ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
|
Dr. Sheetu Raina1, Dr. Anupriya Sachar2 Ph.D., Deptt. Of Zoology, University of Jammu, J&K, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The present study examined the histological response induced by food deprivation in a fish, Labeo boga inhabiting Jammu waters. Stress of prolonged starvation results in alterations in the histological architecture of the haemopoietic organs viz., liver, head kidney and spleen leading to their dysfunctioning. Liver exhibited necrosis, distended sinusoids besides total degeneration. Kidney Showed tubular vacuolation and necrosis while necrosis and deposition of haemosiderin pigments were the causatives observed among splenic tissue during the experimental period of 60 days
Keywords |
| Stress, Haemopoietic tissue, Starvation, Labeo boga. |
INTRODUCTION |
| Starvation is one of the important causative of fish mortality in nature and in aquaculture [1]. Long-term starvation can cause severe deformity in vital organs [2] and even mortality of fish. Starvation also exhibits a widespread histological degeneration in the haemopoietic organs of fish which can bring about alterations in their cellular architecture. Haemopoietic organs (liver, head kidney and spleen) have been reported to be the most sensitive tissues to be affected by starvation [3]. Structural alterations/degeneration of haemopoietic organs impairs its functional capacity (haemopoiesis) which may prove even fatal for the survival of fish. Thus histopathological alterations in haemopoietic organs can be utilized as tools in order to get a clear idea about the extent an organism is affected at tissue or cellular level [4 and 5]. Presently the exposure of fish Labeo boga to starvation period of 60 days has been observed to result in various histological alterations in the haemopoietic organs viz., liver, head kidney and spleen. Starvation may disrupt the haemopoeitic tissue by disintegrating the functional and structural properties which may directly affect the metabolism of fish and can definitely diminish its health fitness. The paper is organized as follows. Section II describes the methodology adopted for the research work. Section III represents the results of effect of starvation on liver, head kidney and spleen in the form of a, b and c section. Section IV and Section V reflects the discussion and conclusion part of the research work respectively. |
II. MATERIALS AND METHODS |
| a) Sampling site The fish, L. boga (Ham.) for present study were netted from Nagrota stream of River Tawi. The area is located about 8-12 kms from Jammu city, J&k, India. b) Histological analysis After acclimatization, healthy specimens of Labeo boga (size ranging between 10-15cm) were divided into control and fasting group in triplicates. Each group contained ten individuals. Control groups were fed with commercially available pellet fish feed twice a day whereas the starved groups were deprived of food for an experimental period of 60 days. The test fishes were dissected open in ringer solution and their liver, head kidney and spleen were fixed in Bouin’s fixative. After post fixation treatment and routine dehydration and clearing, these tissues were embedded in histowax of 54-56ºC. 5- 7 μm thick section of these was cut on microtome and stained using haematoxylin eosin stain. |
III. RESULTS |
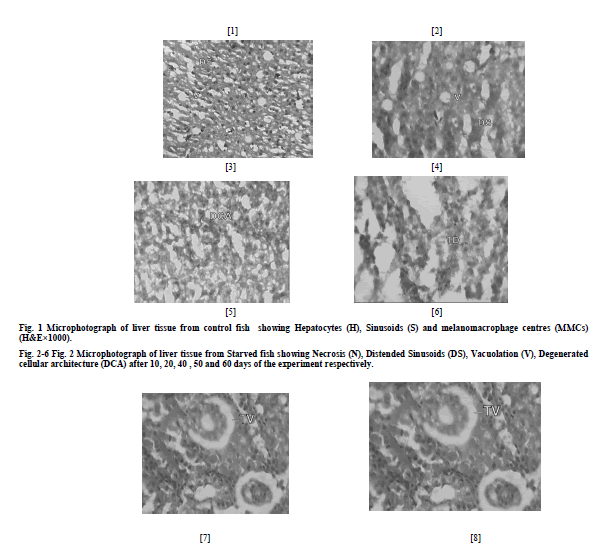
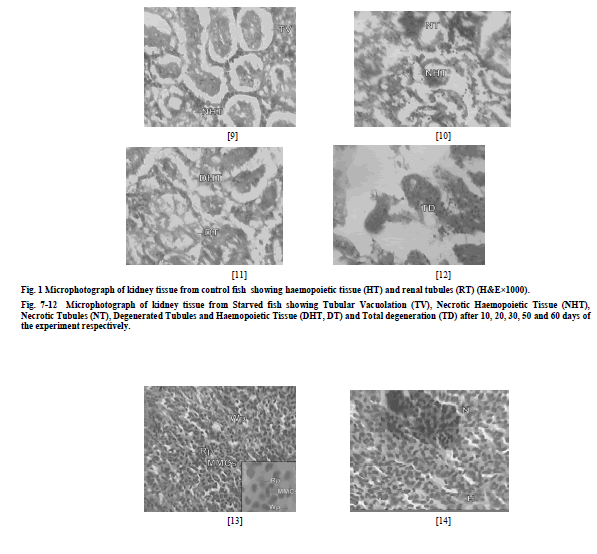
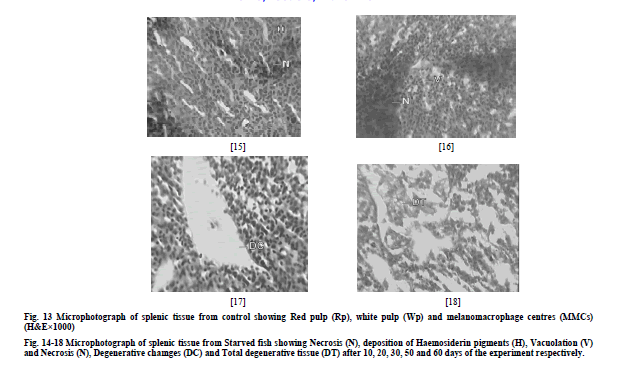
