ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
M. Abdallah1,2,O.A.Hazazi1, B.A.ALJahdaly1, A. S. Fouda3, W. El-Nagar3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The corrosion behaviour of Zinc in 2M sodium hydroxide solution and its inhibition by some coumarin derivatives was studied using weight loss and galvanostatic polarization techniques. Addition of Ba2+ ion to alkaline medium containing the coumarin derivatives increases the inhibition efficiency of the system. The obtained results showed that the inhibition efficiency of these compounds increased by increasing their concentrations and decreased by raising the temperature, so that the adsorption of these compounds is physically adsorbed on the zinc surface. The adsorption of the inhibitors on the zinc surface in alkaline solution was found to obey Temkin adsorption isotherm. The activation energy and some activated thermodynamic parameters for the corrosion process were calculated and discussed. The polarization technique indicates that these compounds act as a mixed type inhibitors but cathode is more polarized when an external current was applied. The results were explained on the basis of the molecular structure and the substituent groups and their charge densities.
Keywords |
| Coumarin, Zinc, Sodium hydroxide, Corrosion, Inhibitors |
I. INTRODUCTION |
| Zinc is a very important metal used in a wide range of applications by various industries, e.g.as a construction material for roofs and facades in structural and civil engineering or as a sacrificial coating to protect iron and steel producing and processing industry, especially the automotive industry [1]. Zinc is a very active metal so that it corrodes quickly in aqueous solutions. For these reasons the industrial use of corrosion inhibitors is often a good solution to prevent corrosion phenomena and to provide a more acceptable life time of metallic structure [2,3].Most of the reported corrosion inhibitors are organic compounds containing hetro cyclic atoms (nitrogen ,oxygen, sulfur and phosphorous),aromatic ring or triple bonds are commonly used to reduced the corrosion attack of zinc in aqueous solutions[4-13] .The inhibition effect of organic compounds arises from the adsorption on the metal surface .The adsorption properties depend on the structure of the organic compounds, such as functional groups, steric factors, and aromaticity. |
