ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Dr. Sheetu Raina and Dr. Anupriya Sachar Ph.D., Dept. Of Zoology, University of Jammu, J&K, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
In the present study an attempt has been made to evaluate the combined effect of heavy metal, zinc and carbamate pesticide, sevin on the haematological parameters of the fish, Labeo boga for the experimental period of 63 and 60 days respectively. The studied parameters were Total erythrocyte count (TEC), Haemoglobin (Hb), Haematocrit (Hct), Mean corpuscular volume (MCV), Mean corpuscular haemoglobin (MCH), Mean corpuscular haemoglobin concentration (MCHC), Total leucocyte count (TLC) and differential leucocyte count (DLC). The mentioned parameters exhibited different responses on exposure to sevin and zinc. The alterations in these parameters have ultimately become the causative for affecting the general health status and hampering the entire metabolic machinery of the fish.
Keywords |
| Sevin, Zinc, Fish Haematology, Labeo boga |
INTRODUCTION |
| Human destructive influence on the aquatic environment in the form of sublethal pollution usually results in chronic stress conditions that have negative influence on aquatic life [1]. Freshwaters are highly vulnerable to pollution since they act as immediate sinks for the human activities [2]. The most prevalent xenobiotics arising out of industrial and agricultural activities are pesticides and heavy metals where the stress response mechanisms have been widely addressed in vertebrates in general and fish in particular. Fishes mostly have a tendency to bioaccumulate such type of xenobiotics and humans can be at great risk being at the top of the food chain [3]. Zinc is one of the most common heavy metal pollutants in aquatic ecosystem. Although zinc is required in small quantities for normal development and metabolism of organisms [4] but can become toxicant, if its level exceeds the physiological limits. Pesticides are directly applied to the agricultural fields mainly to control pests and improve the crop yield. These pesticides finally find their way into the water bodies and cause harm to aquatic flora and fauna including fishes[5]. The carbamate pesticide, sevin is known to affect growth, metabolism and development of fish. Sublethal concentrations of both zinc [6, 7, 8] and sevin [9] have been reported to drastically affect the haematological parameters of fish. Presently the effect of sublethal concentrations (20%, 40% & 60% of LC50) of heavy metal, zinc and carbamate pesticide, sevin on the haematological parameters of fish Labeo boga have been evaluated for an experimental period of 63 and 60 days respectively. |
| The paper is organized as follows. Section II describes the methodology adopted for the research work. Section III represents the results and discussion of the effect of stressors on haematology. Section IV reflects conclusion part of the research work. |
MATERIALS AND METHODS |
| Adult specimens of Labeo boga were collected with the help of cast net from Nagrota stream of River Tawi from Jammu, J&K, India. The fishes were acclimatized for about 15 days. Three different concentrations viz. 20% (0.4mg/l), 40% (0.8 mg/l) and 60% (1.2 mg/l) of LC50 of zinc and 20% (0.3 mg/l), 40% (0.6 mg/l) and 60% (0.9 mg/l) of LC50 of sevin along with controls were maintained during the course of the experimental period. 0.5ml of blood was taken directly by cardiac puncture with the help of heparinized needles using EDTA as an anticoagulant. Among blood parameters TEC and TLC were counted with the help of improved Neubauer cytometer [10]. Hb% was determined by using Sahli’s haemoglobinometer [11], Hct was determined by centrifugation method [12]. MCH, MCV and MCHC were calculated by using formulae: |
| MCV= Hct × 10/ RBC Count Its unit is fentolitre (fl) |
| MCH = Hb% × 10/ RBC counted in picogram (pg) |
| MCHC = Haemoglobin in g/100ml × 100/ Vol. of packed RBCs in 100ml. It is represented as percentage (%). |
| Identification of cellular components: Identification of various blood cells (differential leucocyte count) was done by methodology as adopted by Anderson [13]. |
| Microphotography: Slides of blood smears were scanned and photographed with Sony SSC-DC378P Semi-Digital camera attached with Olympus CH20i Research microscope. |
| Experimental data and those of control were statistically analyzed by means of analysis of variance (ANOVA). Significance was set at P<0.01. All analysis were performed using SPSS software. |
RESULTS and DISCUSSION |
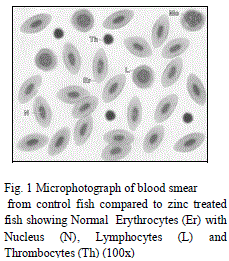
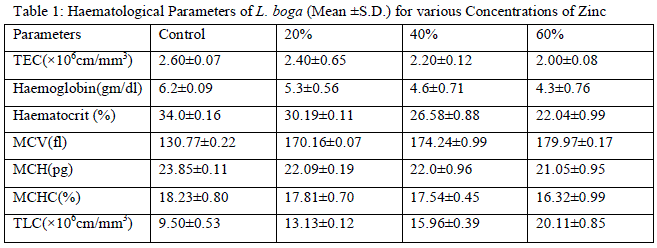
