ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Srabayeeta Basu Roy1, Subhas C Shit2, Ranjan A Sengupta3, Premal R Shukla1
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
In this study, poly propylene (PP)/potato starch (PS) blend films are prepared using the two roll mill machine. The biodegradability of the films is investigated by soil burial method over a period of 4 months. The percentage weight loss of the bio-composites in the compost soil burial test is increasing with increasing PS content. The biodegradability of the bio-composites is enhanced with increasing starch content because the PS is easily attacked by microorganisms. Any changes in the various properties of the PP/PS before and after degradation are monitored using weight loss, FTIR spectroscopy, a scanning electron microscope (SEM) for surface morphology, a thermo gravimetric analyzer (TGA) for rapid determination of starch content and a differential scanning calorimeter (DSC) for crystallanity. It can be concluded from the results that use of these bio-composites will reduce the environmental problems associated with waste pollution and the study findings support the predicted application of bio-composites as green-composites or eco-materials.
Keywords |
| Bio-composite, Polypropylene (PP), Potato Starch, Biodegradation, Soil burial method |
INTRODUCTION |
| In recent times, biodegradable materials have gained importance particularly for the protection of the environment from ever increasing plastic waste [1,3]. A number of biological materials may be incorporated into biodegradable polymer [4,5]. The main characteristic of biodegradable polymers is that they are biodegradable through the action of the microorganism in appropriate environmental conditions [6]. When in contact with the biodegradable polymer, the microorganisms produce enzymes that break the material in progressively smaller segments reduces its average molecular mass, favoring its degradation in the environment [7]. Biodegradable materials can be completely degraded into natural ecosystems such as active sludge, natural soil, lake and marine. Petroleum-based synthetic polymers are widely used in modern society. Many of the physical and chemical properties of plastics make them ideal materials for a variety of products and applications. Various approaches to render synthetic polymers degradable have been considered. However, the annual worldwide disposal of approximately 150 million tons of petrochemical plastics in commonly used commodities such as polyolefin in packing, bottle and molding products is a significant environmental problem, especially with the continuously increasing production and consumption of these materials [3,8]. Most widely used alkane-derived plastics have poor biodegradability and may have lifetime of hundreds of years when buried in typical solid-waste sites. |