| In previous work, the inhibiting effect of some coumarin derivatives toward the corrosion of Zinc in 0.1M HCl solution was studied[14]. The aim of the present work is to study the inhibiting effect of 4-coumarin derivatives toward the corrosion of zinc in NaOH solution using weight loss method and galvanostatic polarization method. Investigate the synergistic effect of Ba2+ions on the corrosion inhibition of the investigated 4- coumarin derivatives in alkaline medium. The effect of the temperature on the dissolution of Zn in free and inhibited alkaline solution was also studied and some activated thermodynamic parameters are computed. |
II. EXPERIMENTAL METHODS |
| Corrosion tests were performed using Zn electrode has the following chemical composition: 0.001 wt% Pb,0.002 wt% Fe , 0.001 wt% Cd, 0.001 wt% Cu and the rest is Zn. |
| For weight loss measurements, the reaction basin used in this method was a graduated glass vessel having a total volume of 100 ml. 100 ml of the test solution was employed in each experiment. The test pieces were 20 x 20 x 2 mm. The samples were first mechanically polished with a fine grade emery paper in order to obtain a smooth surface, followed by ultra-sonically degreasing with acetone and then rinsed with distilled water, dried between two filter papers and weighed. The test pieces were suspended by suitable glass hooks at the edge of the basin, and under the surface of the test solution by about 1 cm. |
| Weight loss measurements were carried out as described elsewhere [15]. The percentage inhibition efficiency (% IE) and a parameter (θ) which represents the part of the metal surface covered by the inhibitor molecules were calculated using the following equations: |
 |
| where, Wfree and Wadd are the weight losses of Zn sample in free and inhibited acid solution, respectively. |
| For galvanostatic polarization measurements, a cylindrical rod embedded in araldite with exposed surface of 0.5 cm2 was used. The electrode was polished with different grades emery paper, degreased with acetone and rinsed by distilled water. Galvanostatic polarization studies were carried out using zinc rod of the same compositions used in weight loss. E vs. log I curves were recorded at temperature 30oC. A constant quantity of the test solution (100 ml) was taken in the polarization cell. Galvanostatic polarization studies were carried out using EG&G model173 Potentiostat/Galvanostat. For accurate measurements of potential and current density. Three compartment cell with a saturated reference calomel electrode and a platinum foil auxiliary electrode was used .Solutions were not deaerated to make the conditions identical to weight loss measurements. All the experiments were carried out at 30ïÃâñ oC by using ultra circulating thermostat. |