| a) Liver The microscopic examination of the histology of liver in control group showed the presence of hepatocytes (H), blood sinusoids (S) and melanomacrophage centres (MMCs) (Fig. 1). Liver of starved fish depicted i) necrosis of hepatocytes and distension of sinusoids (DS) after 10 days of the experiment (N) (Fig. 2), ii) vacuolation of hepatocytes (V) (Fig. 3) after 20 days of the experiment iii) Massive distension of sinusoids and vacuolation was also observed after 40 days of the experiment (Fig. 4) iv) extensive vacuolation and sinusoidal distension resulted in overall degeneration of cellular architecture (DCA) of liver tissue and loss of hepatocellular mass by 50th day of starvation (Fig. 5) v) after 60 days of starvation, liver tissue depicted degeneration (TD) to such an extent that the hepatocytes were totally lost and the tissue was left with extensive distended sinusoids resulting in its decrease (Fig. 6). b) Head Kidney The histological examination of kidneys of control fish showed the presence of renal tubules (RT). The interstices of renal tubules were enriched with the haemopoietic tissue (HT) (Fig. 7) Kidneys of starved fish after 10 days of starvation exhibited i) mild tubular vacuolation (TV) (Fig. 8) ii) tubular vacuolation in kidney became more pronounced by day 20th of starvation (Fig. 9) iii) haemopoietic tissue was also observed to exhibit degenerative changes (Fig. 10), after 30 days of starvation period, iii) kidney tissue showed the initiation of process of necrosis of tubules (NT) and haemopoietic tissue (NHT) (Fig. 10) iv) necrotic tubules and haemopoietic tissue of kidney was observed to undergo acute degeneration from 50th day of starvation (Fig. 11), v) total degeneration (TD) of kidney was observed after 60 days of starvation, where tubules were seen to lose their normal architecture and the haemopoietic tissue appeared almost negligible (Fig. 12). c) Spleen The microscopic examination of histology of spleen of control fish showed the presence of red pulp (RP) and white pulp (WP). Splenic tissue also represented the dark staining melanomacrophage centres (MMCs) (Fig. 13) Histology of spleen in starved fish depicted i) initiation of mild necrosis (N) and presence of accumulated haemosiderin pigments (HP) (Fig. 14) by day 10th of starvation which got more pronounced at the end of 20 days of starvation (Fig. 15) ii) After 30 days of starvation, spleen tissue was seen to exhibit intense necrosis and even initiation of vacuolation (V) (Fig. 16) iii) after 50 days of starvation, acute degenerative changes (DC) were observed in splenic tissue (Fig. 17) iv) these changes became more and more acute and ultimately by day 60th day, spleen exhibited degeneration (TD) with almost total loss of normal cellular architecture (Fig. 18). |
IV. DISCUSSION |
| Various histological alterations in the liver of starved fish exhibited necrosis, distension of sinusoids, vacuolation and degeneration of cellular architecture of liver tissue. Necrosis is an advanced and irreversible stage of degeneration and is characterized by dead hepatic cells. Pal [6], Sarkar et al. [7], Camarago and Martinez [8], Pathan et al. [9] and Radhakrishnan and Hemalatha [10] also reported necrosis of liver hepatocytes in different fishes following stress of starvation or xenobiotics. In the presently studied starved fish, necrosis appears to have caused damage and death of hepatic cells in the liver. Though liver has its own regenerating capacity [11] but under the influence of prolonged starvation, necrosis may cause degeneration of liver hepatocytes to such an extent that its regenerating power may get lost almost totally. This ultimately results in vacuolation of liver tissue thereby impairing its synthetic machinery and thereby may create an imbalance in the rate of synthesis and release of substances in the systemic circulation. Under the influence of prolonged starvation, therefore, it appears that liver hepatocytes lose their integrity (due to necrosis and vacuolation) and hence the energy reserves in the form of glycogen and lipids stored in hepatocytes may apparently get depleted. Since glycogen and lipids stored in liver hepatocytes are the only source of energy for the fish during starvation, their pronounced depletion provoked by starvation, lands the fish in extremely worst condition. Similar viewpoint has been put forth by Gisbert et al. [12] and Rios et al. [13] in fish liver tissue following starvation. Liver tissue of starved fish also depicted distension of sinusoids which