ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Guddi Choudhary1, Arpita Sharma1 and Alka Sharma2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The corrosive behavior of some metals was studied in varying strength of two different acids by employing weight loss method. The experimentation was carried out at room temperature. The effect on corrosion behavior of metals with the change in acidic strength as well as the nature of metal was monitored at different immersion periods. The corrosion parameters were determined with the experimental data. On analysis, at room temperature, HCl (compared to H3PO4) was found to be more aggressive towards aluminium as well as mild steel (in comparison to copper). The kinetic parameters were evaluated and discussed. The surface analysis also carried out with the help of Scanning electron microscope (SEM), Energy dispersive X-ray (EDX), Optical microscope (OM) and X-ray fluorescence (XRF). Overall the result suggest that corrosion was observed significantly less in H3PO4 acidic medium as compare to HCl medium.
Keywords |
| Metal, Acid corrosion, Corrosion parameters, Kinetic parameters, SEM, EDX, OM, XRF. |
INTRODUCTION |
| Corrosion is a prevailing destructive phenomenon in science and technology. As per literature survey, the cost due to corrosion in many countries is as high as 3-5 % of the GNP i.e. wasteful in terms of economy of any country [1-3]. This represents a huge amount of money which should have been channeled into the provision of basic social amenities in these countries. In practice corrosion can never be stopped but can be hindered to a reasonable level by analyzing of behavior of metals in different environments and by applying various inhibition methods according to their nature. Shaw Barber & A. Kelly [4] and R. R. Pierre [5] determined that metals are usually exposed to the action of bases or acids in the industries in a variety of practical applications such as acid pickling, industrial acid cleaning, cleaning of oil refinery equipment, oil well acidizing and acid descaling. The exposures can be severe to the properties of the metals as well as age of metals also, thus lead to ritual failure of materials in service. |
A. Objectives |
| There is need to study the corrosion behavior of metals when exposed to various environments, as this is an important factor in material selection. This work examines the corrosion behaviors of some industrially used metals, such as aluminum, copper and mild steel in hydrochloric acid or phosphoric acid. These metals have been a subject of numerous studies due to their high technological value and wide range of industrial applications. |