| The most attractive renewable natural polymer resource is starch because of its low cost, wide spread availability and potential for mass production from renewable resources [14,16]. Research on biodegradable plastics based on starch began in the 1970s and continues even today at various laboratories all over the world. Starch satisfies the requirements of having adequate thermal stability with minimum interference in melt properties and negligible disturbance of product quality. Starch is the mixture of amylase and amylopectin. The availability of starch is just after cellulose Potato starch obtained from the tuber of Solanum tuberosum L. and it is a very fine white powder. It is insoluble in cold water and alcohol. The most important industrial sources of starch are wheat, corn, tapioca, potato and rice. Incorporation of starch into a polyolefin matrix was proposed by Griffin as an effective means of accelerating the deterioration of plastics under biotic environmental exposure conditions [12,13]. PP is a popular thermoplastic material widely used in automobile, electrical equipment, furniture and packaging applications because of its excellent and versatile properties but PP is very resistant to hydrolysis and is totally nonbiodegradable. There is great potential interest in incorporating biodegradable material such as starch into conventional plastics such as polypropylene. The present study reports the preparation of PP/PS composite films to examine the biodegradability by soil burial method. The changes in the properties of the PP/PS before and after degradation were monitored using weight loss, FTIR spectroscopy, a scanning electron microscope (SEM) for surface morphology, a thermo gravimetric analyser (TGA) for rapid determination of starch content and a differential scanning calorimeter (DSC) for crystallanity. From these results, we suggest the predicted applications of bio-composites that are considered as an alternative to nonbiodegradable plastic products. |
RELATED WORK |
