e-ISSN: 2319-9849
e-ISSN: 2319-9849
Aastha Goyal*
Department of Chemistry, Sri Guru Granth Sahib World University, India
Received Date: 06/09/2018; Accepted Date: 09/10/2018; Published Date: 11/01/2019
Visit for more related articles at Research & Reviews: Journal of Chemistry
In this review article, Metal organic frameworks were prepared by various routes and the prepared MOFs was characterized by various techniques like Fourier transform-infrared spectroscopy (FT-IR), X-ray diffraction (XRD), Scanning electron microscopy (SEM) and Energy Dispersive X-ray (EDX) techniques. MOFs were prepared by combination of various inorganic metals and organic linkers. MOFs have potential for the treatment of various gaseous pollutants. Further, MOFs were used for making composites with the help of doping something like ionic liquid, graphene oxide, CNTs etc. and these MOFs composite helpful in wide range of applications like gas storage, biomedical applications and catalysis and detection of heavy metal ions. MOFs have broad of advantageous properties (e.g., high surface area, high degree of porosity, specific adsorption affinities).
Pharmacology, Calibrants, Relaxation, Deprotonation, Vacuum
Porous materials encompass a broad range of uses in the industrial processes such as absorption and catalysis and they are basically classified as zeolites (inorganic hybrid), polymers (organic hybrid) and metal organic frameworks (inorganic-organic hybrid) [1]. Metal organic frameworks (MOFs) are the microporous material (<2 nm), they are formed by linking metal (it can be ion and a metal node) and organic linker by coordination bond [2]. The organic linkers are molecule or ion that can donate lone pairs of electrons to the metal ions, whereas the metal ions have vacant orbital that can accept these lone pairs of electrons from organic linkers to form a MOFs material. MOFs are subset of coordination networks which are again subset of coordination polymers. The topology of MOFs are depend on geometry of ligands and stability of MOFs is determined by metal-linker combination [3].
They are called THE FUTURE because their composites such as ionic liquid - MOFs, graphene oxide-MOFs etc. are used to capture carbon dioxide which is a greenhouse effect and also store hydrogen gas which is a future fuel [4]. They have other applications such as dye degradation [5], detection of heavy metal ions [6], sensors [7], drug delivery [8], luminescence [9], magnetism [10], gas storage [11] and others has a wide range of applications as they have great porosity, specific adsorption affinities and specific surface area. MOFs are interesting over conventional materials like zeolites, COFs and polymers, as various metals and organic linkers unite to form a large number of materials with different crystal structures and chemical compositions. One can tune their properties according to applications that one requires so due to their structural tunability and function ability, MOFs are excellent substitutes of conventional nanoporous material. The limitation of zeolites is that their pores are restricted. It is difficult to alter these materials for specific use as they have rigid tetrahedral oxide skeletons whereas the pore sizes and chemical functionalities of MOFs can be tuned by using different metal nodes to achieve a desired application [12]. They have been studied for a variety of applications, such as gas storage and separation [13], biomedical applications [14], and catalysis [15]. In addition to MOFs, covalent organic frameworks (COFs) are those which have similar materials without metals are called COFs. They have light elements such as H, B, C, N, and O, which form strong covalent bonds with each other [16]. COFs have lower densities than MOFs although they have similar characteristics as MOFs such as large surface areas, high pore volumes, and permanent porosities [17]. The synthesis of metal–organic frameworks (MOFs) involves the reaction of a metal-containing precursor with an organic linker in an organic solvent at an elevated temperature, in what is termed a ‘solvothermal’ reaction. Many more examples have been reported of MOF synthesis in ionic liquids (ILs), rather than an organic solvent, in ‘ionothermal’ reactions [18].
Composites materials are those substances which contain 2 or more materials that combine to produce a new substances with different physical properties from the original substances. When MOFs are doped with some other materials such as ionic liquid, graphene oxide, CNT then composites are formed. As yet, MOF composites have been successfully incorporating with various metal or metal oxides nanoparticles (NPs), carbon materials, polymers, polyoxometalates, ionic liquids, and so on (Figures 1 and 2) [19].
Figure 2. Representation of Organic linkers and Inorganic secondary building units [20].
Design of MOFs: First row transition metals such as Zn, Cu and Co are well known to be able to coordinate with organic groups under various methods like hydro and solvothermal conditions to form crystals.
Nomenclature
Apparently because of the relatively short path of this family of materials, there is not a normalized nomenclature for MOFs (which is used for zeolites).
Some researchers gave the name to metal organic frameworks by consecutive number of synthesis/chronological sequence of findings, initials of organizations or institutions where they were initially prepared or by consecutive of isorecticular synthesis.
