ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
M. Ismail Shareef1, P. Jagan Mohan Reddy1*, S. M. Gopinath1**, K. S. Dayananda1, Ajay Mandal2, Purushotham K.M***
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Postprandial hyperglycemia is a prime characteristic of diabetes mellitus and has been a focus in the therapy for diabetes. One of the therapeutic approaches which involve decreasing hyperglycemia aims at inhibiting the enzyme α-amylase. The leaves of T. Cordifolia were studied for the presence of amylase inhibitors. In this study area, herbal remedies are considered convenient for management of Type 2 diabetes with postprandial hyperglycemia due to their traditional acceptability and availability, low costs, lesser side effects. Comparative evaluation of alpha amylase inhibitory activities of selected plants extracts. The extracts obtained were subjected to in vitro alpha amylase inhibitory assay using starch azure as a substrate and porcine pancreatic amylase as the enzyme The fractions obtained by successive fractionation using solvents of varying polarity were studied for the presence of primary and secondary metabolites. The total phenolic content of the different fractions was determined by HPLC and was correlated with their amylase inhibitory potential. Similarly, the protein content of the extracts was also estimated to understand the nature of the inhibitor present. This study shows that the leaves of T. Cordifolia were effective in inhibiting α-amylase, thereby proving to be potential anti-hyperglycemic agents.
Keywords |
| α-Amylase, Tinosporacordifolia, hyperglycemia. |
INTRODUCTION |
| Diabetic mellitus (DM) is a condition arising due to abnormal metabolism of carbohydrate, promoted by factors, namely, insulin deficiency and/or insulin resistance. This disorder prevails worldwide with its occurrence increasing at an alarming rate globally. Various complications encompass all the vital organs of the body as a consequence of the metabolic derangement in diabetes [1, 2]. The treatment of DM is based on parenteral insulin and oral antidiabetic drugs. Oral hypoglycemic agents include sulphonylureas, biguanides, and other drugs like a carbose. These drugs have serious side effects and deleterious contraindications [2]. Hence herbal remedies having high therapeutic efficacy with minimal side effects are favoured. The antidiabetic agents from medicinal plants are very promising and traditionally acclaimed medicinal plants are being investigated for their antidiabetic potential [3, 4]. Nearly 200 species of plant with hypoglycemic properties have been studied [5]. Highly processed, calorie-dense, nutrient depleted diet leads to exaggerated postprandial spikes in blood glucose and lipids that induces immediate oxidative stress. Induction of oxidative stress has been observed to increase in direct proportion to the increase in postprandial blood glucose level. Postprandial hyperglycemia culminates in type II diabetes mellitus and ensues in the formation of advanced glycationend products. These glycated products are the promoters of diabetic complications and aging [1]. Hence, one of the important therapeutic approaches for diabetes is to decrease hyperglycemia [6]. Herbal medicines with antidiabetic potential have different modes of action—mimic insulin, act on insulin secreting beta cells, or modify glucose utilization. Herbs which modify glucose utilization act by altering the viscosity of gastrointestinal contents, delaying gastric emptying, or delaying glucose absorption [2]. Absorption of glucose can be delayed by reducing the rate of digestion of starch. Inhibition of the mammalian alpha amylase enzyme in the intestine would delay the degradation of starch and oligosaccharides to monosaccharides before they can be absorbed. This would decrease the absorption of glucose and consequently reduce postprandial blood glucose level [5]. Therefore, screening of alpha-amylase inhibitors in medicinal plants has received much attention. Tinosporacordifolia is a climbing shrub found throughout India, typically growing in deciduous and dry forests. Its preferred scientific name is Tinosporacordifolia (L.) Belongs to family Menispermaceae. The young leaves and green fruits are said to be edible. The stem and leaves possess astringent, hepatoprotective, antioxidant, antimicrobial, and anti-inflammatory activities. The antidiabetic effect of the stem of T. Cordifolia has been well established [7]. However, the potential of the leaves of T. cordifoliahas not been much studied, except for a study by Parthasarathy et al. [4], using the methanolic extract of the T. Cordifolia leaves. The present investigation was initiated to screen the leaves of T. Cordifolia for amylase inhibitory activity and correlate it with the polyphenolic content of the leaf extracts. The ethno-botanical information reports about 800 plants may possess anti-diabetic potentials [15] various natural sources including plant products have been investigated with respect to glucose production suppression from carbohydrates in the gut or glucose absorption from the intestine |