| Metals: Christian Vargel, et al. [6] reported that Aluminum and its alloys, however, are reactive materials and are prone to corrosion. Copper is characterized by its high electrical and thermal conductivities and good mechanical workability [7]. Mild steel is one of the major construction materials, which is extensively used in chemical and allied industries for the handling of acid, alkali and salt solutions [8-9]. |
| Corrosive Media: Hydrochloric acid is the most difficult of the common acids to handle from the standpoints of corrosion. Extreme care is required in the selection of materials to handle the acid by itself, even in relatively dilute concentrations or in process solutions containing appreciable amount of hydrochloric acid. This acid is very corrosive to most of the common metals and alloys. Where Phosphoric acid prepared by hydrates (wet) processes generate severe corrosion problems in containers. |
B. Metal dissolution behavior |
| The corrosion behavior of Aluminium, Copper and Mild Steel and their dissolution mechanism in acidic media, viz., HCl and H3PO4 acid at two concentrations (0.5 N-2.0 N) were studied. The experimentations were carried out at room temperature (303±1K) by using a chemical (Weight loss) method. The morphology of these metals surface before and after immersion in HCl and H3PO4 solutions was examined by SEM and optical micrographs at different concentrations of the acid. |
| The compositions of the metals under study were determined by X-Ray fluorescence spectroscopy and the compositions are tabulated as below: |
METHODOLOGY |
A. Specimen preparation |
| Corrosion tests were performed using by chemical measurements. Rectangular coupons of metals of dimensions (3cm× 2.4cm× 0.16cm) were used with a small hole of 0.12mm diameter near upper edge for hanging in test media. Each coupon was cleaned by emery paper of grade 200 to 800 to give a mirror finish and then degreased by double distilled water, then by acetone and dried in desiccators, prior to each experiment each coupon was surface treated [9]. |
 |
B. Test solution- |
| The solutions 0.5 N and 2.0 N HCl, or H3PO4 was prepared by using doubly distilled water. HCl and H3PO4 acids used were of analytical grade, having the pKa value 1× 10-2 for HCl and three pKa values for triprotic phosphoric acid pKa1= 2.12, pKa2 = 7.21, and pKa3 = 12.67. |
C. Weight loss measurements- |
| The weight loss experiments were carried out using surface treated rectangular coupons viz. (Aluminium, Copper and Mild steel) having the above given dimensions. In this method the metal coupons were dipped in beakers containing 50 ml of respective corrosive solutions in two different concentrations ranging from 0.5 N to 2.0 N, were made after 72 h of immersion at room (303±1K) temperature. Before measurement, each coupon was surface treated dried in desiccators for 24 h and then weight was taken. From