| Plastics that are formed from non-renewable feedstock are generally petroleum-based and reinforced by glass or carbon fibers. Various types of natural fibers may be used as a filler. Starch has been considered a suitable material source due to its inherent biodegradability, availability and relative low cost [15] for bio-composites. Potato is the world’s fourth largest food crop, following rice, wheat and maize. China is now the world’s largest potato producing country and nearly a third of the world’s potato is harvested from China and India. Polymeric composite containing starch has been developed for different applications and is the subject of several patents [9-11]. The advantages of reinforcing the PP with potato starch for improved physicomechanical properties have already been in our previous publication [17]. The microbial consumption of starch component, in fact, leads to increased porosity, void formation and loss of integrity of the plastic matrix [13]. Biodegradable polymers can be biodegraded eventually by microorganisms in the natural environment into carbon dioxide. The future outlook for advancement in the area of biodegradable plastics is ultimately promising. Research in the diverse combination of raw materials and produce multiple outputs, while releasing no emissions. Economic concerns must be addressed objectively as biodegradable polymer materials are developed, because the future of each product is dependent on its cost competitiveness and society’s ability to pay for it. Many governments are introducing initiatives designed to encourage research and development of biologically based polymers. |
EXPERIMENTAL |
| Materials |
| PP injection-grade H200MK obtained from Reliance Industries, Mumbai, India, was used as a polymer matrix. Its melt flow index (MFI) and density as reported by the manufacturer were 20 g/10 min and 0.919 g/cm3, respectively. Potato starch granules (in-soluble) of 60 mesh size with sulphated ash 0.30% obtained from M/S S D Fine-Chem, Mumbai, India, were used as filler. |
| Compounding and film preparation |
| Starch powder was mixed with PP granules and mixed materials were extruded in a twin-screw extruder (ZV-20 HITORQUE) with L/D ratio of 1:40 in the temperature range of 120-150° C. The extruded strands were palletized and stored in sealed packs containing desiccants. Four levels of filler loading (5, 10, 15 and 20 wt %) were designed in film preparation. The film of 0.10 mm thickness was prepared in the two roll mill machine with L/D ratio of 1:40 in the temperature range of 170-175° C. |
| Biodegradability test |
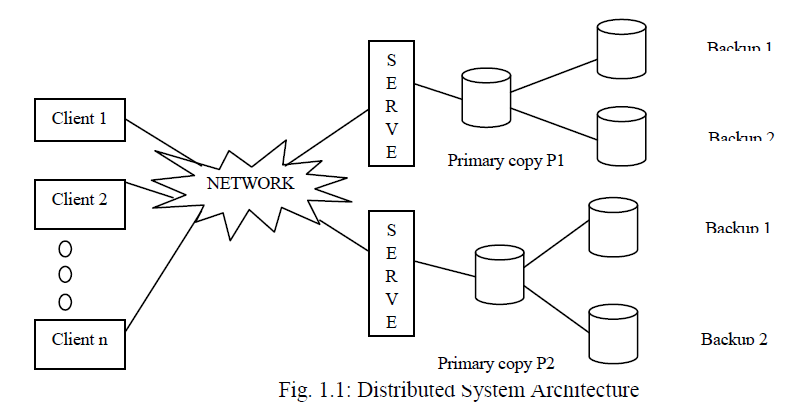