Naming by consecutive number of synthesis: In this method, the MOFs were named as MOF-2, MOF-3, MOF-4, MOF-5, so on, where the number which attached to MOFs designated as the chronological sequence of findings C40H56.
Naming by initials of organization or institution or place of finding: The second method to give name to MOFs is done by forming a phrase or an acronym from the name of an organization, place or any institution where these crystalline MOFs was actually prepared e.g., HKUST-n36 (Hong Kong University of Science and Technology), UiO-66 (University of Oslo).
Naming by consecutive of isorecticular synthesis: Isoreticular means same topology and the naming of MOFs were based on the topology of organic linkers that means they share a common cubic topology which were made from the same type of organic linkers. e.g., IRMOF-1 and IRMOF-0 where IRMOF is Isoreticular metal organic frameworks.
An alternative to this illustrative name consists of using the “empirical formula” of the material, i.e., expressing the metal(s), the ligand(s), and their stoichiometry in the repetitive unit, e.g., [Zn4(O)(bdc)3] (bdc)1,4-benzenedicarboxylate) [21].
History
The very first metal organic framework synthesized was MOF-2 which has its formula Zn (BDC) (H2O) where BDC means 1, 4-benzenedicarboxylate. Other such materials were synthesized which possess porosity were MOF-3 [Zn3(bdc)3 ] and MOF-4 [Zn(btc)NO3) where btc means 1,3,5-benzentricarboxylate [22]. Werner has researched on coordination complexes. At that time many porous crystals were also been identified [23]. Those identified porous crystals did not exhibit permanent porosity. Porous crystal having permanent porosity was invented in 1990s [24]. Yaghi was the scientist who has given the name to these porous crystals that was “Metal Organic Framework” [25].
The most flexible MOF named MOF-5 having neutral framework of composition Zn4O(BDC) in which Zn4O groups are linked by terephthalate where BDC means 1,4-dicarboxylate was synthesized in 1999 [26]. Its name was taken from a known zeolite ZSM-5 [27].
The routine synthesis of metal organic frameworks were done by solvothermal synthesis and hydrothermal methods. The other possibility for the synthesis of metal organic frameworks were done by, microwave-assisted synthesis, electrochemical synthesis, mechanochemical synthesis and sonochemical synthesis. Among all these methods, solvothermal and hydrothermal were popular techniques for the synthesis of MOFs because it is simple and easily controlled process. However, it has some disadvantages also such as it is time consuming and large particle size.
Solvothermal Synthesis
Metal–organic frameworks (MOFs) in which the reaction includes metal-containing precursor with an organic linker in an organic solvent at high temperature, in what is termed a ‘solvothermal’ reaction. In this method a mixture of both organic linkers and metal were heated at high temperature organic solvent. The thermal energy (353 K-453 K) is applied to this process and this process could last between 48 to 96 hours. The organic solvents which were often used in solvothermal reactions were DMF (dimethylformamide), DEF (diethyl formamide), ethanol, methanol, acetone etc. For different kind of starting matter there will be a differing or varying solubility and to avoid this difficulty mixtures of solvents have been used. Solvothermal reactions can be carried out in variety of temperature ranges, depending on the required reaction. For the synthesis of inorganic compounds and MOFs (inorganic and organic hybrid), hydrothermal method has been successfully used [28]. For example, Senkovska et al. synthesized two aluminium based metal-organic frameworks [Al(OH)(ndc)(DMF)1.5(H2O)1.5] and [Al(OH)(bpdc)(DMF)1.8(H2O)3.5] by solvothermal method using N,N-dimethylformamide as a solvent. In a typical synthesis, 2,6-naphthalene dicarboxylic acid or 4,4´-biphenyldicarboxylic acid respectively, was dissolved in DMF.Al (NO3)3.9H2O was added and the mixture was filled in a Teflon liner, placed in an autoclave, heated to 120°C for 24 h and cooled to room temperature. After the product was separated by centrifugation, the sediment was washed with DMF for three times and the product was dried on air [29]. Reinsch et al. synthesized the system Al3 +/H3BTB/DMF/additive was systematically investigated using high-throughput methods and the new, microporous MOF [Al (BTB)] (BTB=1,3,5-benzenetrisbenzoate), named CAU-4 (CAU=Christian-Albrechts-University). The optimized synthesis of CAU-4 in the HT reactor system is as follows: Al (NO3)39H2O, benzoic acid and 1,3,5-benzenetrisbenzoic acid were dissolved in N,N-dimethylformamide. The reactor was heated for 24 h at 180°C. The reaction can be scaled up to the fourfold amount, using Pyrex glass tubes, or to the tenfold amount, using larger steel autoclaves with Teflon inserts. The same molar ratios Al3 +: H3BTB: benzoic acid: DMF=7:14:10:142 as well as the same temperature program were used. A yield of 47% (based on H3BTB) was obtained from the reaction in the 30 ml reactor [30]. Pradip et al. synthesized fluorinated MOFs by Solvothermal reactions of Cu(NO3)2.3H2O with 4,40 -(hexafluoroisopropylidene) bis(benzoic acid) (C17H10F6O4, H2hfbba) and terminal monodentate ligand 3-methyl pyridine (3-picoline/3-mepy) in the presence of N, N-dimethyl formamide (DMF) and N,N-diethyl formamide (DEF) solvents gave rise to two structurally different two dimensional (2D) fluorinated metal organic frameworks (FMOFs). The effect of the choice of solvent has been clearly reflected in the structures obtained. The F-MOFs reported in this paper are formulated as [Cu2(hfbba)2(3-mepy)2]. (DMF)2(3-mepy) (F-MOF-4), [Cu2(hfbba)2(3-mepy)2] (Cu-F-MOF-4B), and [Zn2(hfbba)2(3-mepy)2]3(3-mepy) (Zn-F-MOF-4B) which displays interesting 2D structures with and without interdigitation depending on the solvent used.
