ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Seema J Patel1, Jyothi2, Soumya S R3, Apoorva4, Amrin Banu5
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Growing evidence of the role of free radicals and antioxidants in health and ageing has focused great interest on polyphenols. Antioxidant activity of polyphenols in wine samples of different stages of fermentation was compared with that of citrus fruit juices like lemon, lime and orange juice. Total phenolic content of the samples was determined. It was observed that red wine sample showed highest phenolic content and good radical scavenging activity. It also showed antimicrobial activity against Streptococcus epidermidis (acne inducing bacteria) thus confirming the antiacne activity.
Keywords |
| antioxidant, antiacne activity, ABTS, polyphenols, wine |
INTRODUCTION |
| Anti-oxidants are the compounds which can inhibit or delay the oxidation of an oxidizable substrate in a chain reaction triggered by free radicals, seem to be very important in the prevention of diseases. Free radicals, reactive oxygen species (ROS) and reactive nitrogen species (RNS) are associated with many pathological conditions such as atherosclerosis, arthritis, ischemia, reperfusion injury of many tissues, central nervous system injury, gastritis, cancer and AIDS [7]. Synthetic antioxidants like butylatedhydroxy anisole (BHA, butylated hydroxyl toluene (BHT), tertiary butylatedhydroxyquinone and gallic acid esters have been suspected to be carcinogenic. Hence, strong limitations have been placed on their use and there is a trend to replace them with naturally occurring antioxidants. Moreover, these synthetic antioxidants also show low solubility and moderate antioxidant activity [1]. Hence, search for natural antioxidant has greatly been increased in the recent scenario[5]. In the literature many crude extracts and pure natural compounds have been reported which have potent antioxidant potential [2]. However there is still a need to find out more effective antioxidant having fewer side effects from natural source. |
| In the past years the analysis of these compounds in different food-stuffs; such as wine, fruits and tea has attracted the interest of the researchers due to their beneficial effects. Wine is rich in antioxidant compounds and their beneficial effects may be in part explained by the presence of phenolic compounds exhibiting different biological activities. |
| Citrus fruits and juices are an important source of bioactive compounds including antioxidants such as ascorbic acid, flavonoids, phenolic compounds and pectins that are important to human nutrition[4, 6]. Epidemiological studies on dietary Citrus flavonoids improved a reduction in risk of coronary heart disease [3]. |
| Acne vulgaris is a most common skin disorder of Pilosebaceous unit. That affects areas containing the largest oil glands, including the face, back, and trunk. It is generally characterized by formation of seborrhea, comedone, inflammatory lesions and presence of bacteria Propionibacterium acnes, Staphylococcus epidermidis and Malassezia furfur in the follicular canal and sebum production. Staphylococcus epidermidis, an aerobic organism, usually involves in superficial infections within the sebaceous unit [10]. These factors provide a potential target for treatment. So compounds inhibiting its growth will be showing antiacne activity. |
| In this sense, the main objective of this work was to determine the total content of phenolic compounds in different wines produced by simple fermentation in laboratory and to determine the antioxidant capacity of these samples compared with that of citrus fruit juices (rich in vitamin C). The work also included testing of antiacne activity of these samples. |
II. MATERIALS AND METHODS |
| Wine Samples: Red wine and white wine were prepared using black grapes and green grapes respectively by fermentation carried out in the lab. Samples were taken at different stages (like after 21, 42, 63 and 84 days of fermentation) for the qualitative and quantitative studies. |
| Fruit juices: Lemon, lime and orange extracts were prepared and used. |
| The samples of wine and fruit juices were qualitatively tested for the presence of various phytochemical constituents, using standard procedures to identify the constituents like alkaloids, steroids, flavonoids, tannins, saponins, phenols and anthroquinone[8, 9]. |
| Ethanol content [14], sugar content and total acidity parameters in the samples were tested[15]. |