| The percentage inhibition efficiency (% IE) was calculated from corrosion current density values using the equation |
 |
| where Ifree and Iadd are the corrosion current densities in absence and presence of inhibitors, respectively. |
| Chemical structure of coumarin derivatives |
 |
III.RESULTS AND DISCUSSION |
| III.1. Weight - loss measurements |
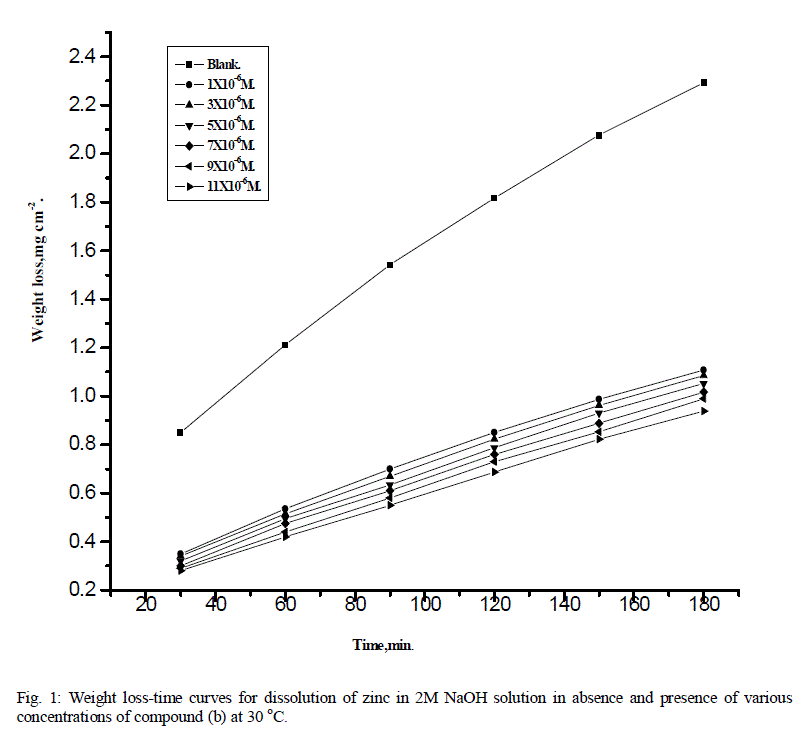
| Figure1 shows the weight loss-time curves for zinc in 2M NaOH solution in absence and presence of different concentrations of compound (b). Similar curves were obtained for other two compounds (not shown).Inspection of this figure, by increasing the concentration of these derivatives, the weight loss of zinc samples are decreased. This means that the presence of these derivatives retards the corrosion of zinc in 2M NaOH or in other words, these compounds act as inhibitors. The linear variation of weight loss with time in uninhibited and inhibited 2M NaOH indicates that the absence of insoluble surface films during corrosion. In the absence of any surface films, the inhibitors are first adsorbed on to the metal surface and thereafter impede the corrosion either by merely blocking the reaction sites (anodic and cathodic) or by altering the mechanism of the anodic and cathodic partial processes. |
| The inhibition efficiency (%I) were calculated using the equation 1and given in Table1.The order of the percentage inhibition efficiency of 4-coumarin derivatives decreases in the following order: b > c > a |
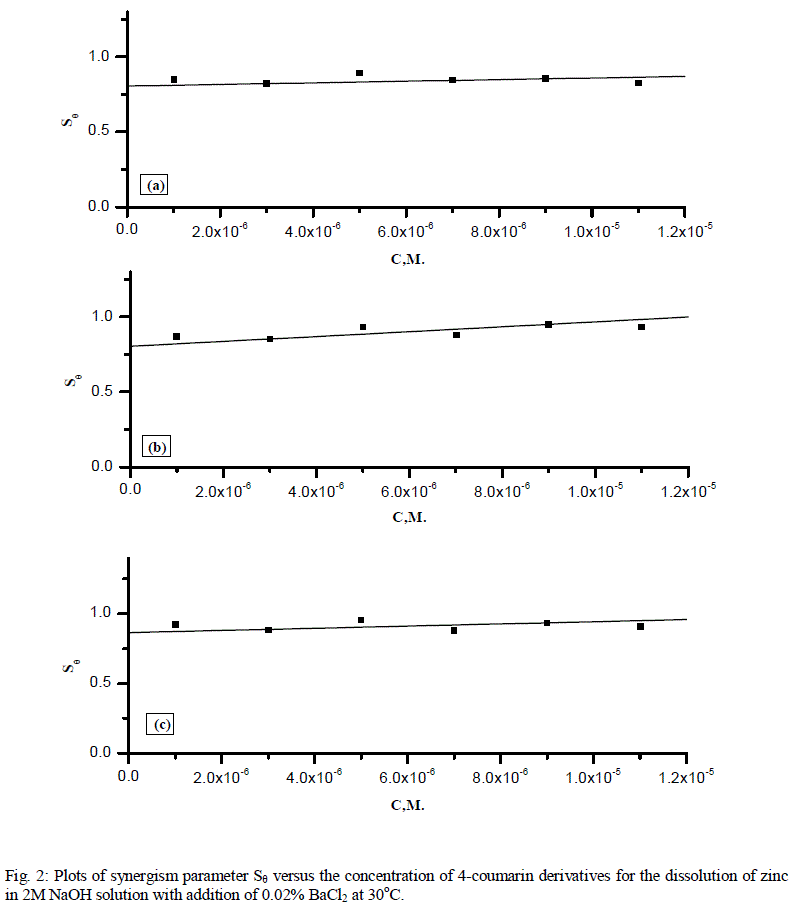 |
| Fig. 1: Weight loss-time curves for dissolution of zinc in 2M NaOH solution in absence and presence of various concentrations of compound (b) at 30 oC. |
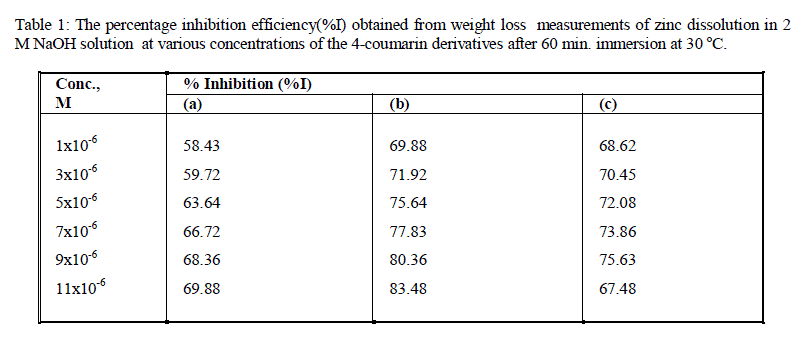 |
| III.2. Synergistic effect of BaCl2 |
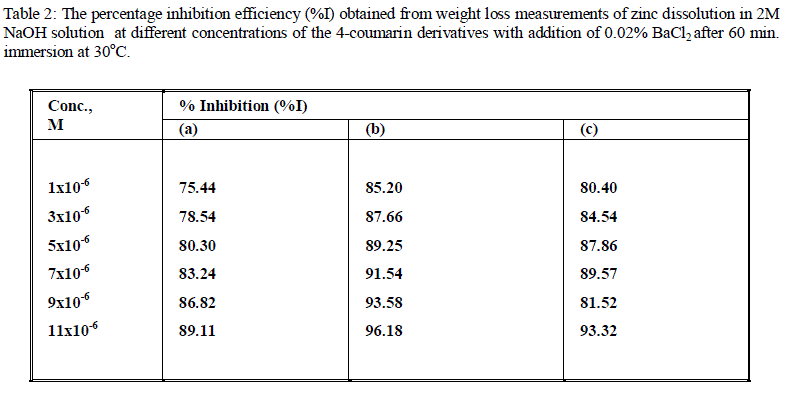
| The effect of addition of 0.02%BaCl2 on the corrosion inhibition of zinc in 2M NaOH solution in presence and absence of the coumarin derivatives after 60 min. immersion at 30oC was studied by the weight loss method. Similar curves to Fig. 1 are obtained (not shown).The values of inhibition efficiency (% I) for various concentrations of inhibitors in the presence of Ba+2are given in Table 2. It is observed that, %I of the inhibitors increases with increasing the concentrations of 4_coumarin derivatives, also the order of inhibition efficiency decreases in the following order : (b) > (c) > (a). |