| Biodegradability test (Fig. 1) of all the blends was performed by the soil burial method as per the procedure reported by Potts et. al. [18]. For this test, a number of plastic containers of about 250 ml capacity were filled with compost. Rectangular polymer samples (in triplicate) were cut into 30 x 50 mm pieces and buried in the soil at a depth of about 5 cm. The compost was kept moist by sprinkling water at regular time intervals to maintain 45-50% humidity. The excess water was drained through the hole at the bottom of the container. The containers are stored at about 30-35° C. The degradation of the samples was studied at regular time intervals by removing the samples carefully from the soil and washing them gently with distilled water to remove the soil adhering on the surface. The samples were dried at 60° C under vacuum until a constant weight was obtained. Weight loss of the polymer with respect to time was recorded as a measure of degradation. The soil burial test was studied by evaluating the weight loss of the film over time. The weight loss was calculated using Equation (1) |
| Weight Loss (%) = (Mo – Md)/Mo x 100… (1) |
| Mo = Initial mass; Md = Degradation mass at each designated day |
 |
| Scanning Electron Microscopy |
| The surface morphology of the PP/PS films before and after biodegradation was observed by means of Nova Nano FEG-SEM 450. The micrographs of the samples were taken at different magnifications to identify cracks, holes and other changes on the surface of the samples during degradation. |
| Transmission electron microscopy |
| The diffraction analysis of the sample was determined by transmission electron microscopy by JEOL JEM 2100. Resolution of the instrument was 1.4 Angstrom Unit and the accelerating voltage was 200kV. The sample was prepared by ultra-microtome method and the filament used was LaB6 (Lanthanum hexa boride), made in Japan. |
| Atomic force microscopy |
| AFM imaging was performed on flat bio-composite films of Polypropylene using NT-MDT NTEGRA Prima Scanning Probe Microscope in tapping mode, operated with 100 X 100μ scanner and NSG tip having size at the edge around 10 nm and frequency of 1.01 Hz. The coated area of composite film was subjected to 50μ X 50μ scan under SPM. The topography image was developed with Nova software supplied with the instrument. Average roughness analysis and particle size distribution in the scanned area were studied. |
| Thermal Properties |
| DSC analysis was performed using Mettler Toledo (Star DSC1 SW 9.30). The instrument was calibrated with indium (calibration standard, purity >99.999%) for melting point and heat of fusion. A heating rate of 10 ºC/min was employed in the range of 25 ºC to 500 ºC. Analysis was performed under a nitrogen purge (50 ml/min). An empty pan was used as a reference. |
