ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Shreenidhi, K.S. 1, Niveditha, C.2, Sarayu, K.3, Ramachandra Murthy, A.4, Nagesh R. Iyer5
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Biomineralization is an important and ubiquitous process resulting in biominerals, produced by a range of taxa, from bacteria and to eukarya. Calcium carbonate precipitation was reported in many bacterial species, both in vivo and in vitro conditions which is influenced by various abiotic factors and could transform soluble organic nutrients into insoluble inorganic calcite crystals. Biologically controlled mineralization (BCM) of calcium carbonate involves a remarkable control in size, morphology and phase selection, resulting in complex hierarchical organic- inorganic structures with unusual physicochemical properties. The calcium carbonate thus secreted can be used in various applications and as an additive admixture in building materials. In this study calcareous bacteria were isolated from marine sediments, which were able to produce calcium carbonate crystals by utilizing organic and inorganic nitrogen source indicating the nitrogen and ureolytic cycle as their basic metabolic pathway. Characterisation studies were performed and the most efficient isolate was obtained.
Keywords |
| Bio-mineralization, Bio-minerals, Calcite, Phenomenon, Characterisation studies. |
INTRODUCTION |
| Biomineralization is the process of secreting calcium carbonate, biologically. In other words, it is the synthesis of inorganic crystalline or amorphous mineral-like materials by microorganisms or living organisms. It is quite widespread among the prokaryotic and the eukaryotic organisms [2]. These organisms are known to secrete calcium carbonate naturally. Many bacterial species originated from various natural habitats have been reported to precipitate calcium carbonate [11] in invitro and natural conditions which is influenced by various abiotic factors that determine the route and environment for the calcium carbonate precipitation [10].These are the composite materials that are made up of both mineral and organic components. As they are formed in the controlled conditions, they have distinguishing properties based on the phase properties like shape, size, crystalline nature, elemental composition (isotopic and trace), etc.; hence, they