| The strong chemisorptions of these cations on the metal surface is responsible for the synergistic effect of these ion leads to greater surface coverage and, therefore, greater inhibition. |
 |
| The synergistic inhibition effect was evaluated using a parameter, Sθ, obtained from the surface coverage values (θ) of the anion, cation and both. Aramaki and Hackerman [16] calculated the synergism parameter Sθ using the following equation. |
| Sθ = 1-θ1+2 / 1-θ'1+2 (4) |
| where: |
| θ1+2 = (θ1 + θ2) – (θ1θ2); |
| θ1 = surface coverage by anion; |
| θ2 = surface coverage by cation; |
| θ'1+2 = measured surface coverage by both the anion and cation. |
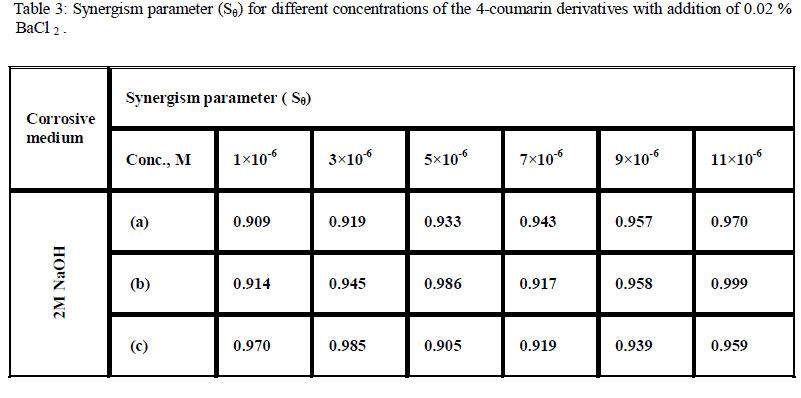
| We calculate synergism parameters from the above equation. The plot of the synergism parameter (Sθ) against various concentrations of 4-coumarin derivatives given in Fig. 2 and the corresponding values are shown in Table 3. As can be seen from this Table, values of Sθ are nearly equal to unity, which suggests that the enhanced inhibition efficiencies caused by the addition of these cations to 4-coumarin derivatives is due mainly to the synergistic effect. as following: |
| (b) > (c) > (a). |
| Synergistic adsorption is classified into two types plus a mixture of the two: (i) specific co- adsorption of anions and cations, and (ii) ionic or physical overlap adsorption of cations over the anion covered zinc surface. |
| From previous results it is known that BaCl2 would be considered as one of the effective anions for synergistic action. The net increment of inhibition efficiency shows a synergistic effect of Ba2+ ions with 4-coumarin derivatives. |
 |
 |
| III.3. Adsorption isotherms |
| 4-coumarin derivatives inhibit the corrosion of zinc in 2 M NaOH solution by the adsorption on metal surface. Theoretically, the adsorption process can be regarded as a single substitutionally process in which an inhibitor molecule, Inh., in the aqueous phase substitutes an "x" number of water molecules adsorbed on the metal surface [17] vis, |
| Inh(aq) + xH2O(sur) Inh.(sur) + xH2O(aq) (5) |