the weight loss method, the weight loss or corrosion rates of metals were calculated by helping of area and density and time periods [3, 10-11]. A is surface area taken in cm2, D is density of metal in g/cm3 and T is time period in hours and ïÃÂÃâW= W1-W2 Here W1 is the initial weight of the metal coupons and W2 is the weight at a particular immersion time. The procedure similar as reported in our earlier work, ie, by Rekha N. Nair et al. [11] and Alka Sharma et al. [12-13]. The densities for Aluminium, Copper and Mild Steel are 2.7, 8.9 and 7.8 g/cm3 respectively [14]. |
RESULTS AND DISCUSSIONS |
A. Qualitative Solution Characterization: |
| The dissolution of Aluminium, Copper and Mild steel were observed in different concentrations of aggressive media (HCl, and H3PO4). This was evidenced by the decrease in the original weight of the metal coupons. Experimental results reveal HCl medium to be more corrosive, than H3PO4 acid medium. In general the observations depend on the type of material. During initial few hours of the test, in hydrochloric acid coupons began to react vigorously as compare to phosphoric acid. The color of the solution change according to metals dipped in it; solution with Cu coupons appeared light blue which turned dark blue as the immersion time increases. It is because of the formation of cupric chloride (CuCl2) due to reaction of copper with chloride ions of HCl media. |
| The aggressive solution containing mild steel coupons gained color (light brown) which later on darkened with increasing immersion period. This change in color is clearly due to dissolution of Fe to form ferric chloride (FeCl3). The HCl medium with aluminium coupons does not show any considerable change while in H3PO4 medium turbidity appears. At the bottom of the test solution in the MS Coupon of 2.0 N phosphoric acid solutions, a dark and rusty solids in appearance. Literature suggests that the solid product is formed due to Iron phosphate FePO4 [15 - 16]. |
B. Weight loss Measurements: |
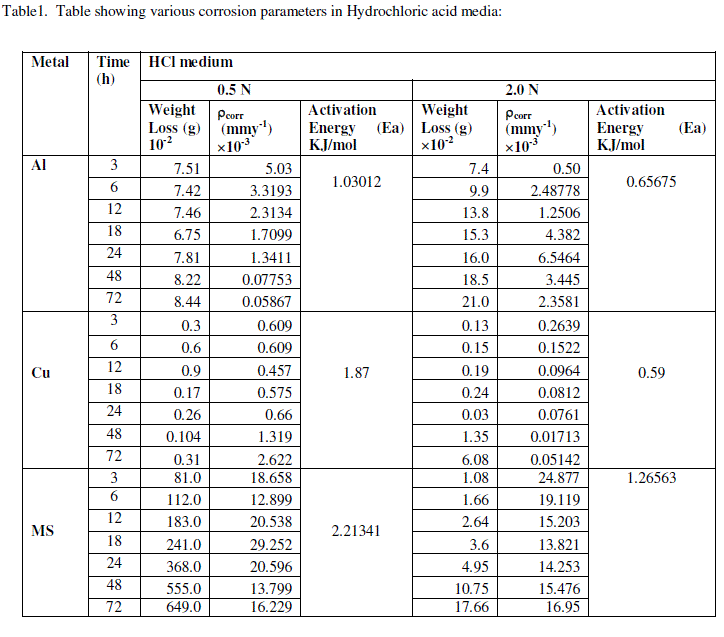
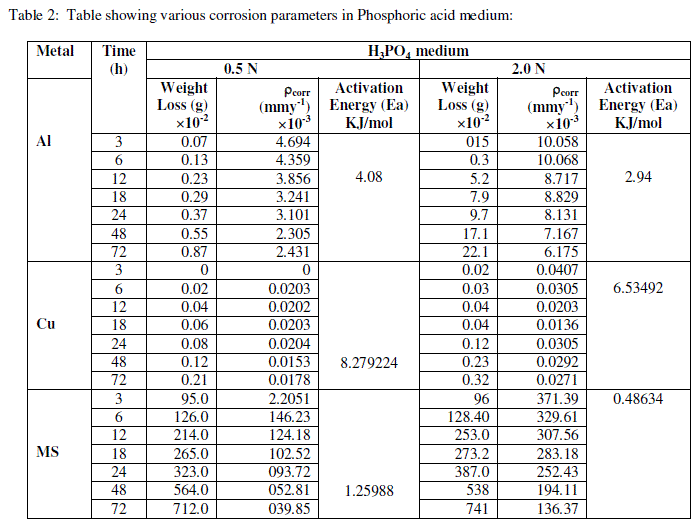
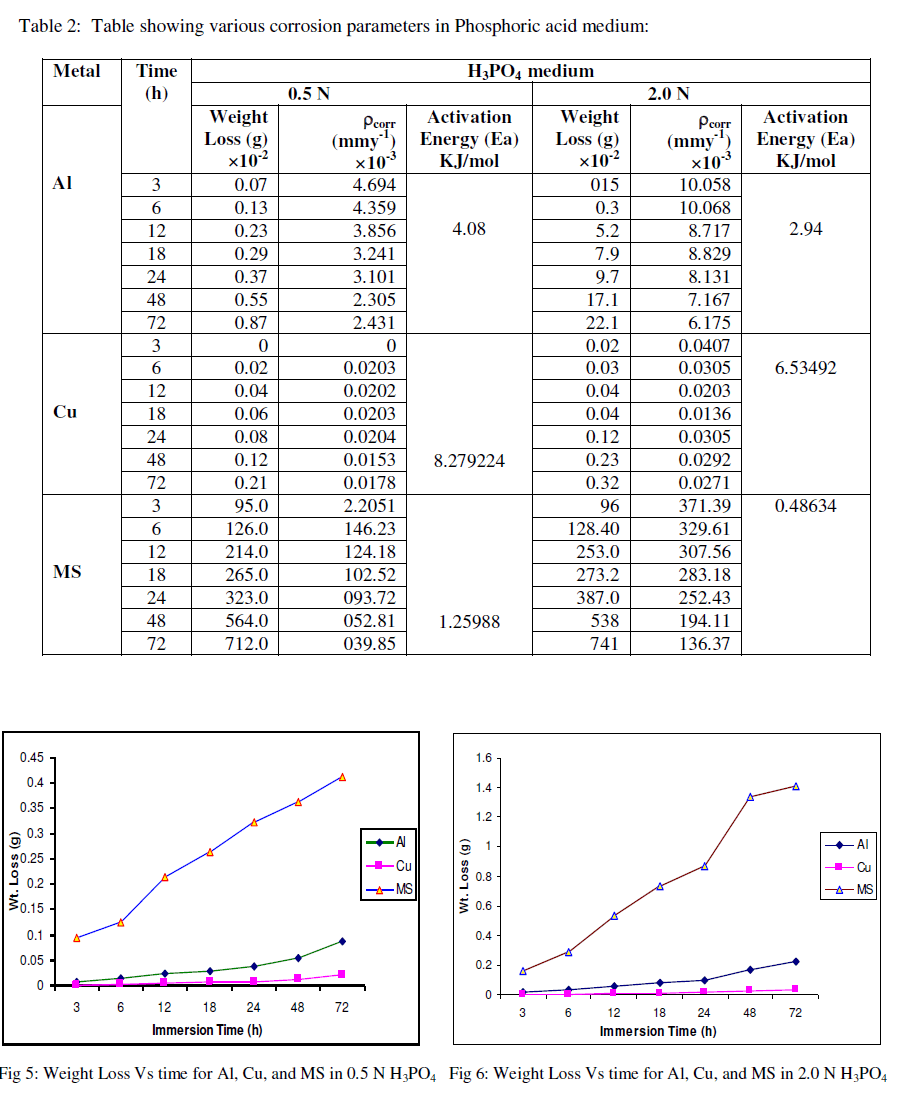
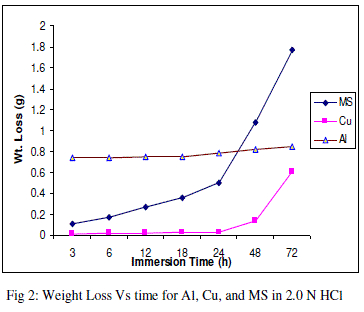
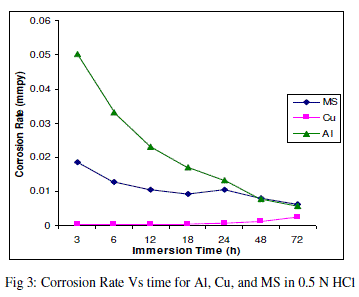
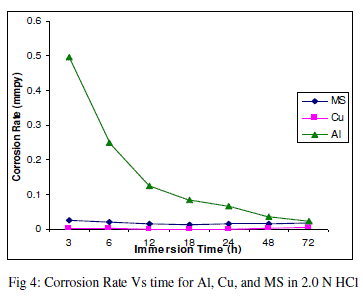
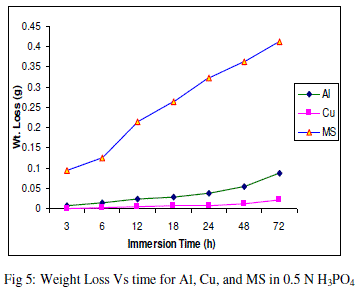
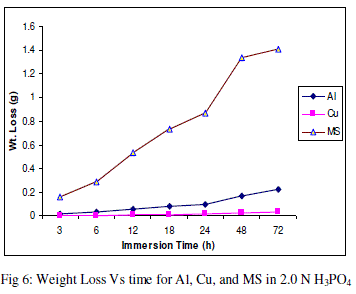
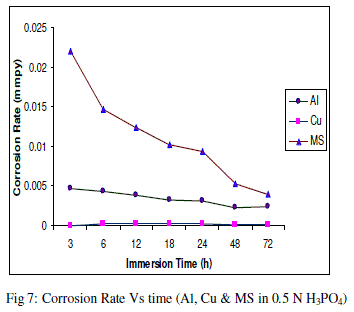
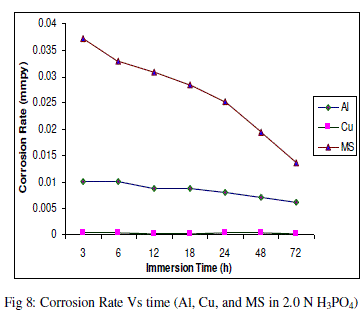
| The experimental data obtained by weight loss measurements were evaluated to determine various corrosion parameters, viz., corrosion rate and activation energy. These have been summarized in Tables 1 and 2. These tables (1 and 2) and figures (1-4) represents the variation in HCl acid and H3PO4 acid as a function of two different concentration 0.5 N and 2.0 N. All experimental results obtained from measurements are interpreted as described under. |