are called as true minerals[1][9]. Biominerals have unusual morphologies as they are decided by the intricacy and diversity of the secreting microorganism [3] which mimics the structure of the microorganism secreting it. Biomineralization process can be divided in to two different groups such as biologically induced and biologically controlled based on the degree of biological control they have over the system of secretion of biominerals. Minerals that are precipitated as a product of the interaction between the environmental and biological activity is termed as biologically induced biomineralization. Minerals that are precipitated as a product of cellular activities of the microorganisms under optimum conditions is termed as biologically controlled biomineralization. Bacteria are capable of forming inorganic crystals intracellularly or extracellularly through different metabolic processes. Calcium carbonate (CaCO3) is one of the most useful, versatile material and also a common filler in many industries (Eg: cement industry). It is critical to the construction industry as marble and as an ingredient of cement. It decomposes to form carbon dioxide and lime, an important material in making steel, glass and paper. |
| Calcium carbonate forms three types of crystals namely calcite, aragonite and vaterite. Calcite crystals are rhombohedron shaped, white, transparent and readily usable form. It is a well-known example of extracellular bacterial biomineralization. The technique is highly desirable because the mineral precipitation induced as a result of microbial activities is pollution free and natural. Aragonite crystals are orthorhombic shaped and formed by biological and physical process. Vaterite crystals are spherical shaped. It converts to calcite at low temperature and to aragonite at high temperature. |
| The calcium carbonate thus secreted can be used in various applications and as an additive admixture in building materials. Bacteria were able to transform soluble organic nutrients into insoluble inorganic calcite crystals which when applied to concrete, seals the cracks. Biomineralization process forms precipitation and fills the pores that enhances the mechanical properties in concrete materials such as strength and durability. |
MATERIALS AND METHODS |
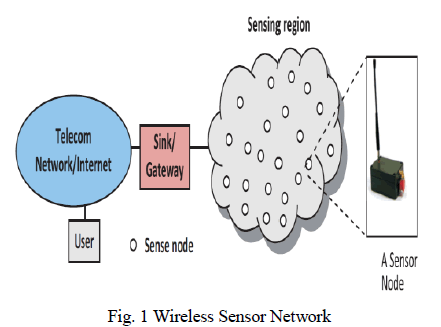 |