| Glass transition temperature study |
| DSC analysis for glass transition temperature was carried out using PYRIS DIAMOND DSC (Perkin Elmer, USA) at a heating rate of 10° C/min over a temperature range of -40-200° C. |
| Infrared Spectroscopy |
| Fourier transform infrared (FTIR) spectra of PP/PS films before and after biodegradation, were obtained in SHIMADZU CORP (model A210044 01377) in the spectral region between 4500 and 450 cm-1. |
RESULTS AND DISCUSSION |
| Soil burial test |
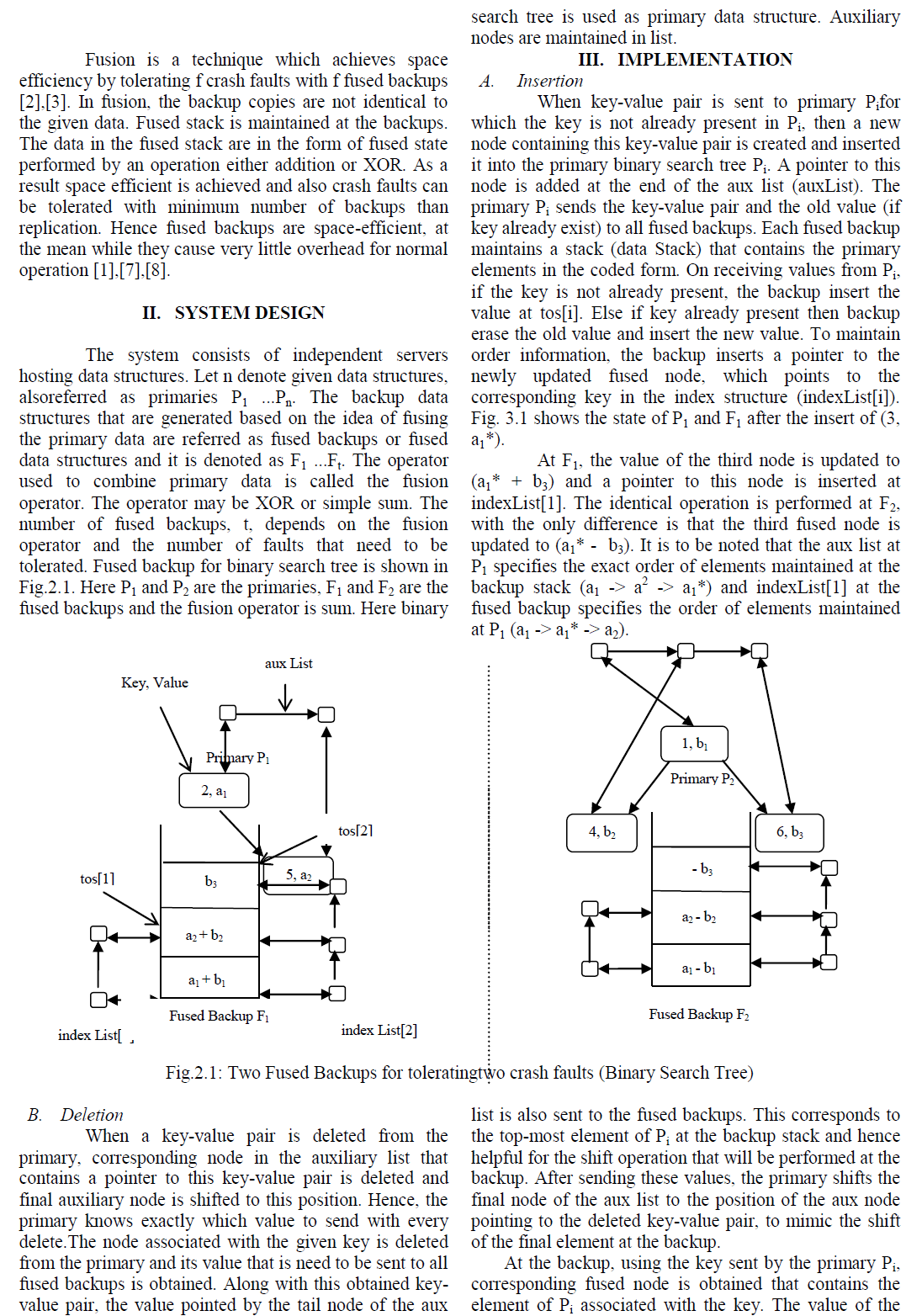
| Fig. 2 shows the weight loss of pure PP and PP/PS films in soil. In the figure, biodegradability increased up to 10.6% as the burial time increased in the soil for 120 days for 20% PP/PS bio-composite. All the films in soil degraded rapidly in the first 30 days. This rapid degradation was due to the composting process, which occurred in two stages: an active composting stage and a curing period. In the first stage, the temperature rose and remained elevated as long as there was available oxygen, which resulted in strong microbial activity. In the second stage, the temperature decreased but the film continued to compost at a slower rate [19]. In the soil burial test pure PP showed almost no loss in weight. |
 |
| Surface morphology |
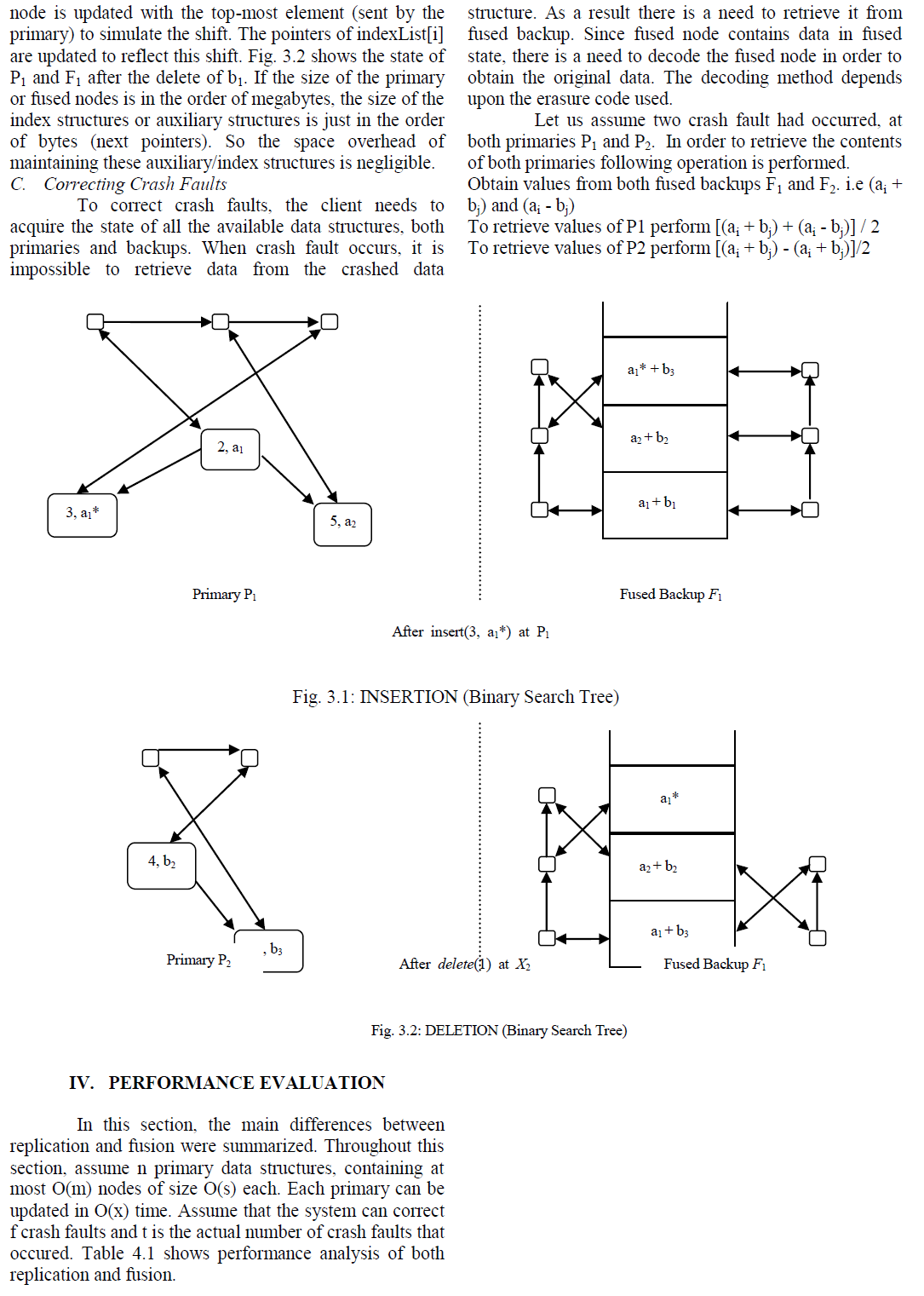
| Scanning electron microscopy (SEM) has been used widely to study the surface morphologies. Enhanced resolution of samples by SEM permits higher magnification probing of surfaces without loss in detail. In addition, due to the depth of the field of SEM, the micrographs retain a three- dimensional appearance of the samples. SEM was used to study the surface morphological changes after the biodegradation of the samples. Fig. 3 was used to exemplify the effect of soil on the PP/PS (80/20) blend. After soil burial the blend exhibited weight loss due to microbial growth on the starch. Micrographs [Fig. 3(a) and (c)] show continuous phase before biodegradation and [Fig. 3(b) and (d)] show cracks, voids and damages on the surface after biodegradation. This is a general phenomenon observed in otherthermoplastics filled with natural filler [22]. |
 |
| TEM Studies |
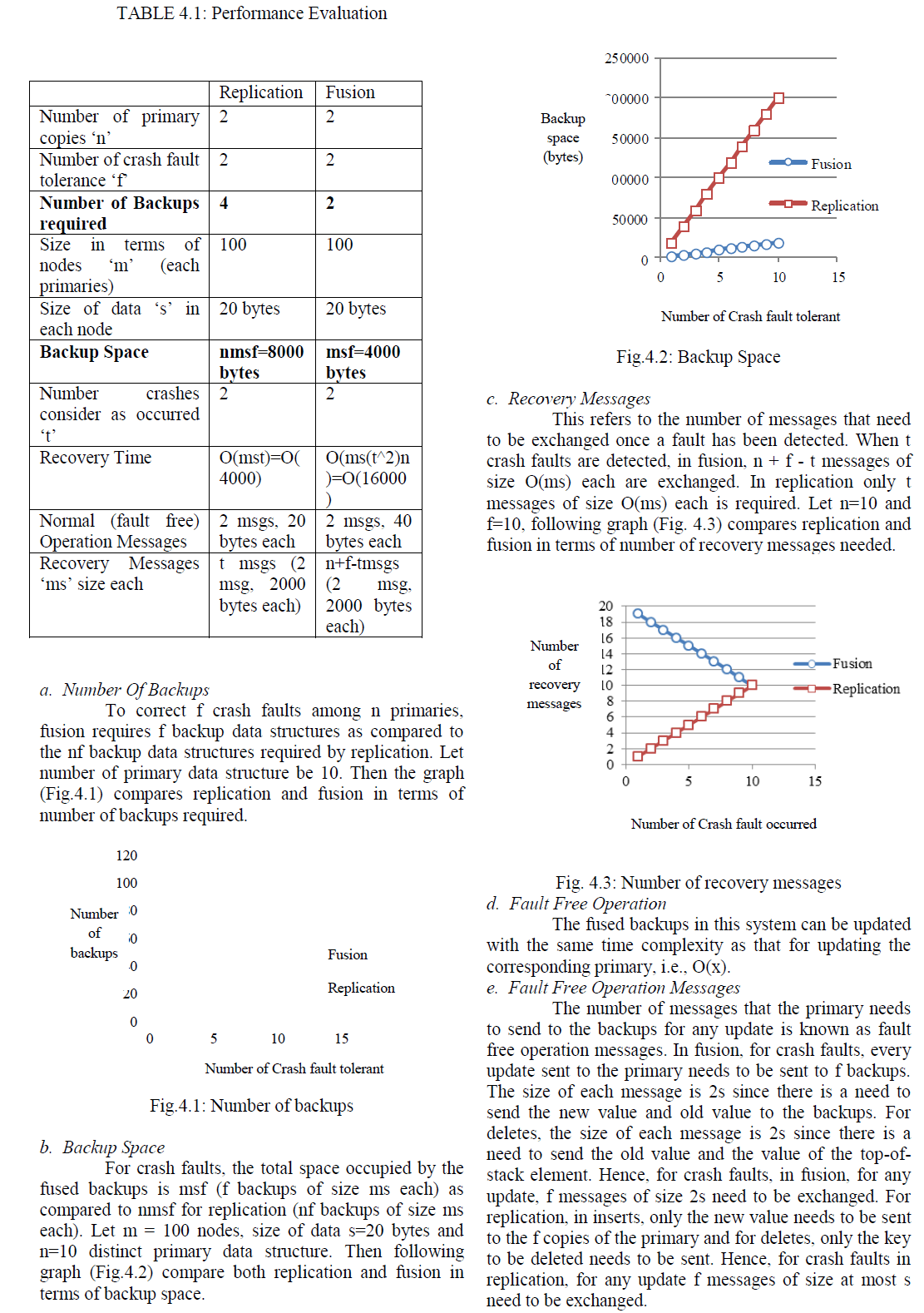
