ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Siva sankara pandian G, Mariaamalraj S and Anant Achary Centre for Research, Department of Biotechnology, Kamaraj College of Engineering and Technology, Virudhunagar, Tamil Nadu, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The present study demonstrates green process for the synthesis of Silver nanoparticles (AgNPs) by bacteria Pseudomonas aeruginosa AMB AS7 and the role of biosurfactant in enhancing the stability of AgNPs. An aqueous solution of silver nitrate was treated with both cell free broth and biomass of P. aeruginosa AMB AS7 which resulted in the synthesis of AgNPs. The nanometallic dispersions were characterized by surface plasmon spectrum measuring at a range of 280 to 700nm. The particle distribution analysis and Scanning Electron Microscopic (SEM) results showed the formation of AgNPs in the range of 30 – 80 nm. Further analysis carried out by Fourier Transform Infrared Spectroscopy (FTIR) showed the presence of functional groups from proteins and biosurfactant. The stability of AgNPs synthesised by both chemical and biological methods was analysed. Biological method of AgNPs synthesis showed remarkable stability. They were stable for 24 days whereas AgNPs synthesised by chemical method were stable for only 3 days. Also the influence of biosurfactant and chemical surfactant on the stability of AgNPs synthesized by chemical method was analyzed. The biosurfactant showed superior property (19 days) over the chemical surfactant (7 days) in terms of increasing the stability of AgNPs. The use of bacterium for nanoparticles synthesis and the influence of biosurfactant on the stability of AgNPs offer the benefits of eco-friendliness and amenability for large-scale production
Keywords |
| Silver nanoparticles, biosurfactant, stabilizing agent, biological synthesis, Pseudomonas aeruginosa. |
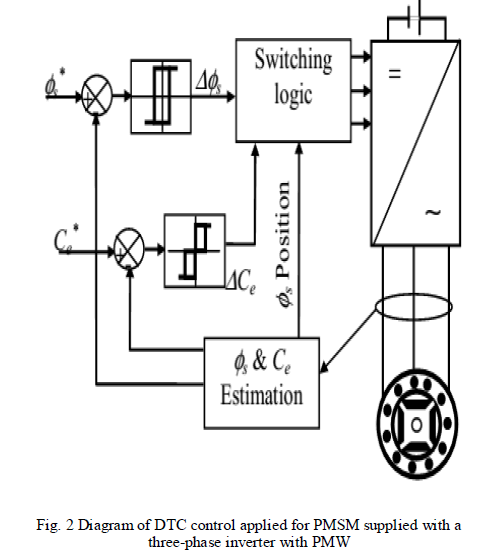
INTRODUCTION |
| Silver nanoparticles (AgNPs), as one of nanomaterials of noble metals, have extensive applications in many fields. They are widely used as an additive for numerous materials and products including plastics, ceramics, glass, cement, rubber, lubricants, paints, ointments, adhesives, pigments etc. They can also be used for antibacterial materials, antistatic materials, cryogenic superconducting materials, and biosensor materials. Synthesis of AgNPs has attracted a lot of attention due to their unusual optical [1], photoelectrochemical [2] and electronic properties. The general methods of synthesizing AgNPs is by physical method, which yield low amounts of product and generates lots of heat and the chemical protocols generally applied for synthesis of AgNPs, suffer from one or the other limitations like high cost, use of toxic chemicals, etc. |
| Many microorganisms such as bacteria, yeasts, fungi and actinomycetes have been used in synthesis of metal nanoparticles. Microbial synthesis of metal nanoparticles can take place either intracellularly or extracellularly [3- 5]. Additional downstream processing steps like ultrasonication, cell disruption by chemicals to release AgNPs are required in case of intracellular synthesis of AgNPs [6]. On the other hand extracellular biosynthesis is cheap and AgNPs can be separated easily which favours large-scale production of AgNPs to explore its potential applications. Due to this, many studies were focussed on extracellular methods for the synthesis of metal nanoparticles[7]. The approach on using culture supernatants from different bacteria, yeasts, fungi and actinomycetes for the synthesis of silver nanoparticles is well documented. |
| One of the hurdles in AgNPs synthesis and its application is stability. Recently, surfactants are emerging as potential nanoparticle stabilizing agents. However, the synthetic surfactants are not eco-friendly and toxic. Therefore, the biosurfactants can be a better, green alternate for the synthesis and stabilization of nanoparticles. It also helps in synthesis of monodispersed, uniform nanoparticles. |
| In this view, the present studies focused on biosynthesis of silver nanoparticles using bacteria, Pseudomonas aeruginosa AMB AS7, their characterization using UV-visible spectrophotometer, SEM, FTIR, Particle size analyser and study the influence of biosurfactant and chemical surfactant on the stability of AgNPs. To the best of our knowledge, this is the first report on the comparison between biosurfactant, rhamnolipid and chemical surfactant, Sodium dodecyl sulphate (SDS) in enhancing the stability of AgNPs. |