| Fig. 1 shows the flow diagram of the process carried out for this investigation. A brief explanation about each step has been discussed below. |
| A. Sample preparation: |
| Bacterial samples were collected from Rameshwaram sea water ,the individual bacterial species were identified by the different plating techniques on nutrient agar. |
| B. Growth in Specific Medium: |
| The bacterial species were grown in specific media for maximum calcium carbonate precipitation which was supplemented with organic and inorganic nitrogen sources. Culturing of bacteria was carried out in both solid and liquid media. The growth and development were observed. |
| C. Characterization studies: |
| The study has evidenced the precipitation of calcium carbonate by bacteria which was confirmed through characterisation studies like optical micro-graphs, SEM and EDS. Elemental, structural, morphological, atomic characterization studies were performed to confirm the presence and metabolic pathway of calcium carbonate crystals. The bacteria cultured in both media were taken for optical microscopic observations in order to identify the presence of calcium carbonate crystals and its location. Structural morphology of calcium carbonate crystals were studied using scanning electron microscope. Elemental characterizations of crystals were done by Energy-dispersive X-ray spectroscopy (EDS). Optical microscopy images were recorded using a Carl Zeiss optical microscope Discovery V20 with Axio vision (Version:4.8.3.0)-64 bits).SEM images and EDS mapping were recorded with SEM - ZEISS EVO 18 to know the surface morphology of the material. |
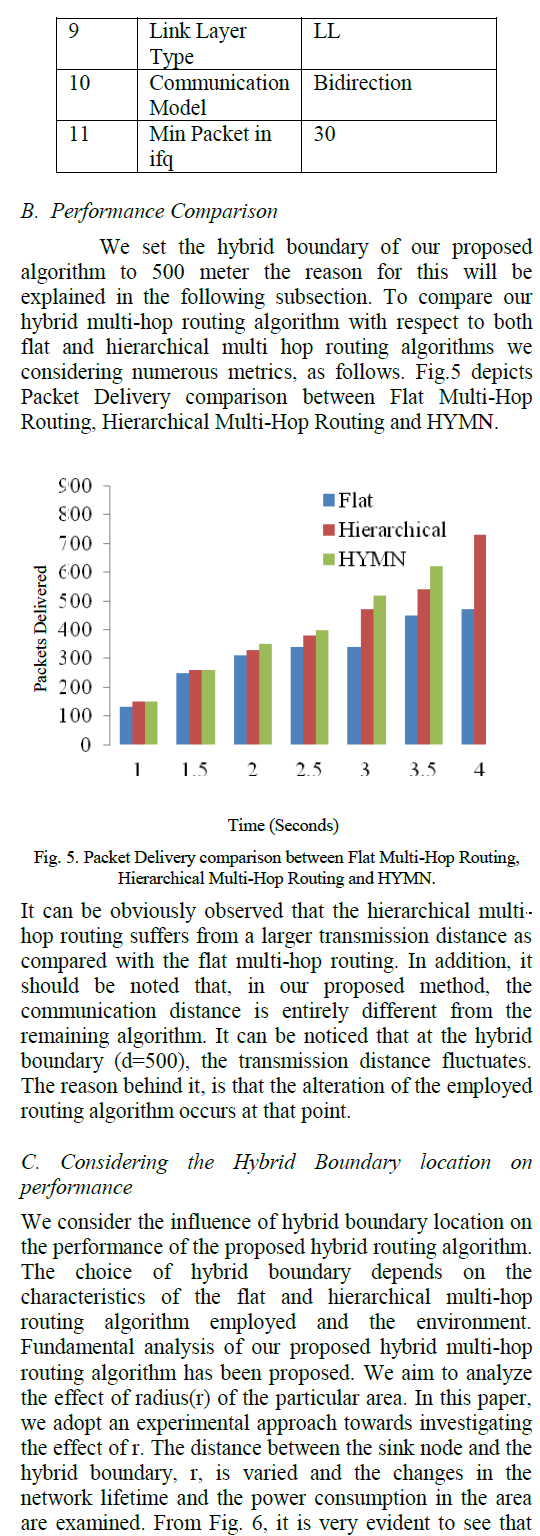
RESULTS AND DISCUSSION |
| A. Identification of bacterial strains: |
| Unique individual colonies were identified based on the colony morphology observed on the nutrient agar plates. |
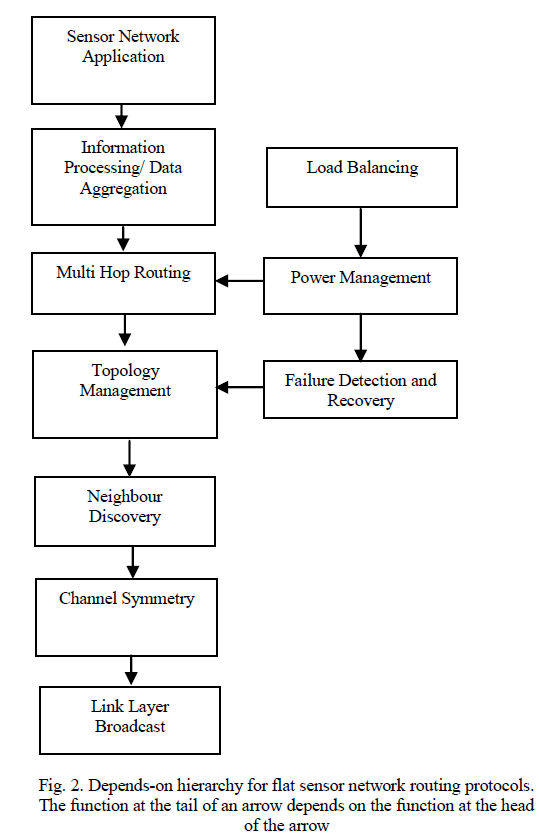 |
| The appearance of colony results from characteristics of the individual bacteria viewed collectively as shown in fig 2.From the nutrient agar plate, the visible characteristics of different colonies were noted. Colonies that differ in appearance were typically different bacterial strains, species or genera. Description of colony’s morphology includes its shape, the margins or edges of the colony, the colony’s colour and surface features [8]. |