| where, x is known as the size ratio and simply equals the number of adsorbed water molecules replaced by a single inhibitor molecule. The adsorption depends on the structure of the inhibitor, the type of the metal and the nature of its surface, the nature of the corrosion medium and its pH value, the temperature and the electrochemical potential of the metal-solution interface. Also, the adsorption provides information about the interaction among the adsorbed molecules themselves as well as their interaction with the metal surface. Actually an adsorbed molecule may make the surface more difficult or less difficult for another molecule to become attached to a neighbouring site and multilayer adsorption may take place. There may be more or less than one inhibitor molecule per surface site. Finally, various surface sites could have varying degrees of activation. For these reasons a number of mathematical adsorption isotherm expressions have been developed to take into consideration some of non-ideal effects. |
| Adsorption isotherm equations are generally of the form [18]. |
| f (θ, x) exp (-a, θ) = KC (6) |
| where, f (θ, x) is the configurationally factor that depends essentially on the physical model and assumptions underlying the derivation of the isotherm a is a molecular interaction parameter depending upon molecular interactions in the adsorption layer and the degree of heterogeneity of the surface. All adsorption expressions include the equilibrium constant of the adsorption process, K, which is related to the standard free energy of adsorption (ΔG°ads.) by: |
| K = 1/55.5 exp-ΔG°ads. / RT (7) |
| where, R is the universal gas constant and T is the absolute temperature. |
| A number of mathematical relationships for the adsorption isotherms have been suggested to fit the experiment data of the present work. The simplest equation is that due to Temkin isotherm according to the following equation: |
| In K C =a θ (8) |
| where, K is the equilibrium constant of the adsorption, C is the inhibitor concentration in the bulk of the solution, a is the interaction parameter and θ is the surface coverage. The surface coverage, i.e, the fraction of the surface covered by the inhibitor molecules. |
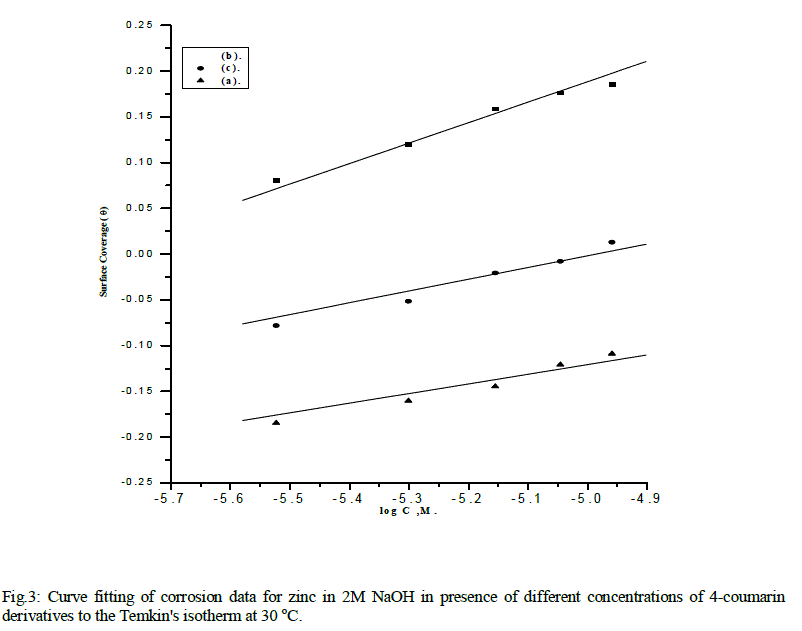
| Plots of θ vs. log C (Temkin plots) for adsorption of 4-coumarin derivatives on the surface of zinc in 2M Sodium Hydroxide at 30 °C is shown in Fig.3. The data gave straight lines indicating that Temkin isotherm is valid for these systems. Temkin's isotherm is applied for ideal case of physical and chemical adsorption on a smooth surface with no interaction between the adsorbed molecules. |
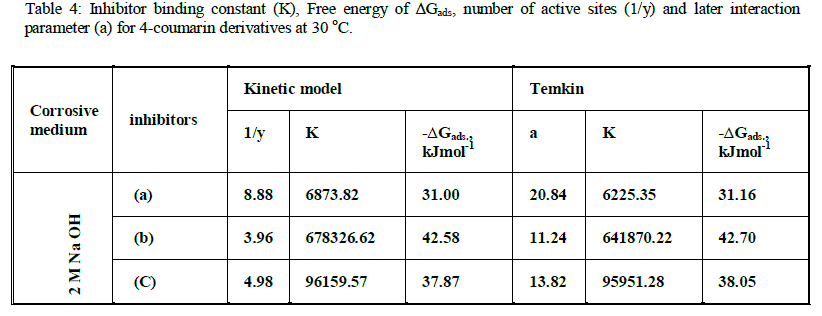
| On the other hand, it is found that the kinetic-thermodynamic model of El Awady et al [19] |
| log θ / (1-θ) = log K' + y log C (9) |
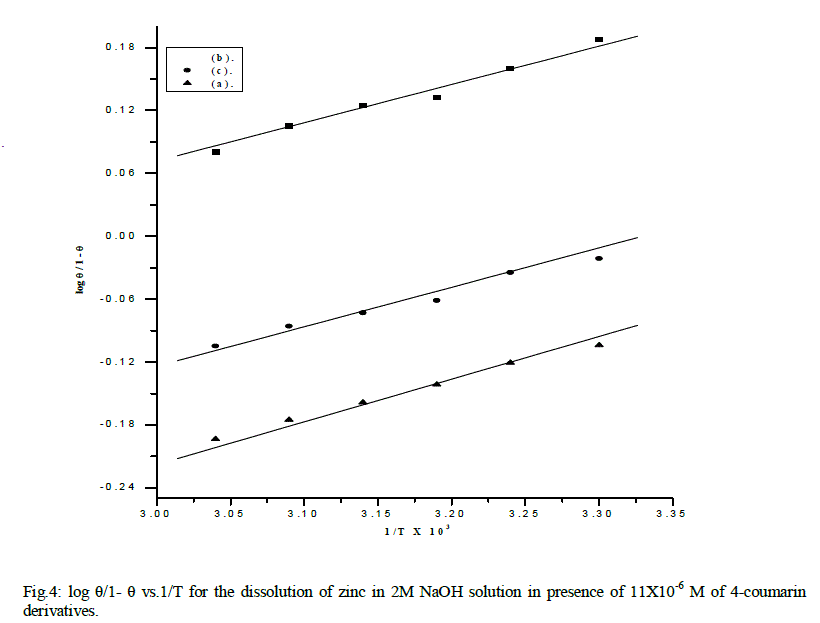
