ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
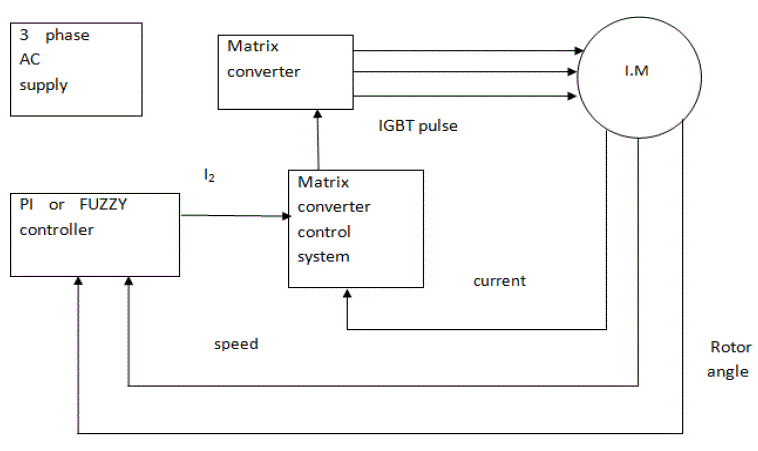
K. K. Bandyopadhyay1, Debarati Paul2*
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Qualitative studies of colour removal of distillery effluent were carried out continuously in packed bed reactor to screen out the suitable combination of modified soil. The continuous reactor (voidage: 0.39-0.4, packed density: around 1) studies enable to remove colour and COD degradation of effluent (COD 22000-23000 mg/l) to the tune of 97-99% respectively for the period of more than 10 days. Formulation of modified soil, kinetic data, voidage, space velocity etc are important parameters in assessing the efficiently of the reactor performance.
Keywords |
| Biological Oxygen Demand (BOD), Chemical Oxygen Demand (COD), Capillary Seepage System, Immobilized Cells, Lignocellulosic Derivative (LCD), Melanoidin |
INTRODUCTION |
| Molasses based distilleries are listed in the “Red category” industries as per the Ministry of Environment of Forests (MOEF) due to their high polluting potential [1]. They discharge huge volume of wastewater at the rate 8 – 15 lit of rectified spirit. The main problem in treating distillery effluent is its colour, which contains nearly 2% (w/w) of dark brown recalcitrant pigment, melanoidin and is highly resistant to microbial degradation and other biological treatments [2]. These compounds are also toxic to the existing microbial population in soil or water. The other associated problems are high Biological Oxygen Demand (B.O.D), Chemical Oxygen Demand (C.O.D), low pH and toxic substances such as phenols [3]. In 2003, Central Pollution Control Board (CPCB), the national agency responsible for environmental compliance, stipulated that distilleries should achieve zero discharge inland surface water courses by the end of 2005. Bioremediation of melanoidin containing waste water with white – rot fungi and their lignin degrading enzymes have been reported [4]. These ligninolytic enzymes are non - specific in their substrate requirement. The extremely wide range of degradative activity is dependent on the lignin – degrading enzymes, lignin peroxidase, manganese peroxidase, laccase and H2O2 producing enzymes [5]. |
| The proposed method involves a combination of physical and biological methods for adsorption on “modified soil” and simultaneous biodegradation of melanoidin by immobilized cells. This technique is novel to the best of our knowledge and can be extended to the field to the benefits of farmers. Objective of current study is to evolve a packed bed immobilized cell (in modified soil) reactor system to understand its impact on decolourisation and COD removal |
RELATED WORK |
| Bacterial systems have also been reported to decolourize and reduce C.O.D of the distillery spentwash [2, 6]. Physical methods of treatment involve adsorption through activated carbon or organic resin, however, high costs are involved. Biosorption, via live or dead microorganism of algae, yeast, bacteria and fungi has been used for this purpose [7-10]. The process of biosorption is reported to be governed by the type of melanoidin lipids, [11], pH and hydrophilicity [12]. Sonication is another physical method performed as a pre-oxidation step before biological treatment for increasing biodegradability [13, 14]. However its effect on decolourization of industrial effluents is not cost effective and practical. The available technologies to purify melanoidin containing effluents are highly energy intensive, expensive, land – oriented, unstable and no return on investment. So, today we need to develop a cost effective technology with some return on investment (ROI). |
MATERIALS AND METHODS |
| 1.Organism and Medium: A mixed culture population was isolated (unpublished report) from the soil of Uttar Pradesh and Maharashtra, India having high water retention capacity as well as base exchange capacity, by acclimatization with effluent. For medium preparation, 20% molasses solution was used as carbon source for cultivating the microbes. 2. Modified Soil Preparation: Natural Balarampur soil was mixed with 2 to 3 lignocellulosic derivatives (composition under patent) and traces of inorganic salts. This soil was named as ‘modified soil’ and physic-chemical properties of natural soil and ‘modified soil’ were determined before and after treating the effluent and photographed (for visual changes). |
| 3. Cell Immobilization: Prior to lab scale studies the column was packed with modified soil (Fig 1). The concentrated suspension of cells (melanoidin degrading isolates) was run continuously through the column for 2 days to activate the column and immobilize the cells on the ‘modified soil’. This was done in order to attain cell numbers in the range of 2.5-3 x 1010 per gram of modified soil. 4. Continuous Experiments: A mild steel column (1.1 m height, 5.25 cm internal diameter, having five different ports at 30,50,70,90 and 110 cm high) was employed for the studies. After secondary treatment, effluent of different COD values (9500-22000 mg/l), was passed through the column to maintain constant level in the column (Table 1). Samples were drawn at four hour intervals from the sampling ports to assess the colour removal and decrease in COD values in effluent samples. The residence time in the column was approx. 12 h. A steady state was assumed when the colour removal and COD degradation was achieved off as evidence by four successive samples assayed. Figure 1 shows a schematic diagram of the immobilized cell –modified soil column. 5.Batch Experiments: Qualitative studies of suspended cells were carried out in 500 ml conical flask containing 200 ml effluent (COD ranging 9500-22000 mg/l) inoculated with 10% inoculums and incubated at 30°C for six days. After incubation, decrease in COD values and decolorisation of effluent were computed. 6. Analytical Procedure: COD was measured as per the standard methods for the examination of water and wastewater; APHA (1995) [15]. |