became more massive as the experimental prolonged further. Distension of sinusoids seemingly may be due to the mechanical obstruction of blood supply to liver tissue. Therefore it is the combined effect of necrosis, vacuolation and distension of the sinusoids which seemingly appear to result in overall degeneration of the cellular architecture of liver tissue towards the end of the starvation period. The total degeneration of the liver tissue may definitely results in disruption of various metabolic processes including erythropoiesis. All these histopathological observations indicate that exposure of fish Labeo boga to prolonged starvation caused destructive changes in its liver tissue ultimately disrupting the functional efficiency of liver. Presently, microscopic examination of the head kidney of starved fish showed tubular vacuolation which became more pronounced with the advancement of the starvation period ultimately leading to necrosis of tubules and even haemopoietic tissue. The kidney tissue was observed to undergo total degeneration with loss of normal architecture of tubules and haemopoietic tissue by the end of starvation period. Such histological variations observed in kidney tissue are in conformity with the findings of Velmurugan et al. [14], Mohamed [15] and Prashanth [16] in fishes when exposed to different periods of starvation. . Degenerative changes in kidney tissue as observed presently can be causative of different excretory disorders and internal exaution. Moreover continuous decline in the volume of haemopoietic tissue of the kidney can lead to impairment of haemopoiesis. This may result in disruption in the blood forming efficiency of head kidney and hence inhibits further release of normal erythrocytes into the general circulation. Thus prolonged starvation degrades the normal architecture of the fish kidney thereby affecting its functional efficiency which has a direct influence on physiology of fish. Spleen of the starved fish exhibited conspicuous histological alterations viz., necrosis, appearance of haemosiderin pigments, vacuolation and total degeneration of the splenic tissue following prolonged starvation. Necrosis of splenic tissue followed by Vacuolation surfaced due to starvational stress. Spazier et al. [17] also observed necrosis and vacuolation in splenic tissue of European eel Anguilla anguilla following stress resulting in impairment of normal physiology of fish. Haemosiderin is one of the breakdown products of Hb from senescent and degenerated erythrocytes [18]. Deposition of haemosiderin leads to pathological state known as haemosiderosis. Hibiya [19] also reported haemosiderosis and attributed it to an increased rate of erythrocyte destruction in the spleen resulting eventually in a decreased number of mature erythrocytes in general circulation. Similar deposition of haemosiderin pigments has been observed in the spleen of presently studied starved fishes also. |
| Under the influence of prolonged starvation, the histological architecture of splenic tissue manifested severe degeneration ultimately affecting its blood cell forming capacity i.e. both erythropoiesis as well as leucopoiesis. |
V. CONCLUSION |
| From the above discussion, it can be concluded that prolonged starvation caused structural abnormalities in the haemopoietic organs viz., liver, head kidney and spleen of the fish Labeo boga resulting in severe physiological problems. These conditions can cause even the death of the fish. The findings of the present histological investigations demonstrate a direct correlation between stress of starvation and histopathological disorders observed in several tissues. It is therefore recommended that long term food deprivation should be avoided in aquaculture to maintain healthy conditions for the better survival of fishes. |
ACKNOWLEDGEMENTS |
| The authors extend heartfelt thanks to the Department of Zoology, University of Jammu, Jammu for providing necessary facilities regarding the research work. |
 |
 |
 |
References |
|