| is valid to operate the present adsorption data. The equilibrium constant of adsorption K=K' (1/y), where 1/y is the number of the surface active sites occupied by one 4-coumarin molecule and C is the bulk concentration of the inhibitor. Plotting log θ/(1-θ) against log C at 30 °C is given in Fig. 4 .Straight lines relationships were obtained suggesting the validity of this model for all cases studied. The calculated values of 1/y, K and ΔG°ads. are given in Table 4. Inspection of the data of this Table shows that the large values of ΔG°ads. and its negative sign, indicate that the adsorption of 4-coumarin compounds on the zinc surface is proceeding spontaneously and is accompanied by a highly-efficient adsorption. It is worth noting that the value of 1/y is more than unity. This means that the given inhibitor molecules will occupy more than one active site. In general, the values of ΔG°ads. obtained from El-Awady et al model are comparable with those obtained from Temkin isotherms. |
 |
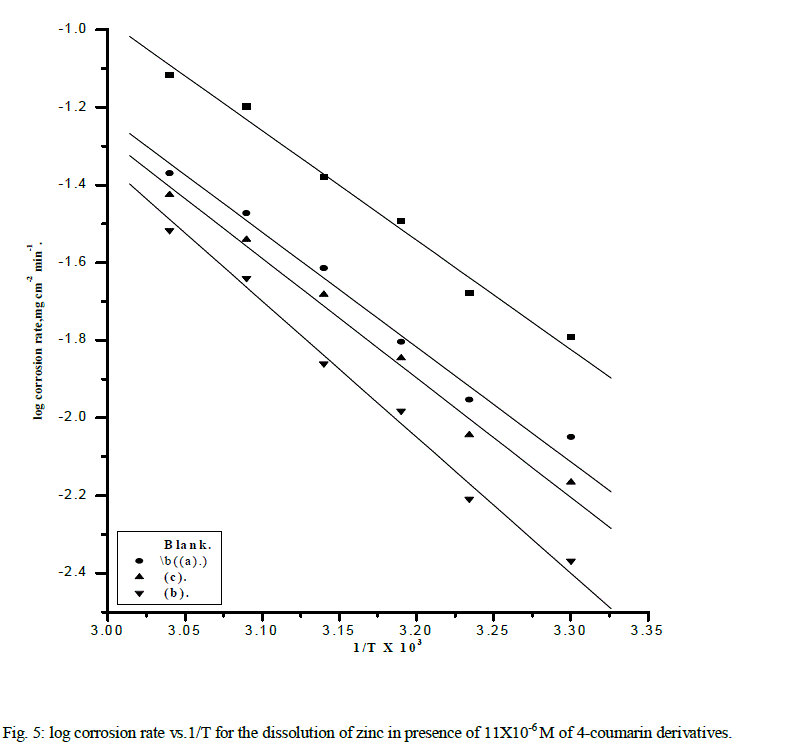 |
 |
| III.4. Effect of temperature |
| The effect of rising temperature on the rate of dissolution of Zn in 2M NaOH solution containing11x10-6 M of three compounds of 4-coumarin derivatives was studied by weight loss measurements over a temperature range from 35 to 55 oC. Similar curves to Fig .1were obtained (not shown). |
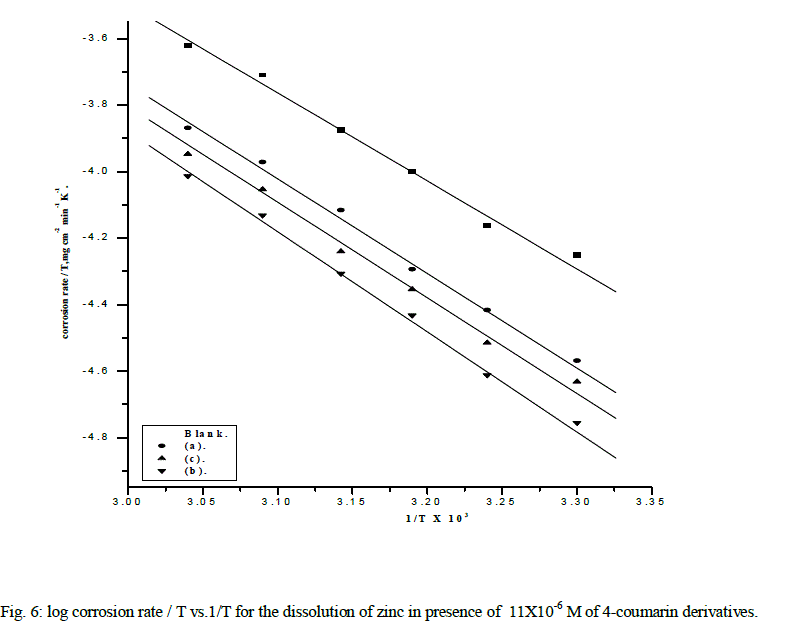
| The apparent activation energy Ea*, the enthalpy of activation ΔH* and the entropy of activation ΔS* for the corrosion of zinc samples in 2M NaOH solution containing 11x10-6 M concentrations of 4-coumarin derivatives were calculated from Arrhenius-type equation: |
| Rate = A exp (-Ea*/RT) (10) |
| and transition-state equation: |
| where A is the frequency factor, and R is the universal gas constant ,T is absolute temperature, h is Plank's constant, and N is Avogadro's number. Fig. 5 represents Arrhenius plot (log Rate vs. 1/T ) for uninhibited and inhibited Zinc in 2M NaOH solution containing containing11x10-6 M of the studied compounds. The values of Ea can be calculated from the slope of the straight lines and given in Table 5. The increase of the activation energies in the presence of inhibitors is attributed to an appreciable decrease in the adsorption process of the inhibitors on the metal surface with increase of temperature and a corresponding increase in the reaction rate because of the greater area of the metal that is exposed. |