Synthesis of [Cu2(hfbba)2(3-mepy)2] (Cu-F-MOF-4B): 0.5 mL of 3-methyl pyridine stock solution and 1.5 mL of H2hfbba solution (0.20 M) in DEF were mixed in a 5 mL vial. 0.5 mL of Cu (NO3)2 .3H2O solution (0.20 M) in DEF was added to this solution and it is heated at 85°C for 96 h and the products were washed with DEF (15 mL) three times. Dark blue colored crystals of Cu-F-MOF- 4B were collected by filtration and dried in air.
Synthesis of [Cu2(hfbba)2(3-mepy)2]3 DMF)2(3-mepy) (F-MOF-4) 9b: 0.5 mL of 3-methyl-pyridine stock solution (0.20 M) and 1.5 mL of H2hfbba stock solution in DMF (0.20 M) were mixed in a 5 mL vial. 0.5 mL of Cu(NO3)2. 3H2O stock solution (0.20 M) was added to this solution and it is heated at 85°C for 96 and the products were washed with DMF (15 mL) three times. The blue colored crystals were collected by filtration and dried.
Synthesis of [Zn2(hfbba)2(3-mepy)2]3(3-mepy) (Zn-FMOF-4B): 0.5 mL of 3-methyl pyridine stock solution and 1.5 mL of H2hfbba solution (0.20 M) in DEF were mixed in a 5 mL vial. 0.5 mL of Zn(NO3)2 6H2O solution (0.20 M) in DEF was added to this solution and it is heated at 85°C for 96 h and the products were washed with DEF (15 mL) three times. Colorless crystals of Zn- F-MOF-4B were collected by filtration and dried in air [31].
Tian et al. synthesized nickel based MOF by using methanol as solvent. Ni(NO3)3.6H2O and 2.4 mmol of BTC were dissolved in 70 mL absolute methanol. The mixture was stirred for 1 h at room temperature, and then transferred into a Tefon-lined stainless steel autoclave with a volume capacity of 100 mL and heated at 150°C for 24 h. After the heat treatment, the autoclave is allowed to cool naturally to room temperature, and the products are collected by centrifugation at 10000 rpm for 5 min and washed with absolute methanol several cycles, and then dried at 60°C in vacuum 12 h. Glover synthesized various MOF-74 building units using cobalt, magnesium, zinc, nickel metal centres [32].
Cobalt MOF-74 (Co-MOF-74): In a 400 mL jar, with sonication, 0.5 g of 2,5 dihydroxyterephthalic acid (DHTA) and 1.5 g of Co(NO3)2 .6H2O were dissolved in dimethylformamide, ethanol, and water. The jar was capped tightly and placed in a 100°C oven for 2.75 days. After cooling, mother liquor was decanted and the products were washed with methanol and immersed in methanol. The products were then evacuated to dryness and heated under vacuum to 250°C. After 24 h, the sample was cooled to room temperature and stored [33].
Petit and Bandosz synthesized MOF- 5: MOF-5 was prepared by mixing zinc nitrate hexahydrate and 1,4-benzenedicarboxylate in N,N-dimethylformamide (DMF, 140 mL) until complete dissolution of the solids. Then, the mixture was transferred into a round-bottom flask connected to a condenser and heated at 115-120°C for 24 h. After cooling, the supernatant was removed and crystals deposited on the bottom of the flask were collected, washed with DMF, and immersed in fresh chloroform overnight. The chloroform was changed twice during two days. Finally, crystals were collected, placed inside a closed filtering flask connected to an aspirator, used to create vacuum inside the flask, and heated at 130-135°C for 6 h. The resulting crystals were then kept in a desiccator [34].
