ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Dr.A.Narender Department of physics, Kakatiya University, Warangal (A.P) – 506009, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
A systematic measurement of dielectric constant (ε) and loss (tanδ) on powder pressed pellets of AMPS with AA, MAA and MBA copolymers in composition 50/50 have been carried out in the frequency range 200Hz – 100 kHz and in the temperature range from room temperature to180oC covering through the glass transition temperature (Tg). Relaxation peaks were observed (above Tg )from tanδ versus temperature curves. The peaks are attributed to α – relaxations. Further the peaks shift towards higher temperatures for higher frequencies. The AC conductivity has also been evaluated from the data. The results are discussed in the light of substituent effect in the copolymer.
Keywords |
| Dielectric constant (ε), Dielectric loss (tanδ), AC conductivity. |
INTRODUCTION |
| The use of polymers in engineering industry as dielectric is becoming increasingly important. Polymers substituted by sulfo groups have unusual properties, allowing a wide range of applications. Introduction of the sulfo groups into fibers improves dyeability and imparts antistatic properties. Sulfo polymers are employed in biomedical systems including biomembranes[1]and blood compatible materials [2-5]. Sulfo polymers are also having potential applications in bioengineering, biomedicine, chromatography, water purification[6]and in enhanced oil recovery(EOR)[7-10] .Ranga Rao carried out the synthesis and studied dielectric property of AMPS with Vinyl Amides[11] . As no reports are available on dielectric properties of AMPS with acrylamide copolymers, an attempt is made to investigate the dielectric properties of AMPS+AA, AMPS+MAA, AMPS+MBA copolymers in 50/50 composition. |
II.EXPERIMENTAL |
| The powder samples of AMPS with AA, MAA and MBA copolymers of 50/50 composition were used to prepare the pellets of suitable size for the present measurements. The dielectric constant (ε) and dielectric loss (tanδ) were measured using a GR-1620A capacitance measuring assembly in conjunction with an indigenously built three terminal high temperature cell.The measurements have been carried out in the frequency range from 200Hz to 100 kHz and in the temperature range from room temperature to 180oC, covering through the glass transition temperature (Tg). |
III.RESULTS AND DISCUSSION |
| The results on the measurement of dielectric constant (ε) and loss (tanδ) for AMPS with AA, MAAand MBA copolymers of 50/50 compositions at room temperature are given in table-1 for frequencies 200Hz and 100kHz and the variation are shown in fig.1. |