| Fig.6 represents a plot a log (Rate/T) vs. 1/T for uninhibited and inhibited Zinc in 2M NaOH solution containing 11x10-6 M of the studied compounds give straight lines with slope of - ΔH*/2.303R, and intercept is equal to log R/Nh +ΔS*/2.303R .The values of ΔH* and ΔS* are calculated and listed in Table 5.. The positive values of ΔH* reflect a strong chemisorptions of the inhibitor on the surface. The values of entropy of activation ΔS* in the absence and in the presence of the studied coumarin compounds are negative. This implies that the activated complex in the rate determining step represents an association rather than a dissociation step [20]. This means that the activated molecules were in higher order state than that at the initial stage. |
 |
 |
 |
| III.5. Galvanostatic polarization |
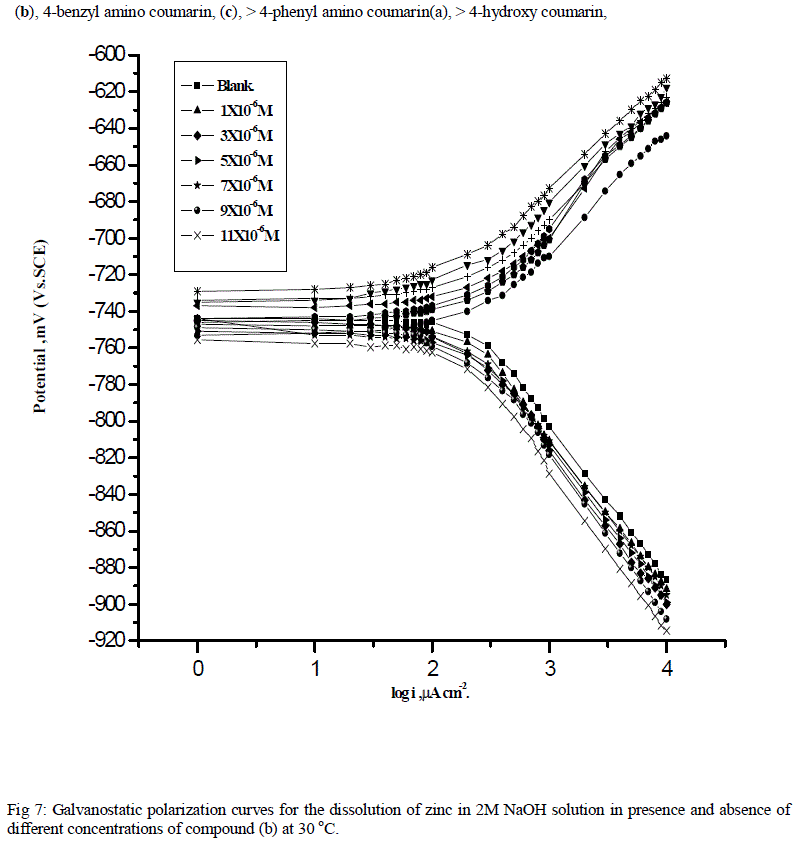
| Fig.7 represents the galvanostatic polarization curves of zinc electrode in 2M NaOH solution in absence and presence of various concentrations of compound (b) as an example of the tested 4-coumarin derivative. Similar curves were obtained for other two compounds (not shown). Corrosion parameters such as corrosion current density (icorr.), corrosion potential (Ecorr.), Tafel slopes (βa and βc), degree of surface coverage (θ) and inhibition efficiency (%I) were determined and are given in Table 6. Inspection of this Table, it is obvious that. |
| a) The anodic (ïÃÂâa) and cathodic (ïÃÂâc) Tafel slopes are approximately constant suggesting the inhibiting action of these compounds acted by adsorption at the metal surface according to blocking adsorption mechanism. These compounds decrease the surface area available for anodic dissolution and cathodic hydrogen evolution reaction without affecting the reaction mechanism. |
| b) The values of Ecorr. change slowly to less negative values and the value of Icorr. decrease and hence the values of IE's increases indicating the inhibiting effect of these compounds. |
| c) The values of IE's of the three tested compounds decrease in the following order: b > c > a. |
| The order of the inhibition efficiency is in a good agreement with that obtained from weight loss measurements. |
| III.6.Chemical Structure of Inhibitors and Corrosion Inhibition |
| Inhibition of the corrosion of zinc in 2M NaOH solution by some 4-coumarin derivatives was studied using weight loss and galvanostatic polarization measurements, The observed corrosion date were obtained as the concentration of inhibitors increases, namely, |
| i- The linear variation of weight loss with time. |