Determination of total polyphenol content (TPC) |
| The total phenolic concentration was determined spectrophotometrically according to the Folin-Ciocalteu (FC) colorimetric method. The TPC was calculated from a calibration curve using gallic acid as a standard (50-500 mg/L). Data was expressed as mg of gallic acid equivalent (GAE), averaged from three measurements [11]. |
Measurement of the antioxidant capacity |
Determination of ABTS radical scavenging activity |
| The antioxidant capacity of wine samples was determined by ABTS assay. This method is based on the inhibition of the absorbance of the radical cation of 2,2´-azinobis (3-ethylbenzothiazoline 6-sulfonate)ABTS•+, which has a characteristic long-wavelength absorption spectrum showing a maximum at 734 nm. Results were compared with a standard curve prepared with different concentrations of Trolox, a water-soluble analogue of vitamin E, and were expressed as milimolarTrolox equivalents [12]. |
Determination of total anti-oxidant capacity: |
| This method is based on the reduction of Mo (VI) to Mo (V) present in the reagent mixture consisting of ammonium molybdate, sodium phosphate and sulfuric acid by the anti-oxidant compound and formation of a green phosphate/ Mo (V) complex, which has maximum absorption at 695 nm. The total anti-oxidant capacity is expressed as the number of equivalents of ascorbic acid [13]. |
Determination of invitroantiacne activity using agar well diffusion method: |
| Acne is one of the common skin disorder and the most popular dermatological complaint. Staphylococcus epidermidis has been recognized as pus-forming bacteria triggering an inflammation in acne. Thus the antiacne activity of samples was determined by antimicrobial activity of the samples against Staphylococcus epidermidisby agar well diffusion method. The inoculum of Staphylococcus epidermidis was swabbed on nutrient agar plates and samples were taken in wells. Inhibition zones around each sample well were measured after incubation [16]. |
III. RESULTS AND DISCUSSION |
| Table 1 shows the results of qualitative phytochemical analysis of the samples to detect the presence of alkaloids, steroids, flavonoids, tannins, saponins, phenols and anthroquinone. Through qualitative phytochemical analysis it was observed that flavonoids, phenols and tannins were observed in all the samples. |
 |
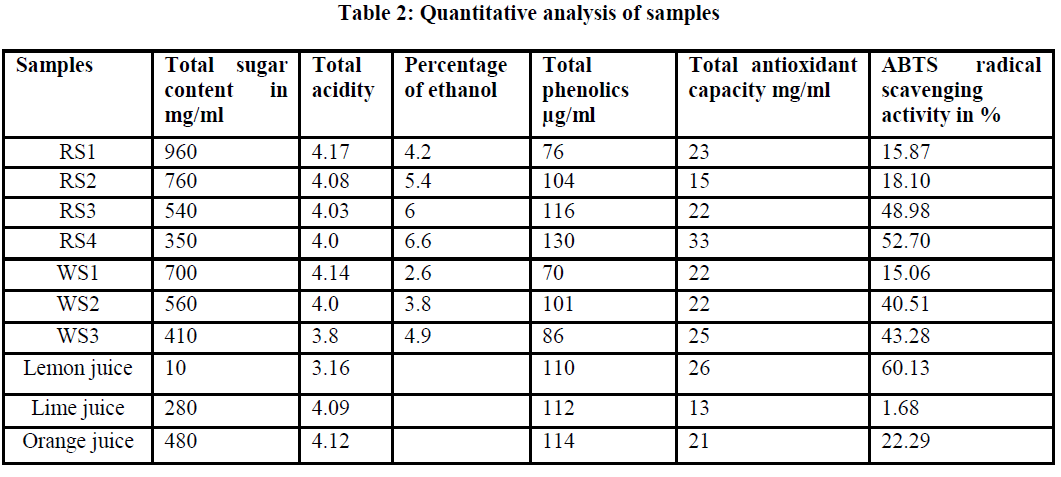
| Phenolic compounds are responsible for the antioxidant activity of wines. The concentration of phenolics, estimated as the Total Phenol Content is shown in Table 2 and the results are expressed in gallic acid equivalents (GAE). The total polyphenol concentration was found to vary between 76μg/ml (Sample 1) and 130μg/ml (Sample 2). Our results confirm a variation in phenolic content among wine samples tested and these data showed that the red wine sample had the highest total phenolic content. |
| The total antioxidant capacity of red wine samples was determined by the bleaching of pre-formed ABTS radical cations and as ascorbic acid equivalents. Table 2 shows that red wines studied presented a substantial antioxidant capacity. |
 |
| The compounds present in wine and fruit juices found to inhibit the growth of Staphylococcus epidermidis which was indicated as a zone of inhibition.Table 3 shows the inhibition zone diameter. Lemon juice showed maximum zone of inhibition followed by red wine sample and it can be compared with inhibition zone obtained from ethanol as control. |
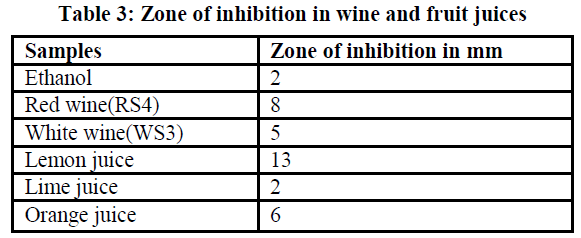 |
| Although lemon juice is showing maximum inhibition zone (Fig1) but it has higher acidity and it is the concentrated extract taken which will be diluted while consuming and also only fresh juice had shown good results. Red wine sample has comparatively less acidity and good antimicrobial property according to the results obtained. It was observed that the older the wine the better was the antioxidant property showing the effect of fermentation process on the antioxidant property of the product. Staphylococcus epidermidis have been recognized as pus-forming bacteria triggering an inflammation in acne, so the inhibitory effect of wine on that shows the antiacne property of wine. |
 |
IV. CONCLUSION |
| Red wine samples showed highest polyphenolic content. Substantial antioxidant and antiacne property shown by red wine through above results can be attributed to its polyphenolic content. These activities of red wine and also it being a natural product can make it very good in skin treatments. |
References |
|