| RBC and related parameters: Presently marked decline have been observed in TEC in both zinc and sevin treated fishes [compared to control (Tables 1 and 3, Figs. 1 and 10)] which can be attributed to i) an increase in the rate of RBC destruction and ii) abnormal/distorted shape of RBCs (Figs. 5 and 14) which then no longer can perform normal function. Support for the present viewpoint can be derived from the findings Palanisamy et al. [1] and Sachar and Raina [2] who too observed decline in TEC as a result of erythrocyte destruction. The microscopic examination of blood smears of both zinc and sevin treated fishes depicted anomalies in shape as well as nuclear malformation of RBCs compared to control ones (Figs. 1, 5 and 10, 14). Distorted shape of RBCs may also result in reduced oxygen carrying capacity of RBCs and thus consequently can lead to tissue hypoxia as earlier also suggested by Yang and Chen [14] and Verma [15]. Sevin, however appeared to be more deleterious to RBCs as compared to zinc which must have resulted in the overall failure of haemopoietic machinery and thereby inhibiting erythropoiesis. |
| Similar to TEC, Hb and Hct, too, have been observed to exhibit overall significant decline (P<0.01) in their values in zinc and sevin treated fishes in all exposures (Tables 1 and 3). Decline in Hb and Hct may be attributed to RBC lysis/destruction (Figs. 8 and 14) and greater prevalence of erythroblasts (immature RBCs) in the general circulation in both zinc and sevin treated fishes (Figs. 6 and 14). However, the erythroblasts make their appearance after second week in zinc treated fishes (Fig. 6) but only after 5th day in sevin treated fishes (Fig. 14) which thereby indicates that sevin has impaired haemopoietic organs (liver, anterior kidney & spleen) more acutely than zinc. Availability of greater number of erythroblasts in general circulation appears to be an attempt on the part of fish to compensate for low TEC. |
| Calculated Values of TEC/Red Blood Cell Indices: These calculated indices showed significant alterations in their values with increasing concentrations of zinc and sevin in all the treated groups compared to control ones (Tables 1 and 3). MCV gives an indication of the status or size of RBCs. MCV exhibited significant increase (P<0.01) in its values in all zinc and sevin treated groups (Tables 1 and 3). Gupta et al. [16] also documented increase in MCV in xenobiotic treated fishes. Increase in MCV values, according to present author, can be attributed to reduction in TEC values because MCV and TEC have an inverse relationship. Hence decline in TEC presently have been observed to result in an increase in MCV values in both zinc and sevin treated fishes. Increased MCV values are also suggestive of RBC swelling/macrocytosis (Fig. 9). Nikinmaa and Huestis [17] who observed stress induced swelling of fish RBCs attributed it to the osmotic disturbances and uptake of electrocytes and water into the cells accompanied by acidification of plasma and cytoplasm of RBC. MCH represents the amount of Hb in the RBC of an organism whereas MCHC is the average concentration of Hb in given volume of blood. As evident from the Tables 1 and 3, MCH and MCHC exhibited gradual decline in zinc treated fishes and fluctuating pattern in sevin exposed fishes. Such changes in MCH and MCHC may be either due to the i) prevalence of large number of small sized immature RBCs (erythroblasts) in general circulation (Figs. 6 and 14) or ii) reduction in cellular blood iron thereby resulting in reduced Hb synthesis. Thus decline or fluctuating pattern of MCH and MCHC definitely appears to be the reflection of marked decline in Hb because of chronic toxicity of zinc and sevin. On the basis of decline in RBC dependent parameters, fish L. boga have been diagnosed to suffer from two types of anaemias under the influence of zinc and sevin toxicity viz., Megaloblastic (occur either due to depression in TEC, Hb and Hct or greater influx of erythroblasts or immature RBCs in the general circulation or even both) and hypochromic macrocytic anaemia (due to decreased level of MCH and MCHC and increased values of MCV). |
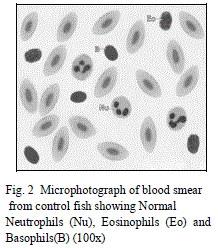
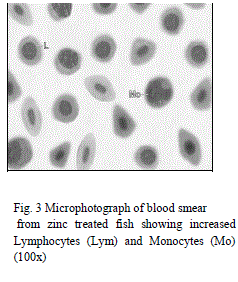
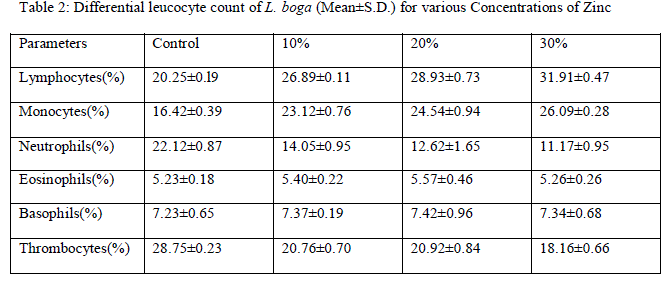
| Leucocytes or White blood cells (WBC): Marked increase have been observed in values of TLC under zinc toxicity (Table 1) which indicates that zinc has stimulated the leucopoietic machinery of fish to release more and more leucocytes in the general circulation thereby resulting in an increase in percental values of TLC. This simply reflects the protective response of defense mechanism of treated fishes to counter the zinc-mediated stress. Contrarily however, in case of sevin treated fishes, sharp decline in TLC (Table 3) of the fish have been observed which plausibly be due to the fact that the immune organs of the treated fish probably gets so much deteriorated that it may not be possible for the fish to cope up the stress caused by sevin and therefore resulted in leucopenia. Among