| ii-The decrease of weight loss . |
| iv. The parallel shift in Tafel lines to higher potential values. and |
| v. The corrosion current density decreases |
| The mechanism of the inhibition processes of the corrosion inhibitors under consideration is mainly the adsorption one The adsorption process is governed by different parameters that mostly depend on the chemical structure of these inhibitors, the number of adsorption active centers in the molecule and their charge density, molecular size, mode of adsorption, heat of hydrogenation and formation of metallic complexes. The percentage inhibition efficiency of the three studied coumarin derivatives toward the corrosion of Zn in 2MNaOH solution are in the order: |
| (b), 4-benzyl amino coumarin, (c), > 4-phenyl amino coumarin(a), > 4-hydroxy coumarin, |
 |
 |
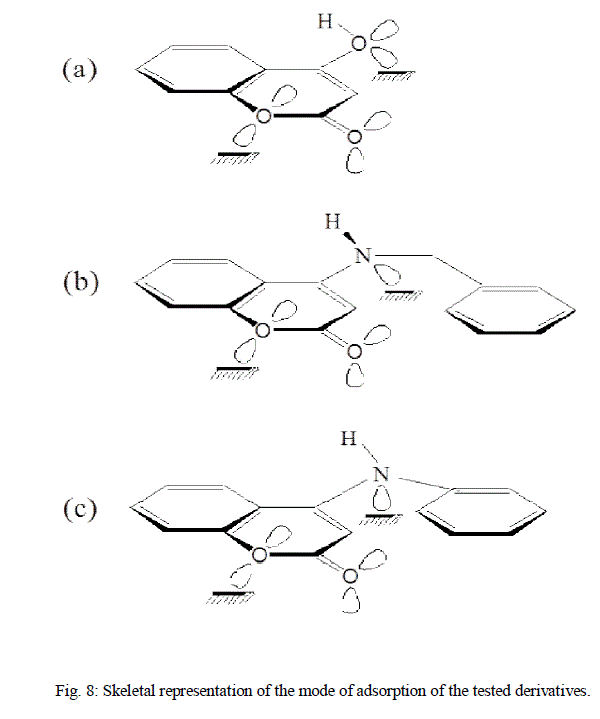
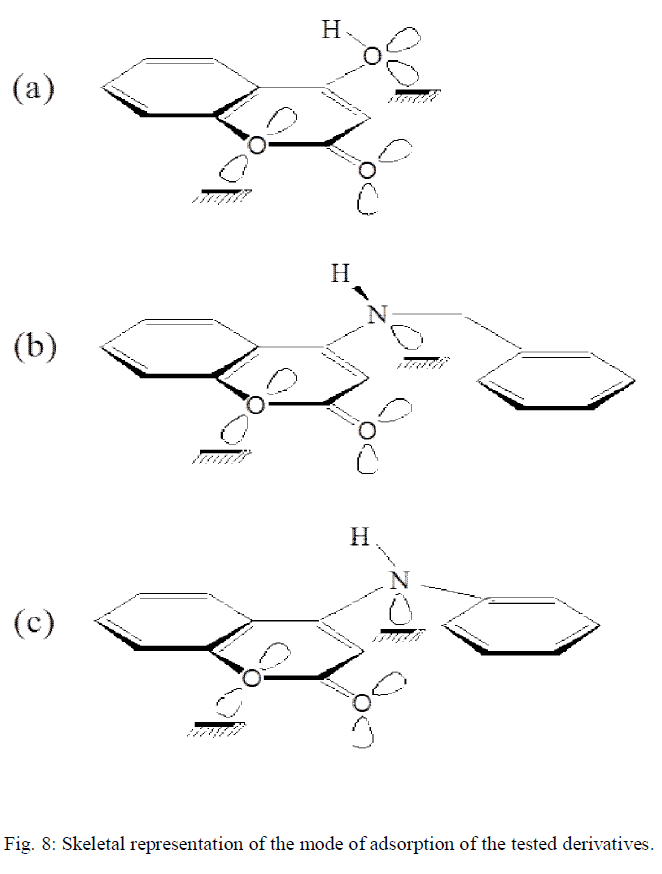
| The extent of inhibition depends on the molecular size of the molecule and the electron density (donating or with drawing) of the substituent groups. Skeletal representation of the mode of adsorption of the tested derivatives is shown in Fig.8 [14]. Compound (b) exhibits excellent inhibition power due to: (i) its larger molecular size that may facilitate better surface coverage, (ii) its adsorption through two active centres as shown from Fig 8, and (iii) the benzyl gp. which is highly electron releasing group which enhance the delocalized π-electrons on the active centres of the compound. Compound (c) comes after compound (b) in inhibition efficiency inspire of it has two active centres, because it has lesser molecular size. Compound (a) has the lowest inhibition efficiency in spite of it has two active centres. This is because it has the lowest molecular |
 |
VI. CONCLUSION |
| 1. The investigated 4- coumarin derivatives are efficient inhibitors for the corrosion of zinc in 2MNaOH solution. |
| 2. The inhibition efficiencies of 4-coumarin derivatives increased with increase in inhibitors concentration but decreased with the increase in temperature. |
| 3. The addition of BaCl2 to coumarin compounds improves the values of the inhibition efficiency due to synergistic effect. |
| 4. The inhibitive effect of these compounds due to the formation of complex adsorbed on the metal surface. |
| 5. The adsorption process follows Temkins isotherm. |
| 6. The inhibition efficiency obtained from polarization measurements showed good agreement with those obtained from comparative weight loss determination. |
References |
|