ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Roshni P. Adiyecha1, Jignasa Joshi2, Pradeep U. Verma3 and Yogesh T. Jasrai4
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
An efficient regeneration system, direct and indirect organogenesis, was developed for Curculigo orchioides-an endangered medicinal herb. Shoot multiples were initiated from in vitro grown leaf explants through direct organogenesis on Murashige and Skoog’s (MS) medium containing ½ strength nitrogen salts and 0.44 μM BA. Almost 10 shoots were obtained per leaf explant (1 cm long). On the other hand MS medium with full strength nitrogen salts and 2.22 μM BA, stimulated callus formation and almost 8 shoots were obtained per leaf explant (1 cm long ). Comparative study of shoots regenerated directly or through callus revealed differences in number of leaves, shoot length, root length, fresh and dry weight
Keywords |
| Curculigo orchioides, Kali-musli, Regeneration, Organogenesis and Leaf explant |
I.INTRODUCTION |
| Curculigo orchioides Gaertn. is a medicinal herb belonging to family Hypoxidaceae. Curculigo orchioides is included in the IUCN category of lower risk near threatened [17]. Whole plant is medicinally valuable including leaves, roots and rhizome. The rhizome of this plant are sweet, cooling, diuretic, aphrodiciac-viriligenic and tonic which can be used against homorrhoides, leucorrhoea, pruritis, skin diseases, bronchitis and jaundice [14] . Methanol extract has shown immuno-stimulant [2], antioxidant [3], antiasthamatic [11], hepatoprotective, anticarcinogenic and antiinflammatory activity [15]. The tuberous roots of this plant are considered to be tonic alterative, demulcent, diuretic and restorative and are used as a poultice for itch and skin diseases [19]. |
| Phytochemical investigations of the rhizome revealed the presence of curculigoside [7], curculigoside A [4], curculigoside B [25], curculigoside C [27], orcinol glycosides [7], 2,6,- dimethoxyl benzoic acid [4], curculigol, curculigo saponins A-M [26], 2,3, 4, 7- tetramethoxyxanthone, 1,3,7- trimethoxylanthine, and daucosterole [6]. |
| Removal of plants for medicinal and edible tuberous roots as a substitute for safed musli, coupled with extensive denudation of forests floor caused by cattle grazing [6], poor seed setting and germination are some of the major causes that contribute to the herb being categorized as a threatened plant [1]. High incidence of viral and bacterial diseases poses yet another constraint in its multiplication and strengthens the need for in vitro multiplication. |
| Earlier reports on Curculigo orchioides described direct shoot induction using different explants like leaf [12], [23], rhizome [22] and apical meristem [5], [24]. However these protocols demonstrated low levels of shoot regeneration. This study aims to establish an efficient regeneration system for direct and indirect organogenesis leading to rapid clonal propagation. |
II.MATERIALS AND METHOD |
| Young leaves of Curculigo orchioides shoots were used as an explant. Leaves were surface sterilized with 0.1% of HgCl2 (1-2 min). Treated leaves were thoroughly washed with sterilized distilled water (3-4 times) and inoculated aseptically on MS medium. |
| For Direct organogenesis: Leaf pieces (1 cm long) were inoculated on MS medium with ½ strength nitrogen salts (ammonium nitrate 0.825gm/l, potassium nitrate 0.9gm/l) and 2 % sucrose (w/v). The medium was supplemented with 0.44 μM 6 BA alone. |
| For Indirect organogenesis: Leaf pieces (1 cm long) were inoculated on MS medium with full strength nitrogen salts (ammonium nitrate 1.65gm/l, potassium nitrate 1.8 gm/l) and 3 % sucrose (w/v). Moreover 2.22 μM BA was added as growth regulator. |
| The pH of all media was adjusted to 5.8 and autoclaved at 121 ºC (20 min). For all media 0.8 % agar-agar was added as gelling agent for semi-solid medium. The cultures were incubated at 25ºC under 16 h photoperiod with 55 μ mol m-2s-1 photon flux density. |
| Fifty replicates each were studied for direct and callus mediated organogenesis (indirect). Means and standard error (SE) were calculated. |
III.RESULTS AND DISCUSSION |
