ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Ms.E.Jamuna, Mr.S.Duraisivam , Mr.N.Gurumoorthi, Mr.S.Subash Assistant Professor, Department of Mechanical Engineering, The Kavery Engineering College, Tamilnadu, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
This work concern hydrocarbon gas sensor principle by transferring a novel zeolite-based on impedimetric, which has been originally manufactured by comprising only thick-film processes and one electroplating step. The sensing effect is based on a thin chromium oxide layer between the inter digital electrodes and a Pt-loaded ZSM-5 zeolite film. When hydrocarbons are present in the sensor ambient, the electrical sensor impedance increases strongly and selectively. The chromium oxide film is electroplated on Au screenprinted inter digital electrodes and then oxidized to Cr2O3. The electrode area is covered with the screenprinted zeolite. The sensor device is self-heated utilizing a planar platinum heater on the backside.
Keywords |
| chromium oxide layer, ZSM-5 zeolite film, Cr2O3, digital electrodes, screen-printed, planar platinum heater. |
INTRODUCTION |
| In the present study, the technology transfer of an earlier zeolite-based sensor concept that employed a combination of thin-film processes, photolithography, and thick-film processes to a robust device manufactured without thin-film and lithography processes shall be studied. It is the aim of this study to demonstrate that the technology transfer was successful, to obtain initial data on the long-term stability of the senor devices, and to test its cross sensitivity towards several gas components that might be present in real exhaust. |
| EXPERIMENTAL PRINCIPLE OF ZEOLITES |
| Zeolites are aluminosilicates built up with SiO4 and AlO4 tetrahedra building blocks that form a ring structure. They form 3-dimensional (3D) frameworks with linked channel systems and well defined microand mesopores and provide an open porosity that gives rise to an by cations that are bound to the host framework but are mobile along the channels. |
| zeolites are of high interest in the field of gas sensing . This relatively young but emerging field is reviewed just recently making clear that due to their adsorptive, high surface area and porosity. |
| The aluminum ions act as acidic sites to catalyze chemical reactions. Zeolites can also be modified in a post-synthesis step by incorporating catalytically active metal clusters, e.g. Pt or Fe. Due to their unique property spectrum. |
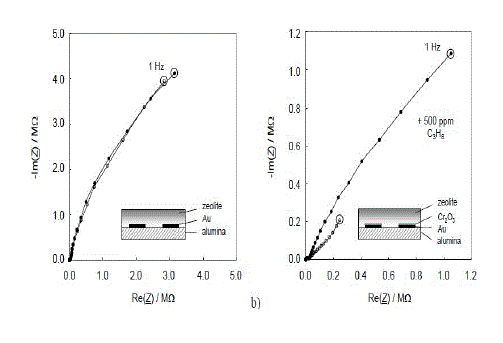 |
| Fig.1. Line graph shown a gas and 500 ppm C3H8 added to the base gas of 2.5 % H2O and 10 % O2 in N2. T = 300 °C. |
| a) Without Cr2O3 layer on the IDE (b) with Cr2O3 interfacial layer. |
| Cr2O3 film on the zeolite-based impedimetric sensor is clearly demonstrated in Fig.1.Two sensors are shown, one without and one with Cr2O3 interfacial layer (Fig. 1a and Fig. 1b, respectively). Due to the preparation in lift-off technique, in the latter case, the spaces between the fingers of the interdigital electrodes are not Cr2O3-covered. The respective sensor setup is diagrammatically shown in the sketches (insets). The sensors were operated in a tube furnace at 300 °C in a base gas mixture of 2.5 % water vapor and 10 % oxygen. 500 ppm propane was added when indicated. |
| The sensor effect is a strong increase of the low frequency impedance when hydrocarbons are admixed. It occurs only in the presence of a metal oxide interfacial layer between the zeolite and the electrodes (Fig. 1b). If the zeolite/Cr2O3 interface is missing (Fig. 1a), the sensor is not sensitive to hydrocarbons at all. As shown in, an additionally sputtered Cr2O3 layer between zeolite and electrode of 100 nm thickness increases the effect, operated at a constant frequency. |
EXPERIMENTAL PROCEDURE |
| Gold interdigital electrodes (lines spaces = 100 μm) were screen-printed with DuPont 5744L paste on alumina substrates (CeramTec Rubalit 708 S) and fired at 850 °C. |