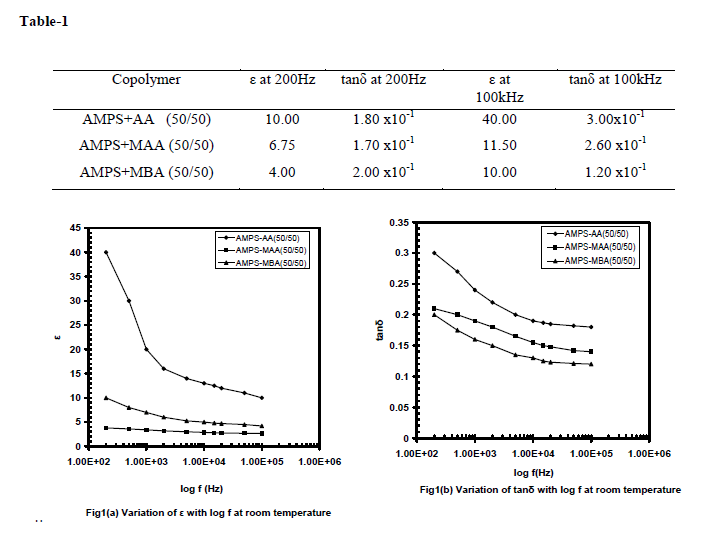 |
| It can be seen that both the values of ε and tanδ decrease, with increase of frequency and the rate of decrease is smaller for high frequencies. Further, an enhanced variation is being observed for AMPS+AA copolymer. It is believed that the large value of ε at low frequencies could be due to effect of space charge polarization,[12] whereas this effect is negligible for the frequencies beyond 10kHz. On comparison among the three copolymers it is seen that an enhanced dielectric property is being established in AMPS+AA copolymer at room temperature. This may be due to more polar nature of AA than that of MAA and MBA in copolymer systems. Figures 2(a) and 2(b) show the variation of ε and tanδ respectively with temperature for AMPS+AA (50/50) and fig.2(c) for ac conductivity (log ζac) with reciprocal temperature for four selected frequencies. From room temperature to 40oC, ε is frequency and temperature dependent and increases at a slower rate. Beyond 45oC and up to 60oC ε decreases giving rise a peak at 45oC for all frequencies, and from 60oC to 80oC ε increases at a rapid rate and then decreases with increase of temperature giving rise a peak at 80oC for all frequencies. Further increase of temperature there is an increase in ε i.e. beyond 100oC. The peak appeared around 45oC in ε for all frequencies is attributed to the slow motion of individual side chain molecular groups like NH2 and CH3. The rapid increase of ε with temperature is attributed to |
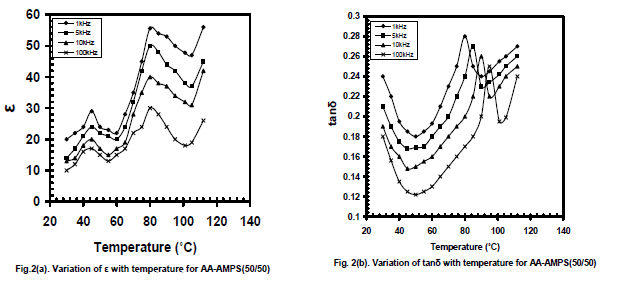 |
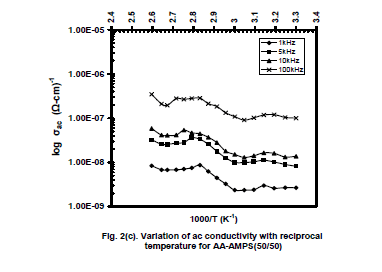
| The variation of tanδ with temperature shows a gradual decrease due to annealing effect, giving rise a dip at 50oC with increase of temperature. Beyond this temperature the increase of tanδ is continued up to 90oC. Further increase of temperature resulted in a rapid enhancement of tanδ giving rise relaxation peaks at 70oC, 75oC, 80oC and 90oC for 1 kHz, 5 kHz, 10 kHz and 100 kHz respectively. The relaxations are known to be α- relaxations, which occur due to segmental motion of polymer main chain above Tg.. The peak positions shift towards higher temperatures for higher frequencies. Further increase of temperature caused a decrease in tanδ. The ac conductivity ( ζac) has been evaluated from the data on ε and tanδ as a function of temperature and plotted as log ζac versus reciprocal temperature (Fig 2c). A frequency and temperature dependent variation in ζac is seen up to 40oC. The trend of variation is same as that seen for ε and tanδ. Beyond 45oCand up to 80oC, a linear rise of conductivity is observed, because the copolymer gets softened and relatively more number of charge carriers available for conduction. Then the conductivity is found to decrease rapidly beyond 80oC entering into a disordered phase of the polymer. The overall change in conductivity is observed to be in the range between 10-9 to 10-6(ohm-cm)-1. |
