against S.aureus 25923. However, highest activity index was in order of A.variabilis, A. fertillisma, Anabaena sp., A. ambigua, and Scytonema Sp. against MRSA (Methicillin Resistant Staphylococcus aureus) 562 with MICs of 64μg/ml. Hence, this strong activity index of cyanobacterial extract directly correlate its antimicrobial potential. Whereas, towards Gram-negative bacteria highest activity index was shown only by A. ambigua and A. variabilis against V. parahemolyticus 451 and Serratia sp., while L. officinalis showed highest activity index against Proteus mirabilis. & Pseudomonas cepacia. Zone of Inhibition and Activity Index of each active extracts were significantly correlated (p, ≤ 0.03). Minimum inhibitory concentrations (MIC) of some of the most active extracts range between 64μg/ml to 128μg/ml, are shown in table 3. It was noted that lower MIC (64μg/ml) was obtained only against S.aureus 25923 and S.aureus 562 treated with A. ambigua, L. officinalis, G. gelatinosa, A. fertillisma, A. variabilis, W. prolifica, and |
| TABLE IIa : Zones of Inhibition Produced by Extracts in the Agar Well Diffusion Assay. (Mean of Three Experiments) |
 |
 |
 |
| Scytonema sp.; against S.aureus 2 and S. epidermidis 35984 treated with Scytonema sp. and L. officinalis respectively. MICs values of bioactive extracts and its zone of inhibition were significantly correlated with p, ≤0.05. The antimicrobial property of cyanobacterial species also significantly correlated with the paired affinity (PA) relation. Cyanobacterial stains extracts also showed cytotoxic against human cervix cancerous cell lines. The cytotoxicity trend showed steep rises with the initially increase of concentration and further became constant at high doses. LD50 against HeLa and SiHa cell lines (Fig. 2) |
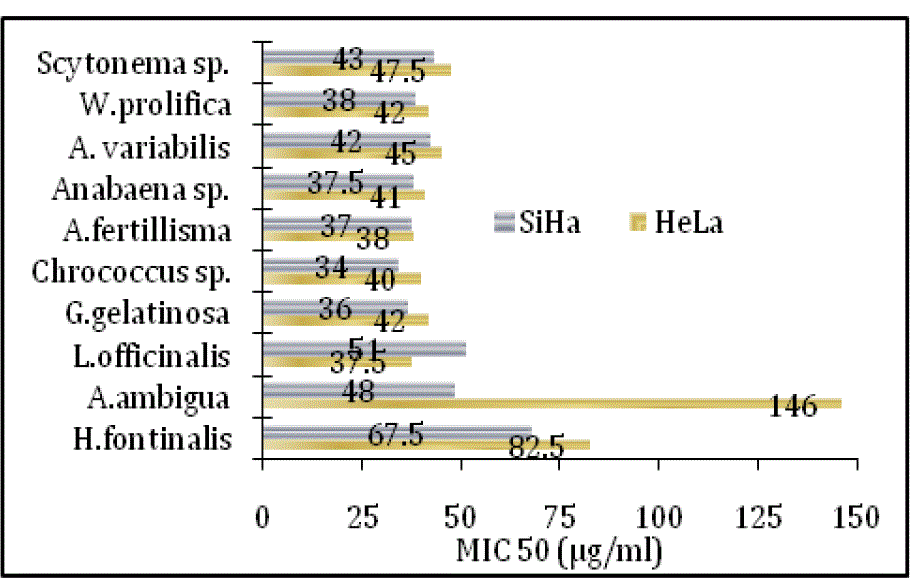 |
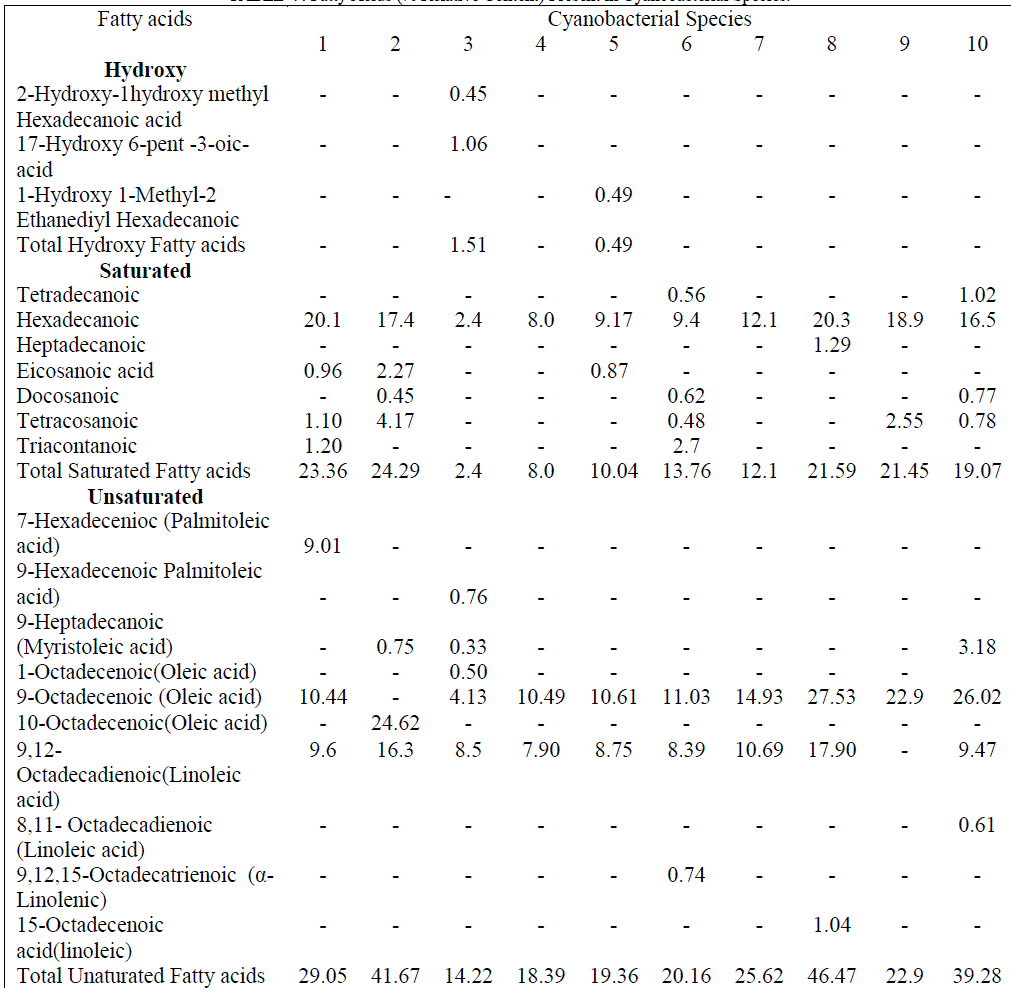
| Comparatively, highest cytotoxicity even at low concentration was observed in L. officinalis, G. gelatinosa, Chrococcus sp., A. fertillisma, Anabaena sp. and A. variabilis extracts, whereas A. ambigua treated HeLa cell line showed lowest cytotoxicity (LD50 146μg/ml). The GC-MS total ion chromatogram of these extracts resolved into several peaks indicating an equally corresponding compounds; 23 in H. fontinalis, 21 in A. ambigua, 51 in L. officinalis,25 in G. gelatinosa, 18 in Chrococcus Sp., 34 in A. fertillisma, 27 in Anabaena Sp., 19 in A. variabilis, 19 in W. prolifica & 25 in Scytonema Sp.(data not shown here).Several important chemicals were identified by GC-MS (Table IV). |
 |
 |
 |
| These extracts contained 03 hydroxy acids (1.51 & 0.49% relative content in L. officinalis & Chrococcus sp); 07 saturated and 10 unsaturated fatty acids (Table V). The percent relative content for both types of fatty acids was as high as 23.36 and 46.47 % in H. fontinalis and A. variabilis respectively. The abundance of saturated fatty acids like palmitic (20.1, 17.4, 20.3, 18.9 &16.5%) & myrestic acid (0.5 & 1.02%) and the unsaturated fatty acids palmitoleic (9.01%), oleic (4.13 – 27.53 %), linoleic (7.9-17.90%) and α-linolenic(0.74%) acids present in respective cyanobacterial species correlates their significant antibacterial action. Of these unsaturated fatty acids the antimicrobial activity of α-linolenic acid is well established. [14] A moderate content of three isomers of benzenedicarboxylic acid (dioctyl, disooctyl and ethylhexyl) were present in each extract except in A. ambigua. These are known as phthalate derivatives; they exhibit a pronounced anti-bacterial action and have been reported by various investigators as cited earlier from different sources. Among these compounds, 1, 2-Benzenedicarboxylicacid bis(2 ethyl |
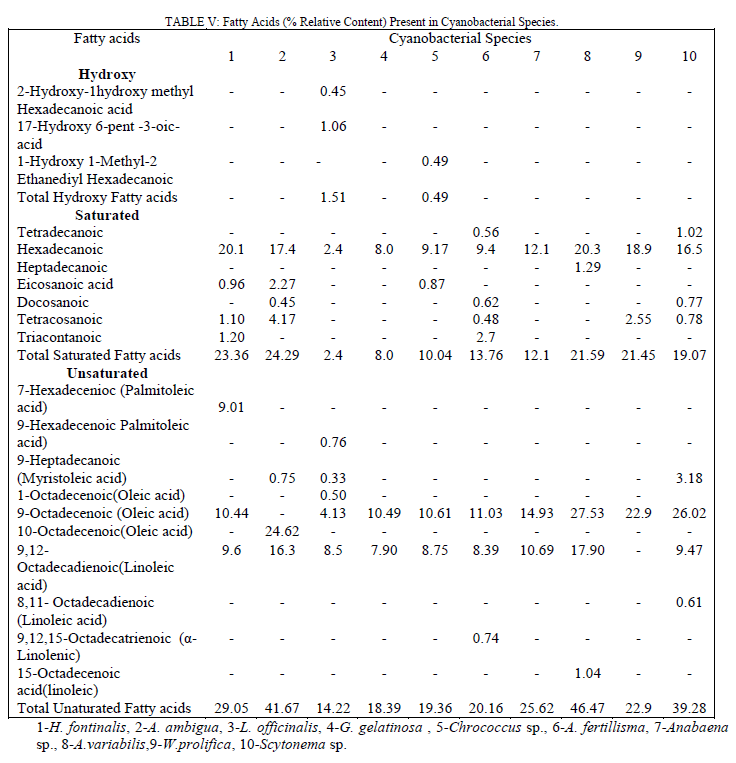 |
| hexyl) has potent antibacterial activity and was present in Chrococcus Sp. (0.56%), A. Fertillisma (10.64%), A. Variabilis (4.55%) & W.prolifica (1.58%) which also showed the higher zone of inhibitions (16.06, 30.04, 33.36 &25.93 mm) and lower MIC of 64 to 128 μg/ml against S. aureus 25923. Among the other two isomers of these phthalates, diisoctyl was detected only in L.officinalis (12.6% ); and dioctyl was detected in H. Fontinalis (2.81%), G. gelatinosa (1.20%), A. Fertillisma (3.67%), Anabaena Sp. (6.1%), W.prolifica (1.9%) and Scytonema Sp. (11.2%). The phytols, also known as terpene alcohols, were present as lower contents in H. fontinalis (2.8%), L officinalis (3.61%), Chrococcus Sp.(2.92%) A fertillisma (2.29%), Anabaena Sp. (3.62%), W. prolifica (2.08%) and Scytonema Sp. (2.57%) while it was present in high amounts in G. gelatinosa (17.48%) A. Ambigua(12.85%). Terpene alcohols, also known as sesquiterpenes were also present in the extracts. In the extracts of A. Ambigua, A. Fertillisma & W .prolifica; a sesquiterpenoid known as farnesol which is antibacterial in its action was present as 6.37, 0.50 & 1.1 % respectively. Besides this, trimethyl alkanes (3,7,11-dodecane) & tetramethyl alkenes (3,7,11,15-hexadecene) were also detected in other extracts. As cited earlier Indole , bisindole, imidazole containing alkaloids and other several analogues have been reported from diverse sources exhibiting pharmacological activities such as antimicrobial, cytotoxic, antitumor and antiviral. In our study three compounds having imidazole and indolinone as functional group were detected as minor fractions in H. fontinalis & L.officinalis. The imidazole or other derivative like imidazolidinedione detected in these extracts is capable of inducing such effects. In these extracts a pool of natural antioxidants like α-tocopherol and an unusual neoacid (2,4-di-tert.butyl phenol/cyclohexanecarboxylic acid) which are reported from plants as well as in marine algae [15],[16] are said to demonstrate good antioxidant and fungicidal properties which occurs in natural peptides and natural alkaloids in cyanobacteria, fungi and in other microbes [17] were present in 50% of the extracts. In this study, six typical steroidal components present were 22, 23-Dibrobostigmasterol, Stigmast-5-en-3-ol (β-Sitosterol), Stigmast-5,22-dien-3-ol (β-Stigmasterol), Cholest-5-en-3-ol, Ergost-5-en-3-ols (campesterol) and Stigmast-5-en-3-yl-9- octadecanoate. These sterols predominantly possess several pharmacological properties. Of these sterols β-Sitosterol is known to have antifungal activity and inhibit carcinogenesis [18, 19], ergosterol, sitosterol and stigmasterol reportedly has antifungal activty [20]. Several other compounds having furan and quinone structures, aldehydes like octadecenal and esters of citronellol and glycerol were present in minute amounts. Majority of cyanobacterial species also contains small amounts of short-chain hydrocarbons [21] like heptadecane. Small amounts of tetradecane (0.56 & 1.02 %) were detected in Chrococcus Sp. and Scytonema Sp. while heptadecane was detected as 0.33 & 20.95% in L.officinalis & Anabeana Sp. respectively. Among the least detected compounds were octadecanthiol, 1-napthelenepentanoic acid, scopolamine and three different nucleoside sugar moiety. The primary aim of this study was to reveal the bioactivity of these Cyanobacterial species. Diethyl ether extracts of these cyanobacterial species were tested against 24 human bacterial pathogens by agar well diffusion assay. It revealed, pronounced antibacterial activity against Gram positive bacteria by all extracts except in Chrococcus and Anabaena Sp. Anti bacterial activity against Gram positive bacteria was more frequent than against Gram negative bacteria. These extracts possessed different intensity of antibacterial action seen as in inhibition zones. This variation in inhibition zones might be attributed due to the difference in total pool of active metabolites present in these extracts. High contents of active fatty acids in these extracts of cyanobacteria are responsible for antimicrobial effects. Total content of saturated and unsaturated fatty acids ranged between 2.4 to 23.36% and 14.22 to 46.47% respectively. Fatty acids largely known in cyanobacteria are Octadecanoic, Octadecenoic, hexadecanoic acids, hexadecenoic acids, hexadecadienioc [21]. The fatty acids 9,12-octadecadienoic (Linoleic); 9,12,15-octadecatrienoic (α-Linolenic) along with saturated fatty acids (tetradecanoic, heptadecanoic, eicosanoic and docosanoic acid) were present in these extracts affording antimicrobial and cytotoxic action. Recently, linoleic, α- Linolenic acids have been reported to have growth inhibitory and proapoptotic effects [22], [23]. In addition to these fatty acids three isomers of 1, 2-benzenedicarobxylic acid (dioctyl, diisoctyl & ehylhexyl esters) were detected. Though in nature dicarboxylic acids are found as components of polymeric compounds in plants but some unusual dicarboxylic acids (short-chain, hydroxyl, branched, saturated and dioic acids) have also been reported from the cyanobacteria Aphanizomenon [24] as minor contents. They have also been reported in water hyacinth (1,2 benzenedicarboxylic acid esters Mw -390) [25]. Dicarboxylic acids such as 1, 2-Benzenedicarboxylic acid are also known to have antibacterial and anti-proliferative potential. Here, in our study at