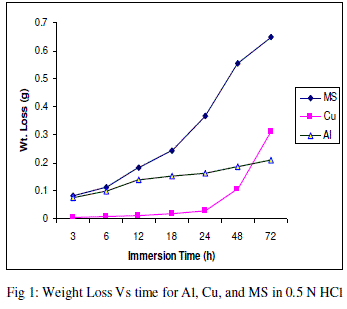 |
| Figures (1- 8) and tables (1 and 2) reveals that aluminium is found to be more corroded in the HCl media as compare to H3PO4 media while in H3PO4 mild steel is found to be more corroded in as compared to aluminium and copper. This is because of the anion effect, the presence of aggressive anions like chloride (Cl-) accelerates the process of corrosion and form a soluble aluminium complex i.e. AlCl3. Aluminium is more reactive in HCl acid solution as compare to Phosphoric acid solution as reported by Peter R. Kosting et al. [17]. This is because aluminium is more reactive than the MS and Cu according to standard Electrochemical potential series and initially aluminium being react violently in initial hours, in HCl medium, as immersion time is increased the film of aluminium chloride formed and considerable weight loss is smaller observed, so Al is active than copper and MS and considerably copper doesn’t frequently affected by these varying acidic strengths. Thus the following trends were observed from weight loss and corrosion rate data in HCl acid and in H3PO4 acid solutions. |
| In HCl medium: Al > MS > Cu ; whereas in H3PO4 medium, the dissolution trend observed was: MS > Al > Cu There are also some final products of reaction of these three metals in HCl as well as H3PO4 acids as explained in literature by Atkins P. W. et al. [18]. |
| Elboujdaini et al. [19] elucidated that the electrochemical reaction of iron in HCl acid than the corrosion reaction involves transfer of two electrons, and then there are two steps, each one represents transfer of one electron and one of them may be considered the rate determining step. This result is in good agreement with the mechanisms that are suggested for iron dissolution. |
 |
 |
 |
| Arrhenius law predicts that corrosion rate increases with increase in the temperature and decrease with increases in Ea and A may vary with temperature (Eq.1). Nowadays it is best seen as below, empirical relationship. The activation energy may be calculated as a straight line of the logarithm of the corrosion rate (mmy-1) and 1/T according to the following relation: |
| ln k = -Ea/RT + ln A |
| The different value of activation energy in different acidic strength of two aggressive media were calculated and summarized in tables 1 and 2. The experimental values obtained clearly indicate that Ea for aluminium is lower in HCl than H3PO4 acid; this reveals that the corrosion rate is higher in HCl media compared to H3PO4 acid media. In other case when we compared the Ea value of Cu and MS in both acidic media Cu corrodes more in HCl while MS more corrode in H3PO4 acid. These results clearly explained by the higher and lower value of activation energy. |