 |
| Figures 3(a) and 3(b) show the typical variation of ε and tanδ respectively with temperature for AMPS+MAA (50/50) and fig.3(c) for ac conductivity (log ζac) with reciprocal temperature for four selected frequencies. Unlike the behaviour observed in case of AMPS+AA, only one set of peaks were seen in the variation of ε at 80oC and without any dip in the variation of tanδ. The relaxation peaks in tanδ have been noticed at 72oC, 75oC, 80oC and 90oC for 1 kHz, 5 kHz, 10 kHz and 100 kHz respectively beyond Tg. The ac conductivity variation is in the range of 10-10 to 10-7(ohm-cm)-1. |
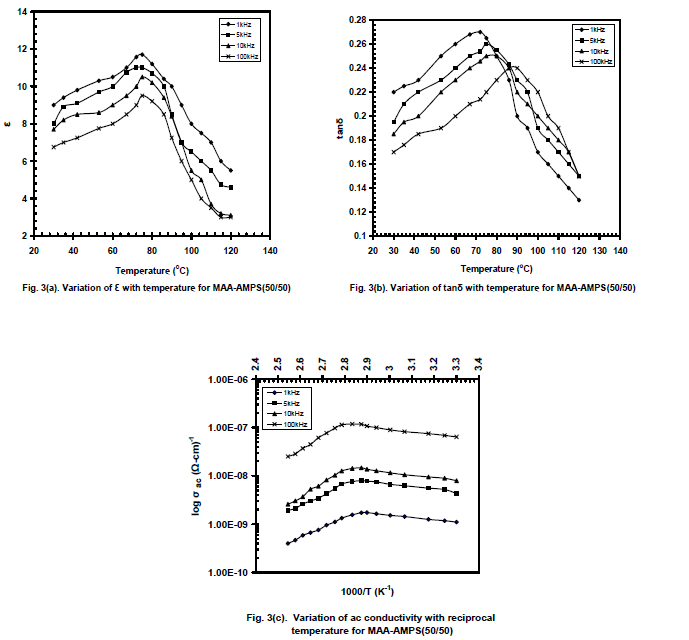 |
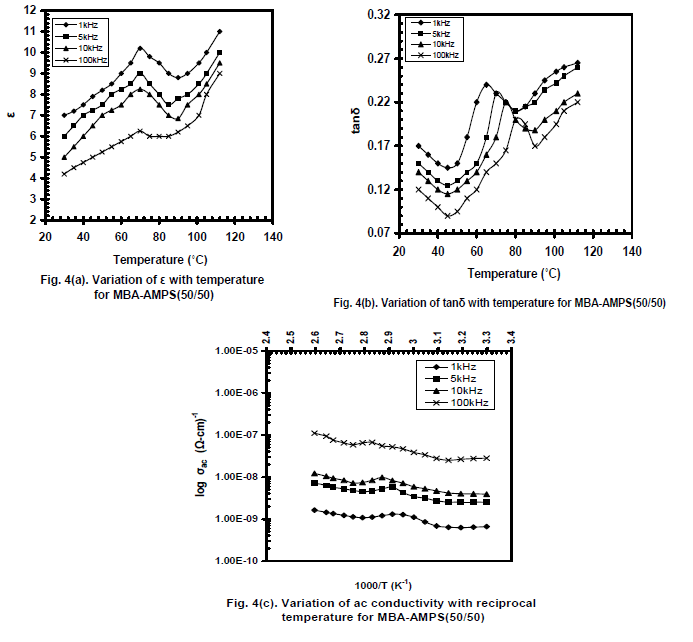
| Figures 4(a) and 4(b) show the typical variation of ε and tanδ respectively with temperature for AMPS+MBA (50/50) and fig.4(c) for ac conductivity (log ζac) with reciprocal temperature for four selected frequencies. A peak is observed at elevated temperatures i.e., at 70oC for all frequencies. It can be seen that tanδ decreases from room temperature to 50oC giving rise a dip around 50oC as observed for AMPS+AA (50/50); then tanδ started increasing giving rise α – relaxation peaks at 65oC, 70oC, 75oC and 80oC for 1 kHz, 5 kHz, 10 kHz and 100 kHz respectively. The ac conductivity variation for AMPS-MBA is in the range of 10-10 to 10-7 (ohm-cm)-1 , one order lower than that for AMPS+AA |
 |
 |
IV.CONCLUSIONS |
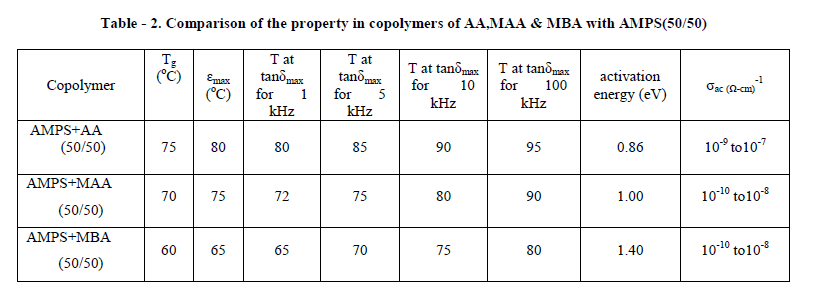
| Among the copolymers AMPS+AA, AMPS+MAA and AMPS+MBA each of (50/50) composition AMPS+AA (50/50) established an enhanced dielectric property around room temperature and also around Tg, the glass transition temperature of the respective copolymer. Presence of α-methyl group in MAA of AMPS+MAA and two acrylamide groups on either side of methylene in AMPS+MBA increases the bulkiness in the side chain of the copolymer system. It is known that, the bulky groups always increase the chain mobility and act in a direction to reduce the Tg values and also α-relaxations temperatures. Accordingly it can be seen that the α- relaxation peaks observed typically for 1 kHz occurred for AMPS+AA, AMPS+MAA and AMPS+MBA at 80oC, 72oC and 65oC respectively. It can be also seen from the Table 2 that Tg values for respective systems are 75oC, 70oC and 60oC, which provides a supportive reasoning for dielectric behaviour described above. |
References |
|