| Transmission electron microscopy (TEM) is a microscopy technique in which a beam of electrons is transmitted through an ultra-thin specimen, interacting with the specimen as it passes through. An image is formed from the interaction of the electrons transmitted through the specimen; the image is magnified and focused onto an imaging device, such as a fluorescent screen, on a layer of photographic film. In this experiment it was observed [Fig. 4(a) to 4(d)] that after biodegradation there is loss of starch content from the composite. This observation is also supported by other researchers for bio- composite biodegradation [22]. |
 |
 |
| Atomic Force Microscopic Studies |
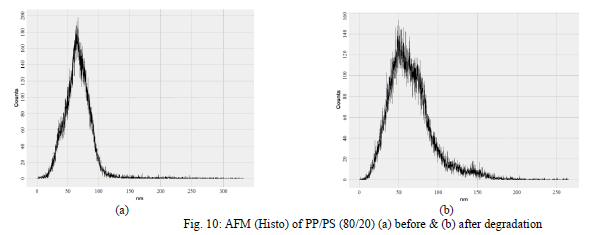
| Atomic Force Microscopic Studies (AFM) has been used widely to study the surface morphologies. In addition, due to the depth of the field of AFM, the micrographs retain a two- dimensional as well as three- dimensional appearance of the samples. AFM was used to study the surface morphological changes after the biodegradation of the samples. Fig. 5 to 10 was used to exemplify the effect of soil on the PP and PP/PS (80/20) blend. After soil burial the |
 |
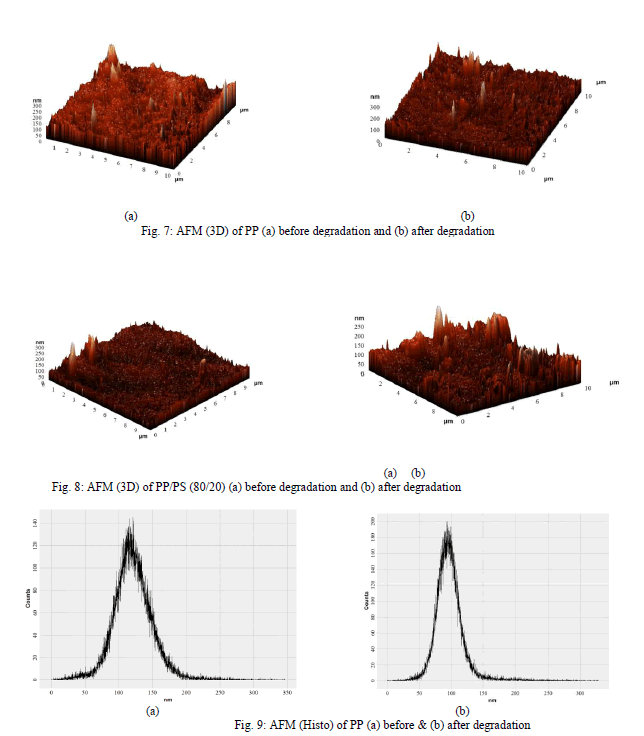 |
| blend exhibited weight loss due to microbial growth on the starch. It is shown in the [Fig. 6 (a) and 8 (a)] that there is continuous phase before biodegradation and [Fig. 6 (b) and 8 (b)] show damages on the surface after biodegradation where as in [Fig. 5 (a) and 7 (a)] and [Fig. 5 (b) and 7 (b)] there is no change for PP films in before and after biodegradation. For PP films after degradation counts are increasing and domain sizes are decreasing [Fig. 9 (a) and 9 (b)] but not much change in the roughness has been seen. For PP/PS films after degradation counts and domain size both are decreasing [Fig. 10 (a) and 10 (b)] and a change in the roughness has been seen. |
 |
| Crystallization Studies |
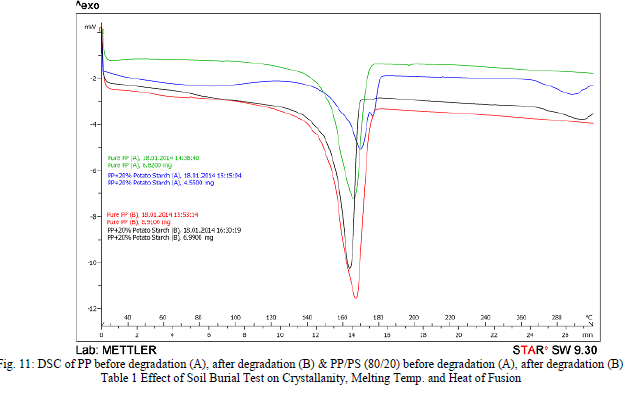
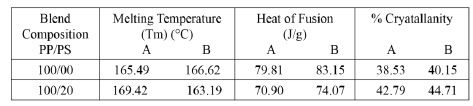
| Crystallanity is a state of molecular structure referring to a long-range periodic geometric pattern of atomic spacing. Cryatallanity can be calculated from a differential calorimetry curve by dividing the measured heat of fusion by the heat of fusion of 100% crystalline material. The percent crystallanity was calculated on the assumption that the heat of fusion of 100% crystalline PP is 207.1 J/g. As the heat of fusion is directly proportional to the amount of crystalline PP in the sample, it decreases as filler percentage increases. An apparent decrease in the heat of fusion [Fig. 11] was due to the decrease in the weight fraction of crystalline PP. Xc (% Crystallanity) = [ΔHf/wΔHf100] x 100 ΔHf = latent heat of fusion of the composite sample; ΔHf100 = latent heat of fusion of a PP with 100% crystallanity; w = weight fraction of PP in the composite. After 