D. The pH effect on Corrosion behavior: |
| Pourbaix M.et al. [21] explained that the acidity or alkalinities of medium affects the corrosion behavior of metals and alloys, and it accelerates strongly where two dissimilar metals are contact with each other and oxide coating is not present. A slightly higher pH about (5-6) was observed in phosphoric acid solutions that than of Hydrochloric acid (0.1- 2.5). The pH of both the solutions was found increased significantly compared to their initial pH values and similar a slight increase in the pH for the 0.5 N HCl acid solutions was also observed. Thus suggesting that a significant amount of hydrogen consumption occurs in these acids solutions .When these metals are dipped for different immersion time period to study the effect of pH, it reveals that corrosion rates are significantly greater at lower pH for Al and as the pH increases, a slight decrease in corrosion rate was observed. Whereas, for copper where the dissolution potential was not significantly affected by a variation in pH. The results for mild steel indicate that both uniform corrosion and localized corrosion are affected by a decrease in pH. It also reveals by reference figures (9-10) for Aluminium as given below: |
 |
E. Microscopic Surface Morphology of Coupons: |
| The microscopic surface morphological analysis was undertaken by Optical micrographs and Scanning Electron Microscope (SEM). Optical micrographs were taken with the help of (LABOMED) microscope and SEM was taken with the help of ZEISS special edition instrument. Several conclusions may be drawn from the results of these scoping experiments that could apply to these different acids. |
Optical Micrographs (OM): |
| Figures 11-13 are optical micrographs of the all three metal coupons i.e. (Al, Cu, Ms) that reveal the morphology of the non-corroded polished metals prior to immersions in aggressive media. |
 |
| Several areas of these coupons were damaged which was evident by their surface appearance, the film present on the surface was cracked and flaked off. There was an insignificant amount of corrosion product on these coupons which exposed to the acid. All coupons exhibited severe general attack and crevice attack where the glass string had been inserted into the hole. In all cases, the corrosive attack had eaten through the top edge of the coupon and the coupon had fallen to the bottom of the bottle. Further crevice attack ensued on the edge of Cu and MS Coupons (figures 15 & 16 and 18 &19 respectively) in 2.0 N HCl acid. The surface of MS coupon shows highly dissolution and also the deposition of ferric chloride was observable. Thus, in general, the HCl acidic medium is more aggressive than H3PO4 acidic medium, wherever Cu coupons are less infected by these media, as compared to Al and MS Coupons. |