II. MATERIALS AND METHODS |
Collection of Plant Material |
| The plant material (leaves) was collected from Bangalore region India, in January, 2012. The plant material was duly authenticated by a Botanist and the specimen is maintained in the department. |
Preparation of Extract |
| The leaves were air-dried, pulverized by grinding using mortar and pestle. Thereafter, the coarse powder of air-dried leaf were subjected to successive solvent extraction by maceration for 72h using solvents of increasing polarity in petroleum ether, chloroform, ethyl acetate, and methanol. Filtered contents were distilled, evaporated, air-dried, freezed, and stored in air tight plastic containers. The respective extractive yields of the extracts were calculated. |
Analysis of Primary Metabolites |
| The primary metabolites like carbohydrates, total proteins, and lipid contents were quantified. Carbohydrates were quantified by the method of McCready et al. [8], proteins by Lowry et al. [9], and lipids by Zak et al. [10]. Analysis of Secondary Metabolites Secondary metabolites like tannins, phenols, and flavonoids were quantified in all the individual extracts. Estimation of Total Phenols The total phenolic content of the purified fractions was determined using the FolinCiocalteau method reported by Singleton and Rossi [11]. Briefly, to 0.1 mL of the extract, 0.5 mL of FolinCiocalteau reagent and 5.0 mL of sodium carbonate were added. The reaction mixture was allowed to stand for 30 min and the absorbance was measured at 640 nm. Gallic acid was used as the standard. Extracts were analysed in triplicates. Estimation of Total Tannins Total tannins were estimated by the method of McDonald et al. [12]. 1 mg of each of the extracts was weighed and dissolved in 10 mL of methanol water (7 : 3). To this 0.5 mL Folin’s phenol reagent (1 : 2) followed by 5 mL of 3.5% sodium carbonate was added and the color intensity was read at 640 nm after 5 min. Extracts were analysed in triplicates. |
Estimation of Total Flavanoids |
| The total flavanoid content of the purified fractions was determined using the aluminium chloride method reported by Zhishen et al. [13]. To 1 mL of the extract, 4 mL of H2O and 0.3 mL of NaNO2 (5%) were added. After 5 min, 0.3 mL of AlCl3 (10%) was added followed by 2 mL of NaOH (1 M). The final volume was made up to 10 mL with H2O and the solution was mixed well. The absorbance was read at 510 nm. Quercetin was used as the standard. Extracts were analysed in triplicates. |
Estimation of Total Phenols by HPLC |
| The total phenolics in both the extracts were detected using a suitable analytical column with the stationary phase Octadecylsilyl silica and mobile phase [A—phosphoric acid : water (0.5 : 99.5 v/v)—B—acetonitrile]. Gallic acid, pcoumaric acid, ellagic acid, ferulic acid, mandelic acid, and vanillic acid were used as reference compounds. 20 μL of the test solution and reference solutions were injected into the column. The detector used for analysis was a UV detector, set at 220 nm with a flow rate of 1.0 mL/min. Assay of Amylase Inhibition In vitro amylase inhibition was studied by the method of Bernfeld [14]. In brief, 100 μL of the test extract was allowed to react with 200 μL of α-amylase enzyme (Hi media Rm 638) and 100 μL of 2 mM of phosphate buffer (pH-6.9). After 20-minute incubation, 100 μL of 1% starch solution was added. The same was performed for the controls where 200 μL of the enzyme was replaced by buffer. After incubation for 5 minutes, 500 μL of dinitrosalicylic acid reagent was added to both control and test. They were kept in boiling water bath for 5 min. The absorbance was recorded at 540 nm using spectrophotometer and the percentage inhibition of α-amylase enzyme was calculated using the formula |
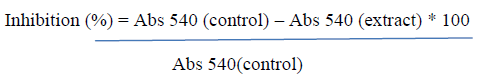 |
| Suitable reagent blank and inhibitor controls were simultaneously carried out. |
Activity Staining of Amylase |