| Earlier, the rhizome explants showed fungal contamination and exuded abundant phenolics in the medium [13]. In present study, leaves were used as an explant for direct and indirect organogenesis. Leaf explants maintained on MS medium with ½ strength of nitrogen salts and supplemented with lower concentration of BA (0.44μM), developed shoots directly from the explant (Fig-1a). This is in accordance with the previous reports [1] [13]. The first visible sign of shoot initiation was observed after 15 days, as white shiny protuberance on the explants (Fig-1a). The shoot bubs enlarged in size and reached an average length of 9 cm in 10-12 weeks. It took about 10 to 12 weeks for complete organogenesis (Fig-1b). Many researchers tried different medium and growth regulators for root induction of C. orchioides [5] [12] [13] [22] [24]. The higher level of BA in the medium inhibited the growth and shoot proliferation from the leaf explants [20]. In fact, the leaf explant of C. orchioides cultured on cytokinin-free medium produced entire plantlets from the cut ends of the leaf explants [1]. In present study, lower level of BA (0.44μM) induced direct organogenesis. |
| In present study rooting was achieved on the same media. Plantlets obtained contained on an average of 3 leaves. The average length of shoots obtained was 9 cm and length of roots was around 2 cm. Maximum 25 shoots per explants were obtained. Clusters having 1-5 shoots were robust and dark green, having 4-8 leaves. Clusters of more than 5 shoots were comparatively weak and light green with 2-4 leaves. The induction of plantlets directly from the parent tissue without intervening callus is of potential value for the in vitro multiplication and storage of a given genotype. It is considered that the direct organogenesis observed in this culture system forms an effective and rapid plantlet regeneration system. |
| The leaf explants maintained on MS medium with full strength nitrogen salts and supplemented with 2.22 μM BA, showed callus formation (Fig-2a). The normal level of nitrogen salts (as in basal medium) and higher amount of BA stimulated callus formation in C. orchioides. Callus production in Juniperus excelsa on MS medium with half strength nitrogen was noted ealier [16]. The present study revealed that callus induction in Curculigo orchioides was stimulated by full strength nitrogen salts of MS medium and higher level of BA. The higher concentration of BA induced callus formation from the cut ends of leaf explants in C. orchioides was also noted earlier[1]. Similarl callus formation in Citruss sinensis with higher level of BA was noted8. Callus formation was initiated within 2 weeks from the cut ends of the leaf explants. High frequency of callus formation was obtained in next 15 days after initiation (Fig -2a). The callus was compact and pale-white with average fresh weight of 2.175 grams. The morphogenic compact callus showed shoot differentiation. These when transferred to MS medium with ½ nitrogen salts and low level of BA (0.44μM) produced shoots (Fig-2b). Shoots and roots were developed within 3 weeks of the transfer. The average length of shoots obtained was 7 cm and length of roots was around 2 cm. |
 |
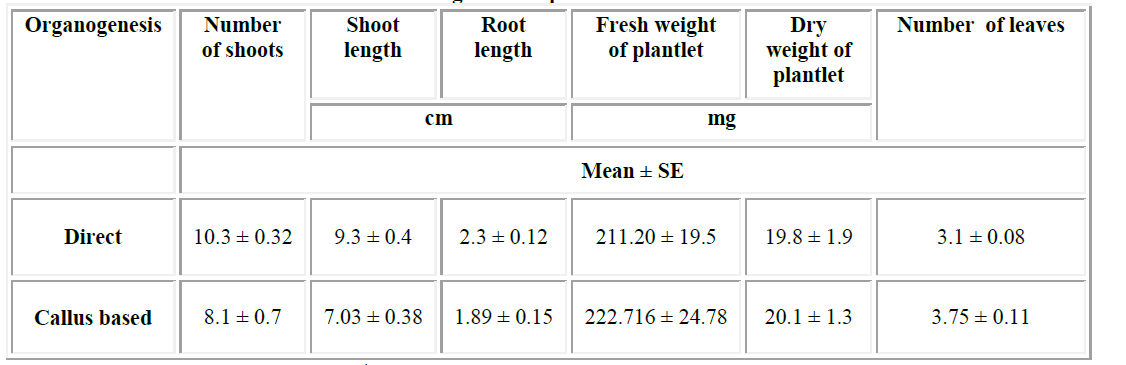 |
| Earlier researchers have tried different explants and growth regulators for the induction of direct and indirect organogenesis of C. orchioides. Single shoot formation directly from leaf explant using BA as growth regulator [1] [12] and about 5 shoots were obtained using 2,4 D as growth regulator12. Similarly, 4-5 shoot multiples [24] and 6-7 shoots [5] [18]8 directly from the shoot bud explants was reported. Through indirect organogenesis, 4 shoots were obtained from leaf explants [20], 8 shoots from rhizome callus [13] and 11 shoots through stem discs [10]. In the present study, almost 10 shoots per explant were derived from direct organogenesis while 8 shoots were observed through indirect organogenesis (Table-1). |
| This is the first report on comparison of direct and indirect plantlet formation of Curculigo orchioides. Results showed that average number of shoots, shoot length and root length were higher in plantlets developed from direct organogenesis. In contrast, average fresh weight and dry weight of plantlets derived through callus was more than direct organogenesis. The growth performance of regenerated plantlets (both through direct and indirect organogenesis) is being evaluated. |
| The protocols studied demonstrated rapid micropropagation of C. orchioides that can be employed for large-scale cultivation and conservation of this endangered medicinal plant. |
References |
|