 |
 |
Energy Dispersive X-RAY (EDX): |
| The EDX technique used to identify the elemental composition of as small as cubic micron of material (figures 35-40). EDX was taken by the help of ZEISS Sanning Electron Microscope equipped with AN-10000 link EDX spectrometer. EDX spectrum of the sample indicated that corrosion product layer has been formed over the surface of the metal samples in different acid media. |
 |
| mild steel in phosphoric acid which is more corroded in H3PO4 media. Hence it can be revealed that the above results are in good agreement with the results suggested by the weight loss. |
XRF graphs of Metal Coupons: |
| The XRF studies were also carried out by the help of Pay Analytical X-ray fluorescence instrument in the frequency range of 25 KV. The study shows the elemental compositions of these coupons (figures 41-43). The highest peak in each graph shows the purity of Al, Cu and Fe for these metals respectively. |
 |
CONCLUSIONS |
| The dissolution behavior of Aluminium, Copper and Mild Steel in 0.5 N and 2.0 N of HCl and H3PO4 solutions have been investigated using weight loss (chemical) method. The dissolution was further endorsed by carrying out surface morphological studies employing micro spectroscopic techniques. The following conclusions can be drawn on the basis of the results: |
| • Weight-loss data and corrosion rate graphs reveals that the Al coupons immersed in HCl acid is more corrode compare to H3PO4 acid solutions, this is due to formation of Aluminium chloride, and the value of activation energy shows the reaction feasibility. |
| • Mild steel coupons shown the insignificant dissolution in H3PO4 acidic medium in 0.5 N as well as 2.0 N concentrations of acids compare to HCl acids due to formation of unstable FePO4 compound. This effect increases with increases of their immersion time. |
| • Dissolution of Copper is less in HCl as well as in H3PO4 acidic medium compare to other two metals, but when compared in different acidic strength than Cu is more reactive in HCl acid than H3PO4 acidic medium. |
| • Scanning electron microscopy (SEM), and Optical micrographs, EDX was used to observe surface defects and corrosion. SEM observations showed that defects or pitting corrosion occurred on the surfaces of the all coupons in tested media. EDX spectrum recorded on the area of dispersed pits and grooves shows the peaks of chloride and phosphate acidic media. |
| ACKNOWLEDGMENTS |
| The authors express their deepest gratitude to the Head, Department of Chemistry, University of Rajasthan, Jaipur for providing necessary research facilities and are also highly thankful to UGC, New Delhi (India) for providing the financial Support. |
References |
|