4 months of exposure to nursery soil environment, the biodegraded samples were again analyzed for crystallanity. The results obtained for PP and PP/PS blend are tabulated in Table 1. Heat of fusion and crystallinity increased after bio degradation. During biodegradation process, generally the amorphous fraction of material is exposed to microorganism attack. Therefore, the microbial degradation results in an increase in the overall degree of crystallanity of the sample. This is similar to the results obtained for most filled thermo- plastic and in agreement with that reported by researchers before, stating that the crystallanity of starch composite increased after biodegradation [19, 20]. |
 |
 |
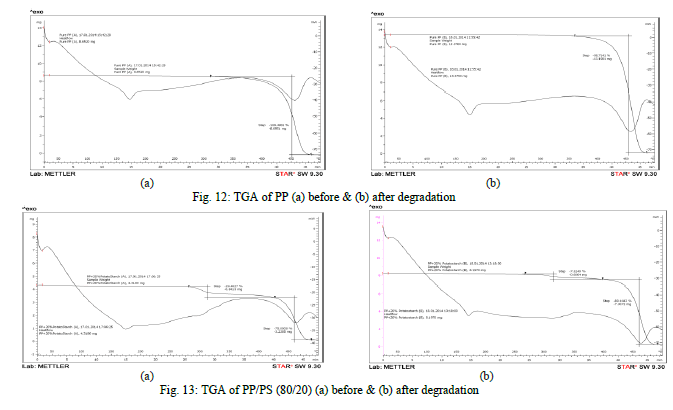
| Thermogravimetry analysis |
| The TGA curves of PP and PP/PS show that a considerable change has occurred in the thermal behavior of PP upon filler addition. A very distinct weight loss is seen [Fig. 13 (a) & 13 (b)] in PP/PS sample after bio-degradation in the temperature range of 250-350°C. The TGA curve of PP sample [Fig. 12 (a) & 12 (b)] shows no weight loss in the same temperature interval, suggesting weight loss is due to partial or complete pyro lytic volatilization of the starch in the blend. This loss in weight might therefore be used to quantify the starch content in the sample, or residual starch content after degradation. In PP/PS blend with starch content 20%, there is significant decrease in the amount of starch (about 12.16%) was observed after soil burial test from TGA data. |
 ` ` |
| Glass transition temperature (Tg) |
| It is used when describing temperature curves of polymers. At low temperatures, the polymer occurs in a glassy state (it appears as a rigid and brittle solid). With increasing temperature it is in the glass transition range which |
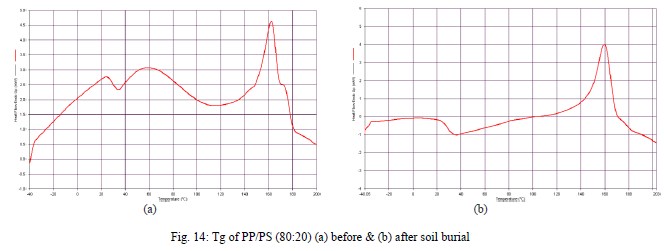 |
| is particularly important and is mostly specified in terms of just one temperature value which is referred to as glass transition temperature. It is assumed to be approximately in the middle of the temperature range in which the glass transition is taking place. In this range the molecules are showing more and more mobility and the polymer occurs in a soft-elastic, rubber-like state. For PP/PS films there is a change in Tg (-10°C) in the enthalpy value after degradation [Fig. 14.(a) and (b)] in the descending direction so it can be concluded that after degradation in the film as the starch is coming out so it would be easier for the flow of the material. |
| Fourier Transform Infrared Spectroscopic Studies |