MATERIALS AND METHODS |
Chemicals |
| All the chemicals used are of analytical grade with high purity. |
Medium and fermentation conditions |
| The bacterial strain, Pseudomonas aeruginosa AMB AS7 was isolated from petroleum-contaminated sample and cultured in Nutrient broth. Fermentation was carried out at 37°C for 48 h in conical flasks with agitation at 100rpm. The cell free supernatant was obtained by centrifuging at 7200 x g for 30 min. Both the biomass of Pseudomonas aeruginosa AMB AS7 as well as supernatant was used for silver nanoparticles synthesis. |
Biosynthesis of silver nanoparticles |
| Silver nanoparticle biosynthesis was carried out as described in by Kumar et al., [8]. Briefly, aqueous silver nitrate (50 ml of 1 mM), was added to 50 ml of the Pseudomonas aeruginosa AMB AS7 culture supernatant (50%, v/v) and kept at 37°C. The bioreduction of silver ions was monitored at regular intervals by sampling aliquots (2 ml) of the reaction mixture. UV–visible spectra of these sample aliquots were recorded as a function of time of reaction from 200 to 700nm on a UV– visible double beam spectrophotometer (Shimadzu, PerkinElmer, Shelton, CT, USA) at 37°C. |
Intracellular biosynthesis of silver nanoparticles |
| The P. aeruginosa AMB AS7 cell mass was harvested and washed twice with sterile distilled-water. For the synthesis of silver nanoparticles were carried out by taking 1 g of wet P. aeruginosa AMB AS7 cell mass along with 50 ml of 1 mM AgNO3 in 250 ml Erlenmeyer flasks. All the flasks were incubated in a shaker at 37 °C for 24 h at 100 rpm. After incubation, the cells from each flask were washed twice with 50mM phosphate buffer (pH 7.0) and resuspended. Ultrasonic disruption of cells was carried out with an ultrasonic processor. The ultrasonic conditions were three 15 s ON periods, and with an interval of 45 s OFF between periods. The sonicated samples were taken and the resulting solution was filtered through a 0.22 μ filter to remove cell debris. The sonicated samples were centrifuged at 7, 200 × g for 30 min at room temperature. |
Characterization of silver nanoparticles |
| The nanometallic dispersions of the synthesized AgNPs were characterized by surface plasmon spectrum measuring at a range of 200 to 700nm. Besides, the size and distribution of AgNPs were confirmed by particle distribution and Scanning Electron Microscopic (SEM) analysis. Further analysis carried out by Fourier Transform Infrared Spectroscopy (FTIR) to confirm the presence of functional groups from biomolecules like proteins and biosurfactant. |
Effect of time on stability of AgNPs |
| The stability of AgNPs with respect to time of storage was studied. The both chemically and biologically synthesized silver nanoparticles were subjected to UV– visible scan from 200 to 700 nm at different day interval till 24 days to study the stability. |
Influence of biosurfactant on synthesis of silver nanoparticles |
| To synthesize AgNPs, were prepared according to the procedure by [9] 50 ml of 1 mM AgNO3 was heated to boiling. To this solution, 5 ml of 1% trisodium citrate and both SDS (at critical micelle concentration (CMC), 2.1 mg/ml) as well as rhamnolipid (at CMC, 0.3mg/ml) was separately added drop by drop and without surfactant was used as a positive control. The solution was heated at boiling point under continuous stirring. The reaction was allowed to take place until the color changed to a greenish yellow solution. The solution was then cooled to room temperature. Synthesized silver nanoparticles were confirmed by UV-visible spectrophotometer. |
RESULTS AND DISCUSSION |
Biosynthesis of AgNPs |
| In this study, both the intracellular and extracellular synthesis of silver nanoparticles by the bacterial isolate Pseudomonas aeruginosa AMB AS7 and the influence of biosurfactant, rhamnolipid on the stability of AgNPs are reported. The cell free broth and biomass of Pseudomonas aeruginosa AMB AS7 was mixed separately with silver nitrate solution (1mM) and incubated at 37° C for 24 h. Fig.1 shows the colour change of culture supernatant from pale green to yellowish brown in silver nitrate treated flask whereas no colour changes in the culture supernatant without silver nitrate, during the incubation in same condition. The similar observation was occurred when P. aeruginosa AMB AS7 biomass was used. The synthesized silver nanoparticle was monitored by UV-visible spectroscopy. |