| Activity staining of amylase was done according to the method of Scandalios [15]. The gel consisted of 1% agar in 0.4 M phosphate buffer of pH 7.5. The plant extracts (1 mg/mL) that were pre incubated with the enzyme were loaded in to different wells. Untreated enzyme served as a positive control in a separate well. The buffer used in the gel was also used in the electrode compartments. A stabilized current of 100 V was passed through the gel for 2 h at 4°C. For visualization of the amylase bands the tray was immersed in 0.5% soluble starch and incubated at 37°C for 30 min. The excess starch was then washed and the gel was flooded with iodide potassium iodide solution for 1 min. Colourless bands against a deep blue background indicated amylase activity. |
III. RESULTS AND DISCUSSION |
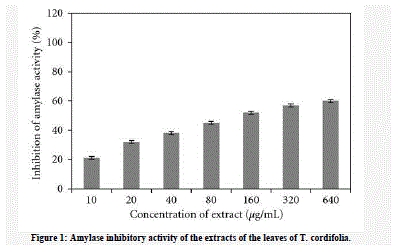
| Hyperglycemia has been a classical risk in the development of diabetes and the complications associated with diabetes. Therefore control of blood glucose levels is critical in the early treatment of diabetes mellitus and reduction of macroand micro vascular complications. One therapeutic approach is the prevention of carbohydrate absorption after food intake, which is facilitated by inhibition of enteric enzymes including α-glucosidase and α-amylase present in the brush borders of intestine. In this study, the α-amylase inhibitory activity of the leaves of T. Cordifolia was investigated. The inhibitory effect of petroleum ether (PETC), chloroform (CTC), ethyl acetate (EATC), and methanol (MTC) extracts was analysed. The percentage inhibition of α-amylase by the extracts of T.cordifolia was studied in a concentration range of 10– 640 μg/mL. Of the four extracts, EATC and MTC were comparatively effective than PETC and CTC in inhibiting α- amylase (Himedia RM638). |
| The IC50 of PETC and CTC was 100 and 120 μg, respectively, and methanol extracts was 20 μg. The EATC and MTC exhibited a maximum inhibition of 98% at 50 μg/mL concentration. The percentage inhibition of all the extracts was not dose dependent (Figures 1(a)–1(d)). |
 |
| The inhibitory effect of all the extracts was also analysed using amylase zymogram. The effects of the inhibitor of two different concentrations were studied. The concentration chosen for EATC and MTC was 1 0 ± I C5 0 and that chosen for PETC and CTC extracts was 2 0 ± I C5 0. Complete inhibition of amylase was observed when the concentrations of the extracts were above the IC50 value. The activity of the extracts was compared with the enzyme control which exhibited a distinct achromatic band against a dark blue background on the agar gel. At concentrations below the IC50 value, faint colorless bands could be observed with EATC and MTC and this indicates partial inhibition of the α- amylase (Figure 2). This indicates inhibition of amylase activity and utilization of starch substrate. |
 |
| Enzyme inhibitors may be proteinaceous or nonproteinaceous in nature. Hence the inhibitory activity of the extracts was correlated with their protein and polyphenolic content. The phenolic content of the extracts was also studied by HPLC. The total phenolic content of MTC was 6.98 mg and that of EATC was 5.26 mg. The quantity of total polyphenols in PETP and CTC was low (2.88 and 1.89 mg, resp.) when compared to MTC and EATC. The amylase inhibitory potential of EATC was greater than that of MTC, as indicated by the zymogram analysis of amylase. Thus there was no correlation between the total phenolics present in the extract and the amylase inhibitory activity of the extracts. |
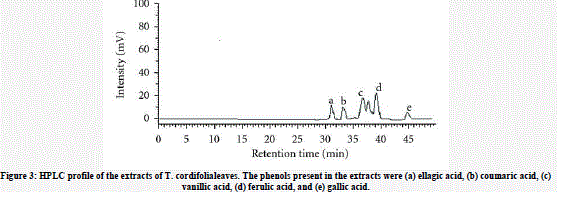
| The presence of phenolic acids in MTC was observed in the decreasing order of concentration of ferulic acid >vanillic acid >ellagic acid >coumaric acid >gallic acid. The phenolic acids in the EATC of T. Cordiofolia were in the following order of decreasing concentration ellagic acid >coumaric acid >vanillic acid >gallic acid (Figures 3(a) and 3(b)). |
 |
| The protein content was found to be high in EATC and low in CTC. The protein content was found in the following order: EATC >MTC > PETC > CTC (data not shown). The protein and phenol analysis of all the four extracts showed that the alpha-amylase inhibitor present in the leaves of T. cordifoliamay be proteinaceous in nature. |
IV. CONCLUSION |
| The current study was to evaluate the amylase inhibitory potential of the leaves of T.cordifolia. The extent of inhibition by the different extracts was compared to the total polyphenolic and protein content of the extracts. The methanolic and ethyl acetate extract of the leaves was found to possess potent amylase inhibitor. The leaves of T. Cordifolia have been also proved to possess antioxidant activity [4]. Thus this study confirms that the leaves of T.cordifolia can mitigate postprandial hyperglycemia and ameliorate oxidative stress and therefore assist in combating diabetic complications. |
References |
|