| The FTIR spectra of PP film show characteristic C-H stretching bands within the 3000-2840 cm-1 region, mediumstrong C-H bending bands within the 1465-1450 cm-1 region and C-H out of plane bending within the 740-719 cm-1 region [Fig. 15(a) and (b)]. All the bands are also present in PP/PS (80/20) [Fig. 16 (a) to (b)]. The characteristic bands for starch in PP/PS blend films showed a broad O-H stretching absorbance in the 3600-3000 cm-1 region and strong set of C=O stretching band in the 1190-960 cm-1 region. |
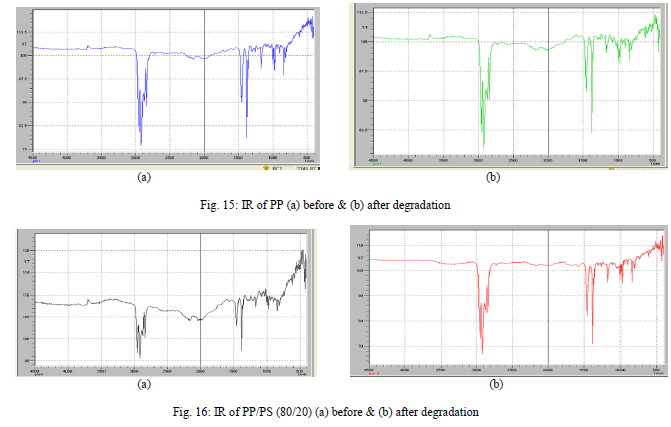 |
| The other bands independent of PP are the O-H bands though this band is unreliable for qualitative analysis due to their dependence on water content [22]. In the blends the 1640 cm-1 band, which is a measure of the water content, vary between degraded and undegraded samples. This could be attributed to a change in the absorbed water starch band as starch is removed rather than any change in the polymer matrix. A decrease in the O-H stretching band in the 3600- 3000 cm-1 region was also observed. The other significant trend that was observed in PP/PS blend before and after soil burial test was a slight change in the C-O stretching band (960-1190 cm-1) after degradation, indicating that the starch is preferentially removed, leaving the PP matrix [Fig. 16.(b)]. |
CONCLUSION |
| The blends containing a higher percentage of starch degraded rapidly in the first 30 days, during which the maximum starch content was accessed (10.6%). Over the next 4 months a gradual decrease of starch occurred. In morphological study has also showed cracks, voids and damages on the surface of the bio-composite after biodegradation. In DSC study, PP/PS blend with starch content 20%, there is significant decrease in the amount of starch (about 12.16%) was observed after soil burial test. Heat of fusion and crystallanity increased after bio degradation. During biodegradation process, generally the amorphous fraction of material is exposed to microorganism attack. Therefore, the microbial degradation results in an increase in the overall degree of crystallanity of the sample. In the AFM, TEM, SEM studies it was observed that after degradation there is changes in the morphology as well as surface roughness and it is also clear from IR that starch is coming out of the bio-composite. |
ACKNOWLEDGEMENTS |
| The authors are grateful to the Central Institute of Plastics Engineering and Technology, Ahmedabad for providing the extrusion and two roll mill machine facilities for film preparation. |
References |
|