ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
R.Rajesh Kumar1, K. Jeyasubramanian1, G.R.Raghav2, S.Anand3 and G.Jeyachandran3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Nano particles use in the electrochromic layer yields less input voltage and consume less power for large surface area. The WO3 is one of the extensively deliberated electrochromic materials. In this work, we prepared WO3 on the aluminum foil via electro chemical deposition. The structural features and the electrochromic behavior of the WO3 thin film are examined by SEM, XRD and cyclic voltammogram with the K+ intercalation using KI as the electrolyte. The cyclic voltammogram indicates large area of the intercalation /deintercalation with K+ ions overcome the difficulties of the intercalation with Li+ ions.
Keywords |
| Tungsten oxide, Thin film, Nanoparticles, Electrodeposition, Electrochromism, Intercalation |
INTRODUCTION |
| The electrochromic (EC) materials have the ability to change its optical properties when voltage is applied [1]. The persistent change in optical properties is attributable to insertion/extraction of electrons and counter ions of the substrates [1,2]. Among inorganic materials, tungsten oxides have been almost extensively studied and famous multifunctional material regarding both hydrogen and solar energy. Also, it exhibits highest coloration efficiency (CE), have wide band gap, n-type metal oxide semiconductor that permits the insertion of positive cations (H+, Li+, Na+ and K+) into its host lattice [3]. |
| Tungsten oxide has wide varieties of applications especially in gas/chemical sensors [1], optical devices [2], pseudo-capacitors [3], electrochromic devices [4], photochromic sensitivity [5], optical absorption [6], hole injection/transport layer in organic-inorganic optoelectronic devices [7] photochemistry [8], lithium-ion batteries [9], photocatalytic activity [10], solar cells [11] |
| Thermal stabilizers [12] and Fuel cells [13], etc. Moreover, the EC WO3 has been prepared by various synthetic method such as physical vapour deposition [1,4,9], electron beam evaporation method [2], aerosol assisted chemical vapor deposition [3], vacuum evaporation [5], pulsed laser ablation [6], Hot-wire chemical vapor deposition[7], Continuous flow reactor [10], electrophoresis deposition [11], electrodeposition [12], sol-gel processing [13], and ultrasonic spray deposition [14]. Haidong Zheng et al used electrodeposition method for obtaining WO3 film from the precursor sodium tungstate in aqueous medium. Employing colloidal particles suspended ethanolacetylacetone mixture as electrolyte, M. Cernea prepared BaTiO3 films by electrophoretic method [20]. |
| With this motivation, we report a simple electrochemical method to deposit WO3 on aluminium foil from the suspension of WO3 powder in ethanol and acetone mixture. Electrochromic properties of the deposited film were examined by cyclic voltammetry with KI as electrolyte. The cyclic voltammogram indicates large surface area of the intercalation /deintercalation with K+ ions that overcome the difficulties of the intercalation with Li+ ions. The developed thin film may be used in electrochromic applications. |
MATERIALS AND METHODS |
Materials |
| Ethyl alcohol, Acetone and Aluminum foil 99.98% were purchased from Merck chemicals, India. Tungsten oxide (Extra Pure) was obtained from Loba chemie, Mumbai. All materials used in this study were of research grade. The aluminum foil of size 5 cm x 2 cm x 0.5mm was consecutively cleaned with ethanol and acetone. |
Experimental procedure |
Bulk Electrolysis |
| 5gms of WO3 powder was mixed with equivalent amount of acetone and ethanol, solution stirred for 30 minutes using magnetic stirrer. The electrodeposition was carried by bulk electrolysis method in electrochemical workstation at constant voltage of -1.5V for 15minutes using aluminium foil as working electrode, Ag/AgCl as reference electrode and platinum wire as counter electrode. The deposition was done under stirring condition to get uniform deposition over aluminium foil. After the electrolysis the film was detached, dipped gently into distilled water for the removal of impurities (2-3 times). After drying at room temperature for 24 hrs, the film was kept at 100°C for 2 hrs. |