| B. Growth in Specific Calcium Media: |
| The bacterial isolates were grown in specific media for maximum calcium carbonate precipitation which was supplemented with organic and inorganic nitrogen sources. Culturing of bacteria was carried out in both solid and liquid media. |
| ïÃâ÷ Inorganic nitrogen source |
| ïÃâ÷ Organic nitrogen source |
| To obtain crystal of considerable size: |
| ïÃâ÷ Isolate 1: organic calcium medium - 4 to 6 days. |
| ïÃâ÷ Isolate 2: inorganic calcium medium- 7 to 8 days. |
| ïÃâ÷ For other two isolates, crystal morphogenesis, development and accumulation were not substantial making the observation difficult as seen in fig 3. |
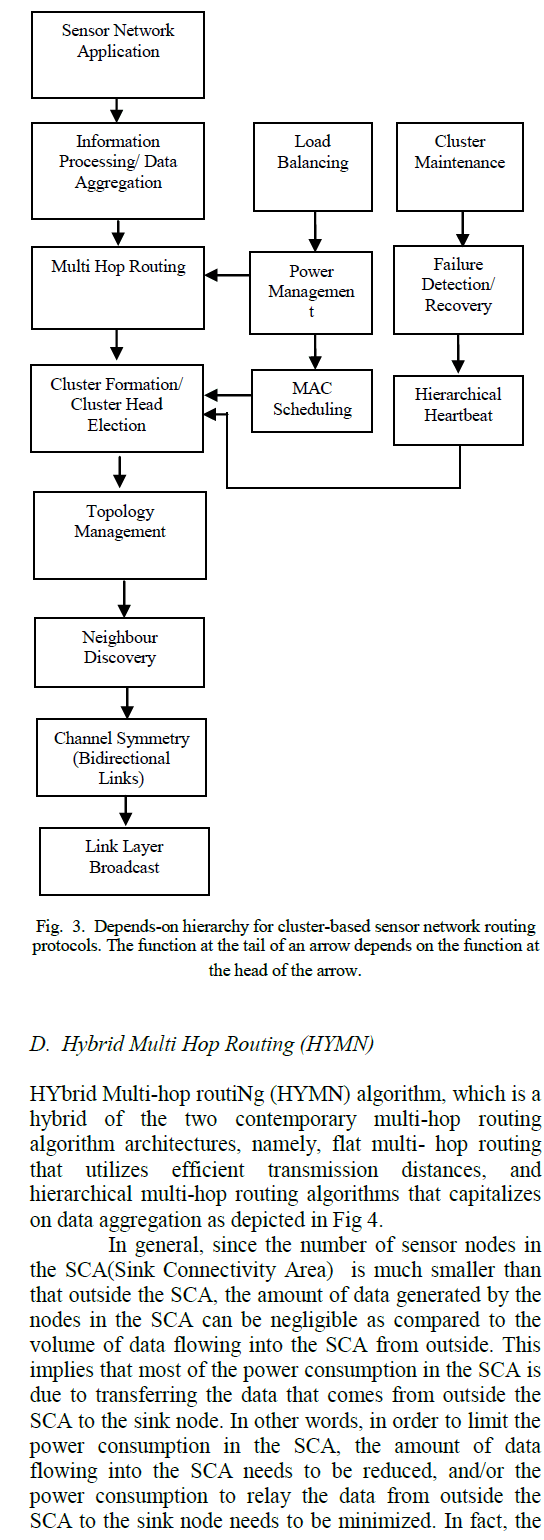 |
| ïÃâ÷ Ammonia on hydrolysis leads to formation of ammonium and hydroxide ions are released. |
| ïÃâ÷ Carbonates formed equilibrate with water and increases the pH. |
| ïÃâ÷ A highly alkaline condition is thus reached. (Extracellular condition = high calcium ion concentration and low protons concentration). |
| ïÃâ÷ Bacteria undergo a stress due to electrochemical gradience. |
| ïÃâ÷ This causes an influx of calcium accumulation and excessive expulsion of protons. |
| This leads to binding of calcium and carbonate forming the calcium carbonate crystal. ATP and proton pump helps in calcium carbonate accumulation. |
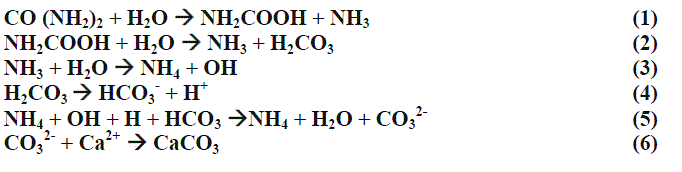 |
| D. Factors Influencing the Calcification Process: |
| Calcium carbonate precipitation mediated by microorganisms is basically decided by six factors: |
| 1. Reactive calcium ions available |
| 2. Concentration of inorganic carbon in the microenvironment |
| 3. pH |
| 4. The availability of an appropriate nucleation site |
| 5. Genetics of urease gene clusters |
| 6. Polymorphs of calcium carbonate crystals[12]. |
| E. Characterization studies: |
| Structural morphology of calcium carbonate crystals were studied using optical microscope. The structure and shape of calcium carbonate crystals was found to be specific for each organism. The location of crystals, secretion and accumulation in the medium varied with each organism. The crystals were exhibited intracellularly or extracellularly. In both solid and liquid media, it was observed that isolate 1 and 3 produced crystals in organic calcium medium and isolate 2 and 4 produced crystals in inorganic calcium medium. Also, isolate 1 and 2 showed colour change in their respective calcium medium as shown in the fig 5 and 6. |