DLC, lymphocytes are the major contributory towards the increase/ decrease of TLC. Increase in TLC in zinc treated fishes may also be due to the release of more and more lymphocytes (compared to control ones) (Table 2, Fig. 1 and 3) in the general circulation from lymphomyeloid tissue. Chronic exposure of sevin might have resulted in inhibition of lymphopoiesis there by affecting the immune functioning of fishes under stress and resulted in TLC in sevin treated fishes throughout the experiment Presently, while monocytes have been observed to exhibit increase (Fig. 3), neutrophils rather were observed to fall appreciably (Fig. 4) during the entire course of experimental period in zinc treated fishes compared to control ones (Figs. 1 and 2). It appears that fish L. boga in a bid to overcome the stress of zinc toxicity, judiciously and efficiently employed increased monocytes for phagocytic function in view of the fact that neutrophils, the other members of the phagocytic machinery have shown a dip under stress. Ishikawa et al. [18] and Devi et al. [3] also reported increase in monocytes and decrease in neutrophils in fish following |
| subjection to heavy metals. Eosinophils and Basophils however did not record any appreciable change in zinc treated fishes (Fig. 4). In sevin treated fishes, monocytes exhibited decline in their number similar to lymphocytes as the chronicity of sevin increased compared to control (Table 4 and Figs. 11 and 13). |
| However, rest of WBCs i.e. neutrophils, eosinophils and basophils depicted decline (Table 4 and Fig. 13) throughout the experimental period of 60 days in contrast to control (Fig. 12). The decline implies that the phagocytic activity of the sevin treated fishes gets almost totally hampered. During the course of present studies, another interesting observation worth mentioning is that macrophages make their appearance in general circulation from 2nd week onwards (Fig. 7) in zinc treated fishes and from only 5th day onwards (Fig.14) in sevin treated fishes. Macrophages otherwise are not the normal constituents of blood but get surfaced in general circulation only under the stressful conditions to enhance the phagocytic activity of the blood components [13]. Present author proposes appearance of macrophages simply strengthen the phagocytic activity of fish for getting rid off debris produced by breakdown of RBCs under stress of zinc and sevin. From the above discussion, it can be deduced that sevin appears more deleterious to WBC than zinc. Under zinc exposure, the fishes were able to stimulate their immune system in order to overcome the zinc-mediated stress by showing increase in TLC in blood. On the other hand, the fish succumbs to the chronic stressful conditions caused by sevin toxicity and its immune system gets totally paralyzed thereby resulting in drastic decline in TLC in the general circulation. Moreover, such fishes can become easily susceptible to diseases and infections thereby affecting their health status. |
| Thrombocytes: Presently, thrombocytes were found to exhibit significant decrease (P<0.01) in their number (Table 2 and Fig. 5) in all zinc treated fishes while in sevin treated fishes, these depicted a significant increase (P<0.01) in all exposures (Table 4 and Fig. 13) compared to control (Figs. 1 and 11). In zinc treated fishes, marked decline in their number may possibly unable to arrest the internal bleeding and hence ultimately may result in haemorrhagic condition under chronic zinc stress. Additionally, decreased thrombocytes also appear to be incapable to contribute to the phagocytic activity of the fish under chronic zinc toxicity. Increase in thrombocytes in sevin treated fishes, it may be mentioned can cause arrest of any sort of internal bleeding. Moreover thrombocytes being phagocytic in function, their increased number may contribute in the strengthening of the phagocytic machinery of the fish under sevin toxicity. |
CONCLUSION |
| On the basis of above discussion, it can be inferred that sevin appeared to be more toxic in nature compared to zinc. Sevin caused drastic decline in both RBC and WBC dependent parameters of fish whereas zinc caused decline in RBC dependent parameters but increase in WBC dependent parameters thereby depicting the higher toxicity level of sevin than zinc. It is therefore strongly recommended that industrial and agricultural discharge(the main source of zinc and sevin ) should be properly treated before disposing their runoff in the aquatic ecosystem otherwise such xenobiotics when introduced in excess into the aquatic ecosystems are capable of affecting the aquatic life negatively. |
ACKNOWLEDGEMENTS |
| The authors extend heartfelt thanks to the Department of Zoology, University of Jammu, Jammu for providing necessary facilities regarding the research work. |
References |
|
 |
 |
 |
 |
 |