Characterization techniques |
| The crystallographic structure of the species deposited was determined by X-ray diffraction (XRD) with a powder diffraction setup Richard Seifert & Co, Ahrensburg, Germany using monochromatic Cu Kα radiation at a voltage of 40 kV over the range of 2θ = 10 – 80o. The surface morphology of the nanoparticles was examined by SEM (Hitachi SUI510 by 10 kV Voltage). Electrochemical characteristics were done using CHI660C, Electrochemical workstation, USA., |
RESULTS AND DISCUSSION |
Surface morphology Analysis |
| The surface morphology of the WO3 deposited on the Aluminum foil has been analyzed using scanning electron microscope. Fig.1 is the SEM image of the WO3 film recorded at the magnification of 10k with detector distance at 8.7 mm at the scale of 5 μm and the size of the nano particle ranges from 60-70 nm of WO3 coated on Aluminum foil. |
 |
XRD Analysis |
| X-Ray diffractogram of WO3 film grown on Aluminum foil is depicted in fig. 2. The XRD of WO3 have characteristic diffraction peaks originating from various planes of WO3, the peaks at 23.0890, 23.5770, 24.295°, 26.57°, 28.73°, 33.325°, 34.077°, 35.077°, 41.62°, 47.22°, 48.29°, 49.88°, 50.51°, 54.19°, 55.70°, 60.5° and 62.230 peaks are labeled as (002), (020), (200), (120), (112), (022), (220), (122), (222), (004), (040), (004), (322), (331), (420), (051) and (242) respectively as per (JCPDS card No. 01-083-0947). Also the lattice parameters evaluated for WO3 matches well with triclinic geometry [15]. |
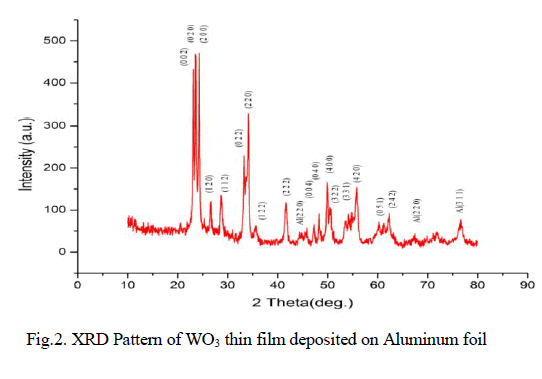 |
| Since WO3 particle are grown over aluminum foil, the peaks at 2θ = 45.25°, 67.23° and 78.44° originally from Al crystal planes are also indexed as (2 0 0), (2 2 0) and (3 1 1) planes as per (JCPDS card No. 01-089-4037) [17]. From the line broadening effect observed in the Xray pattern, the average crystallite size of WO3 nanoparticle was calculated using Scherrer equation [17] and found as 30.5nm. |
Cyclic voltammetry Analysis |
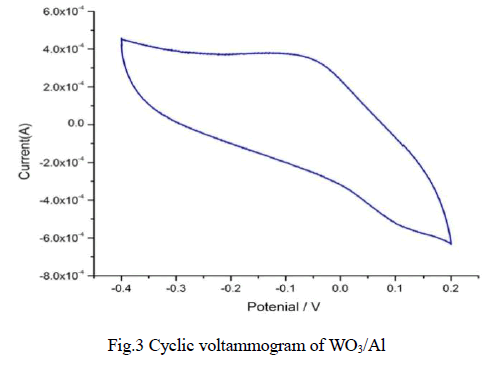 |
| The electrochromic properties of WO3. were examined through cyclic voltammetry. Cyclic voltammetry analyses were done by using three electrodes setup in that WO3 coated aluminium foil as the working electrode, platinum wire as the counter electrode and Ag/AgCl as the reference electrode at scan rate of 10 mVs-1 in 0.1M of KI as electrolyte at room temperature. The voltogramm were scanned in the range of -0.4V to 0.2V. WO3 coated film showed the switching characteristics due to the intercalation/deintercalation of K+ ions [2-3] present in the KI electrolyte. The intercalation/deintercalation effect of as-deposited WO3 film with the electrolyte is given by the following equation. |
| WO3 + xK+ + xe- = KxWO3 [9] |
| WO3 film exhibits its electrochemical response corresponding to the anodic peak voltage at -0.0623V with a cathodic peak voltage noted down at 0.0214V. The CV of WO3 film possesses broad anodic and cathodic peaks with large area. The large area found in between the voltammogram is a measure of its electrochemical activity, which is best suited for electrochromic applications. From these results, it is apparent that, the asdeposited WO3 film exhibit electrochromic property. |
CONCLUSION |
| In this paper we have shown a simple method to prepare WO3 thin film on the aluminum foil by electrodeposition processes at room temperature. The film showed enhanced electro active properties in 0.1 M KI solution when compared to Li+ and H+ ions. After annealing, the film may be used in electrochromic studies, normally thin films are aged for several hours before intercalating with the Li+ ions. This method proves to be the cost efficient method for the preparation of electrochromic thin film used for smart glass, solar cell and gas sensor applications. |
ACKNOWLEDGMENT |
| The authors would like to thank the Management and Principal of Mepco Schlenk Engineering College and KLN College of Engineering for their constant encouragement, support and providing all the essential facilities for completing this research work. |
References |
|