least one among the three isomers of this compound was present in all extracts except in A.ambigua. The antibacterial action by A.ambigua extract remained qualitatively comparable to A. Fertillisma, A. Variabilis, W. prolifica and Scytonema Sp. having low to high content (0.56-11.22%) of these compounds. However, the absence of such compounds in A.ambigua showed lowest toxicity (LD50146μg/ml) against HeLa cell line indicating the importance of this compound as cytotoxic. The high relative content of 1,2- Benzenedicarboxylic acid dioctyl in A. fertillisma (10.64%) & and ethyl hexyl ester in Scytonema Sp.(11.22%) is evidenced with higher cytotoxicities even at lower concentration of extracts against both cell line HeLa and SiHa. It was also noted that A.ambigua had comparatively high content of the sesquiterpenoid compounds also knowns as 2,6,11 & 2,6,10 trimethyl dodecanes (farnesol). This compounds have proven antibacterial action against S.aureus, MRSA (Methicillin Resistant S. aureus) & MSSA (Methicillin Susceptble S. aureus) [26] and fungal growth inhinition [27]. Farnesol has the ability to sensitize S. aureus to antimicrobials when used in syngery with antimicrobials. Therefore, it has been suggested for a potential application as an adjuvant therapeutic agent against biofilm related infections involving MRSA and thereby counteracting drug resistance. Farnesol is also known to posses chemopreventive and anticancerous activity by unknown mechanisms. The compounds belonging to imidazole and indolinones have been previously known for better pharmacological properties such as antibacterial, antifungal and anti-carcinogenesis. Further, the presence of the antioxidants, (alpha- tocopherol), tert-butyl containing compound, gamma butyrolactone and phytosterols are capable of inducing such effects. The heavy presence of sitosterol and stigmasterol seems to potentiate such effects. The aldehyde octadecenal and volatile alkanes (heptadecane and tetracdecane) are also capabale of such activity. The aldehydes (Butanal, heptanal and octanal) have been reported to show biomedical effects [28], [29], [30]. A large fraction of heptadecane was reported in ethyl actetate extracts of three blue green algae (A.variablis, A.flosaquae and O.angustissima) exhibiting antibacterial activity [31]. |
CONCLUSION |
| Several natural products can be explored for the huge needs of novel compounds having medicinal applications. Based on these results it can be concluded that these fresh water strains of cyanobacteria are abundant sources of several pharamaceutically important compounds which can be used as sources of new and useful antimicrobial chemical entities against bacteria. The chemicals present in these extracts are cytotoxic against cervix cancer cell lines which can be potentially explored further. |
ACKNOWLEDGEMENT |
| The authors are thankful to I.A.R.I, New Delhi for Cyanobacterial strains, Ranbaxy Research Laboratories (Gurgaon,India) for microbial strains and I.C.M.R, New Delhi for providing SRF to Naveen Bharat. |
References |
|