| In the intracellular synthesis of silver nanoparticles, maximum peak was observed in the range of 445 to 450nm.The same kind of results was obtained by [10]. They reported that silver nanoparticle synthesized intracellular by Pseudomonas sp.BB4 showed a strong absorption band at 430 nm after 24 hours which is due to the excitation of surface Plasmon vibrations. Similar result was also reported by [11]. They demonstrated that after 72 h of incubation, silver nanoparticles was synthesized in the cell free supernatant of Pseudomonas aeruginosa. However, in the present study 24 h of incubation was enough for the synthesis. |
 |
Characterization of AgNPs |
| The presence of AgNPs was further confirmed by SEM analysis. The SEM micrograph of the dried silver nanoparticles as shown in Fig. 2a showed the AgNPs structure, morphology and the size of nanoparticles. The image elucidated the particles are predominantly spherical in shape with no well-defined morphology was observed. M Safaepour et al., [13] has reported the nanoparticle in the size range of 50 to 100 nm using Enterobacteriaceae family and geranial, respectively. |
| The energy dispersive X-ray analysis (EDX) as shown in Fig 2b revealed the strongest peak at 3 keV indicated the metallic silver confirmed the formation of silver nanoparticles. However, the other elemental signals such as Cl and O were recorded. The similar results was reported by [8], they obtained the optical absorption peak near at 3 keV indicated the presence of nano-sized metallic silver. Particle size analyze was performed with Zetasizer version 6.32 (Malvern instruments) that gave the particle size and size distribution. Particle size analyzer revealed that the particles obtained are size ranging between 30 nm to 200 nm as shown in the Fig.2c and the average size of silver nanoparticles was found to be 60.44 nm. Similar result was reported by [14] where they obtained silver nanoparticles of size 60 nm. |
 |
 |
| This showed the cell free broth not only reduced silver ions, but it also stabilized the particles. Bacterial synthesis of silver nanoparticles produced more stable particles than chemical method. P. aeruginosa AMB AS7 has the ability to produce rhamnolipid which is a biosurfactant. It could act as a capping agent to stabilize the silver nanoparticles for long time. Fig 4 shows the absorption peaks from day 1 to day 24 which clearly shown the changes in the peak position and intensity. The result proved that the AgNPs synthesized by the supernatant of P. aeruginosa AMB AS7 were stable for 24 days which is a more prominent result of this study. |
 |
| The stability of silver nanoparticles might be due to the presence of biomolecules like biosurfactant, proteins in the cell free supernatant. |
Influence of biosurfactant on synthesis of silver nanoparticles |
| The ability of chemical surfactant, SDS and biosurfactant, rhamnolipid to stabilize AgNPs was studied. The result proved that, biosurfactant is superior to the SDS. The AgNPs synthesized using biosurfactant were stable for 13 days whereas in SDS solution, AgNPs were stable for 5 days. This is one of the prominent results of this study where the biosurfactant, rhamnolipid can be used as a capping agent to enhance the stability. Another notable fact is only 0.3 mg/ml of biosurfactant is required to enhance the stability. On the other hand, 2.1 mg/ml of SDS was used; however the stability was not equivalent to biosurfactant. Hence, less amount of biosurfactant is ample to stabilize the AgNPs when compared to that of SDS. |
CONCLUSION |
| The biosynthesis of silver nanoparticles using P. aeruginosa AMB AS7 could produce more stable nanoparticles compared to chemical synthesis. Also, the results proved that the intracellular synthesis of AgNPs was uniform and monodisperse, however, in terms of yield and cost of production, extracellular synthesis of silver nanoparticles find more advantages. The results also shows that the biologically synthesis of AgNPs requires relatively more time than chemical synthesis. However extraordinary stability, uniformity, size of biologically synthesized nano particles could overcome the above said disadvantage. Besides, the current study demonstrated that the addition of biosurfactant during the chemical synthesis of silver nanoparticles could enhance the stability. Also it proves that the biosurfactant has more stabilizing capacity than that of chemical surfactant, SDS. |
ACKNOWLEDGMENT |
| The authors would like to thank the management and Department of Biotechnology, Kamaraj College of Engineering and Technology, Virudhunagar for the support and facilities provided to conduct the experiments. |
References |
|