| Electroplated chromium was applied in a chromic acid (H2CrO4) electrolyte (Atotech CR 843) and subsequently oxidized to Cr2O3. |
| A current of 30 mA with a maximum voltage of 3V was applied to the interdigital electrodes for 20s. |
| The hexavalent Cr6+ is deposited as elemental Cr on top of the electrodes. |
| Experiment observations |
Methods |
 |
| Fig.2. Micro view of the sensor during the chromium electroplating and oxidation Process. |
| (a) Electroplated interdigital electrode area |
| (b) Details with an erroneous structure |
| (c) With oxidized Cr2O3 layer. |
| We are clearly identifies by its color an interruption of a finger, which may be caused by an erroneous screenprinting process. The interrupted fingers are not chromium-covered and remain golden. |
| In Fig. 2c, an oxidized chromium film is shown. The sensor shown on the bottom of the picture represents the pure chromium film, whereas the interdigital electrode area (above) is oxidized. |
| A solution of (NH3)4Cl2*H2O) in water was used and the Na- ZSM-5 powder was added and stirred for 24 hours at room temperature. The dried powder was reduced in a H2/N2 gas flow in a fluid bed at 450°C. Highly dispersed platinum clusters inside the zeolite structure were formed. |
| After screen-printing, the organics were removed by a heat treatment at 450 °C for 6 hours. Additionally, on some sensor elements a screen-printed heater structure was applied on the backside of the substrate before printing the gold interdigital electrodes. |
| The entire setup resembled the ammonia sensor in. The temperature homogeneity at 350 °C was determined by an infrared camera and was found out to be better than 5 °C over the entire interdigital electrode area. |
RESULTS |
| Figure 3 shows a typical impedance spectrum of such sensors. The data are taken in a tube furnace at 350 °C in a base gas of 10 % O2 and 2.5 % H2O in N2. All Nyquist plots show a semicircle at high frequencies and a low frequency “tail”. In the complex plane, the impedance shows a hydrocarbon-dependent part at low frequencies. At higher frequencies, a semicircle, which is attributed to the volume properties of the zeolite, occurs. Its size and shape does not depend significantly on the hydrocarbon concentration in the base gas. A sensor without electroplated Cr2O3 film was also prepared and tested. |
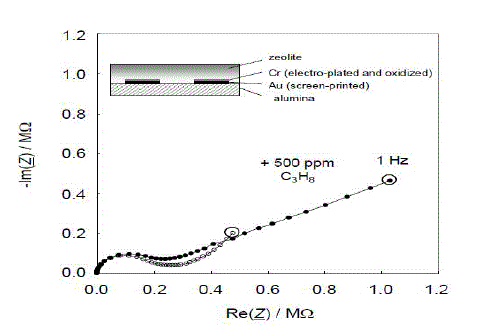 |
| Fig.3 Graph of a thick film sensor with and without 500 ppm C3H8 at 350 °C |
| The impedance response to sudden hydrocarbon changes was evaluated at different temperatures from 250 °C to 400 °C. The measured impedances for each temperature are normalized to compare the signal height and the response times. |
| The fig.4 shows sensitivity S is below 100 %. Between 300 °C and 350 °C the sensitivity reaches its maximum, but the impedance still needs ¾ h to reach 90% of its final value. A response time in the range of 10 min occurs at 400 °C, but here the sensitivity gets reduced. As a result, for further tests approx. 350 °C were chosen as a compromise between response time and sensitivity. |
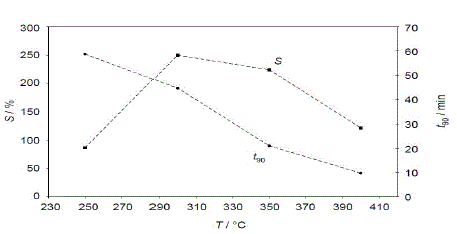 |
| Fig.4 Sensitivity (left axis) and sensor response time (right axis) at a fixed frequency of 3 Hz. |
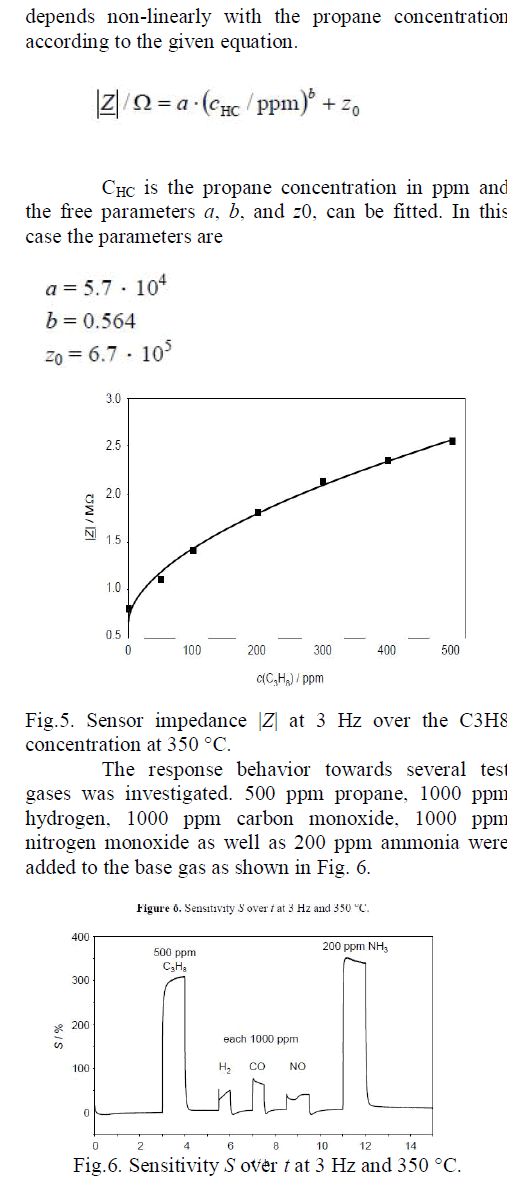
| The sensor characteristics (also at 3 Hz) is shown in Fig. 5. The sensor is exposed to different propane concentrations up to 500 ppm propane. |Z| |
 |
CONCLUSION |
| The sensor was self-heated to around 350 °C, the optimum operation temperature with respect to sensitivity and response time. Best results are obtained at a low measurement frequency of 3 Hz. |
| The sensor remains stable for at least 55 h. The good selectivity of the original sensor was confirmed, but additionally a very strong cross-sensitivity to ammonia was found, which might prohibit its original intention for use in automotive exhausts. |
References |
|