 |
 |
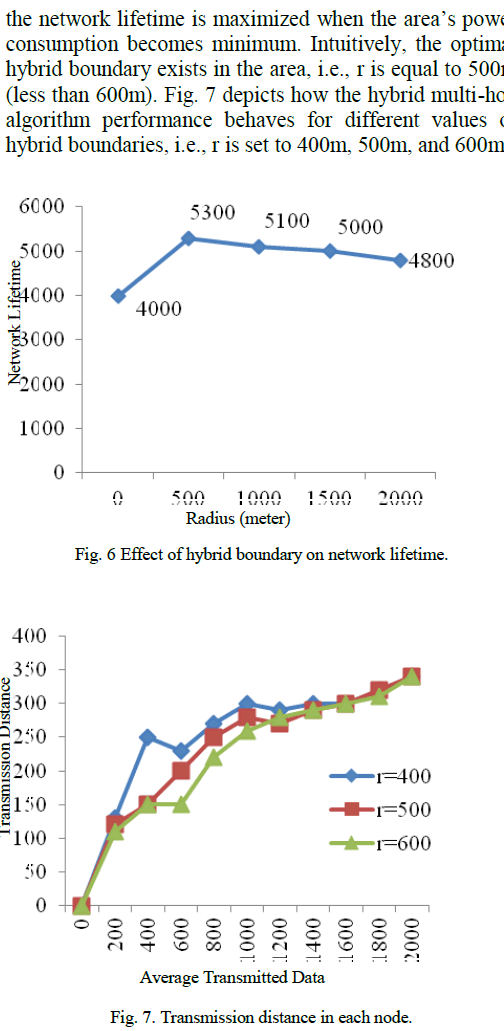 |
| Fig 8 describes the elements along with their weight % present in the bacterial precipitates.The isolate 1 that produced maximum crystals was characterized using Energy-dispersive X-ray spectroscopy(EDS) for elemental analysis. The EDS results indicate the presence of calcium having peaks at different locations. Apart from calcium, magnesium and potassium were also present. Oxygen was also present in higher amount indicating the compound nature of oxygen with all the elements. Elemental composition of the precipitate has demonstrated the presence of 60% oxygen contributed from carbonate, 40% of calcium with 3% of magnesium and 1% of potassium. From the EDS results, it was observed that the organism was capable of secreting 40% calcium carbonate, confirming the existence of calcite as major compound and magnesium-calcite as minor compound unlike the observations made by [4]. |
CONCLUSION |
| It can be concluded that each bacterium is selective to the nitrogen source, which is used in its metabolic pathway to produce crystals. The study has clearly evidenced the precipitation of calcium carbonate by the bacteria with the optical micrographs. The shape and location of crystals precipitated was species specific. From above analysis, isolate 1 calcite production was more and further characterization studies like SEM, EDS were conducted. The crystal was found to be flaky, ball-like structure with size of 10 micrometers The presence of calcium carbonate was confirmed by the EDS results.Microbially induced calcium carbonate precipitation can be used for a number of applications such as the removal of heavy metals , removal of radio nucleotides, removal of calcium from water , biodegradation of pollutants, modification of the soil properties for the enhancement of oil recovery, plugging, strengthening of sand columns. |
ACKNOWLEDGMENT |
| The Authors are thankful to the Director, CSIR – Structural Engineering Research Centre, Chennai for allowing us to carry out this work. The help and cooperation received from scientists and technical staffs of Computational structural mechanic group – Biofacility laboratory are deeply acknowledged. |
References |
|