 |
| Estimation of the colour intensity of effluent was carried out by diluting the sample with 0.1M acetate buffer solution, pH 6 after being centrifuged at 6000xg for fifteen minutes. The extent of colour removal of the diluted solution was measured spectrometrically at 475 nm. The percentage of colour removal was calculated as the colour intensity of decolorized sample against that of original effluent. The removal yield was expressed as the degree of decrease in the absorbance at 475 nm (pastel yellow colour) against the initial absorbance (dark brown colour) at the same wave length (Fig 2). This is further quantified from the standard curve drawn against COD and optical density of decolorized samples. Qualitative HPLC analysis was performed to determine decrease in melanoidin concentrations. Since the brown coloured samples were from effluent, presence of various impurities interfered the analysis and so qualitative data was not obtained. |
RESULTS AND DISCUSSION |
| 1. Batch Studies: The extent of COD reduction and colour removal of suspended cells are represented in Table- 2. From the table it is clear that for lower strength of effluent (COD 5000-5500 mg/l) although COD degradation is marginal (15-18%) but colour removal is approximately 45-50%. But in case of high strength effluent (20000-22000 mg/l) the COD reduction and colour removal were as low as 8-10% and 20% respectively. |
 |
| 2. Continuous Reactor Studies: |
| 2.1. Point to point analysis of colour removal and COD degradation along the height of column reactor: Point to point analysis of COD degradation and colour removal were monitored in the reactor employing immobilized modified soil with different strengths of effluent (COD 10000-22000 mg/l). The objective of this work was for a better understanding of the system at different sections of the reactor and its thorough analysis to establish a kinetic pattern. Fig 3 and 4 show that under the condition of operation (mentioned in the figure) around 77-79% of reactor volume was sufficient to attain COD degradation and colour removal to the extent of 82-92% and 92-96% respectively. Balance 20- 30% of reactor volume is essential as polishing reactor to attain final COD of seepage water in the range of 300-500 mg/l and colour removal of 97-99% depending on the strength of the effluent. |
| 2.2. Operational Stability: The reactor performance was found to be steady for more than 10 days and required no cell input. Fig 5 shows the pattern of colour removal to the extent of 97-98% constantly for the input effluent (COD 21000- 22000) fed to the reactor at 30°C. Continuous removal of colour might be due to combined effect of melanoidinase activity of cells (unpublished data) and adsorbing effect of some colour substances on modified soil. Microbial decolorisation of melanoidins may be due to successive decomposition mechanisms, that is the smaller molecular weight melanoidins are firstly attacked and the larger molecular weight melanoidin are finally attacked is supported from the work of [6]. |
 |
| 3. Soil quality |
| After undergoing column studies for 10 days the reactor operation was stopped and soil was removed from the column, dried and soil quality was monitored. Figure 6 shows the difference between ‘modified soil’ and natural soil after the treatment process. The report has been tabulated (Table 3). From the data it is clear that the soil quality has improved in terms of electrical conductivity, phosphorous and potash. The improvement in quality of soil would enhance crop yield when data is translated at commercial level. |
 |
 |
CONCLUSION |
| In recent years considerable interest in removal of melanoidin pigments present in various types of effluents has stipulated several kind of research on improving clean environment and eco-system. Continuous decolourisation of distillery effluent using immobilized whole cells is one of them. In the present study, it was possible to remove colour and COD of effluent to the extent of 97-99% and 95-97% respectively for a period of approximately a little over ten days through whole cell immobilization on ‘modified’ soil by capillary seepage system. The system will greatly enhance soil quality. |
ACKNOWLEDGEMENT |
| Authors express their deep sense of gratitude to Founder President, Dr. A.K. Chauhan, Amity University, Uttar Pradesh for his continuous encouragement throughout the period of investigation. We are also indebted to Balarampur Chini Mills for their help and assistance. |
References |
|