Mueller et al. synthesized MOF-2. MOF-2 was prepared by mixing 24.9 g of terephthalic acid (BDC) and 52.2 g of zinc nitrate tetrahydrate (Merck) were dissolved in 43.6 g of N-methyl-2- pyrrolidone, 8.6 g of chlorobenzene and 24.9 g of dimethylformamide and heated up to 70 μC for a total of 3 h. After hour, 30 g triethylamine was added. After filteration, white precipitates was formed which was dried in air. The molar yield based on zinc amounted to 87% [35].
Microwave Assisted Synthesis
MOFs were synthesized very quickly by using microwave-assisted synthesis method. As solvothermal method, this method also make nanosized MOFs crystals very quickly. The advantages of this method includes rapid crystallization, phase selectivity, small particle size distribution, and facile morphology control. The crystals were obtained by heating a solution of metal salt, organic linker, organic solvent which kept in microwave at 303 K-373 K. The kind of crystals which were made by microwave-assisted method were similar as produced by solvothermal method. The first reported MOFs by microwave synthesis was Cr-MIL-100 [36].
Jianwei Ren et al. synthesized Zr-based MOFs by microwave-assisted method: The microwave-assisted synthesis of Zr-MOFs was conducted in a microwave reaction system (Anton Paar Synthos 3000). It was prepared by 0.75 g terephthalic acid and 1.05 g zirconium tetrachloride were dissolved in 40 ml DMF ultrasonically and add 17 ml formic acid to that solution and that solution was divided into four portions. Four microwave vessels were programmed for reaction at 120ºC for 5 min under static conditions. After reaction, the sample was treated in the same manner as the Zr-MOF-Oven sample [37].
Choi et al. synthesized MOF-5 by MW:MOF-5 was synthesized in N,N'-diethylformamide (DEF) using microwave irradiation. Equimolar amount of H2BDC (1.57 mg, 0.60 × 10-2 mmol) and zinc nitrate tetrahydrate (1.50 mg, 0.60 × 10-2 mmol) in a DEF (0.6 mL) were place in a 10 mL tube and the tube was sealed and placed in a microwave oven (Discover, CEM, maximum power of 300 W). The resulting mixture was heated at 95°C, held for 9 min and then cooled to room temperature. The colorless crystalline materials (2.10 mg, 27% yield) were obtained by centrifuging, washing with N,N'-dimethylformamide (DMF, 3 × 5 mL) and drying briefly in the air [38].
In this method of synthesis metal ions were used instead of metal salts and these metal ions were supplied constantly through aniodic dissolution which act as a metal source and these metal ions were react with the mixture that contain both organic linkers and electrolytes. The electrochemical synthesis of MOFs was first reported in 2005 by researchers at BASF for HKUST-1. In this MOFs were synthesized by electrochemical route for e.g. bulk copper plates were arranged as the anodes in an electrochemical cell with the carboxylate linker, viz. 1,3,5-benzenetricarboxylic acid, dissolved in methanol as solvent and a copper cathode a greenish blue precipitate was formed. The pure Cu-MOF was obtained and after activation at 250°C a dark blue coloured solid of octahedral crystals is formed. For certain applications it is necessary to synthesized large crystals under moderate conditions by adjusting some parameters like pH/solvent at room temperature [39].
Mueller et al. synthesized Cu-MOF using an electrochemical route: Bulk copper plates, thickness 5 mm, are arranged as the anodes in an electrochemical cell with the carboxylate linker, viz. 1,3,5-benzenetricarboxylic acid, dissolved in methanol as solvent and a copper cathode. Details are to be found in. 10 During a period of 150 min at a voltage of 12–19 V and a currency of 1.3 A, a greenish blue precipitate was formed. After separation by filtration and drying at 120 uC overnight pure Cu-MOF was obtained which was dark blue coloured solid of octahedral crystals from 0.5 to 5 mm size [40].
Mechanochemical synthesis
Mechanochemical is basically combination of mechanical force and chemical reactions, so it is clear from the name mechanochemical synthesis that it is a solvent free process which can occur at room temperature where MOFs were constructed via a process of performing various chemical reactions by applying mechanical force. To get a desired MOFs, a mixture of both metal salts and linkers were ground together in a ball mill. Mechanochemical synthesis of a Cu-isonicotinic acid MOF was first reported in 2006 by Pichon et al. Recently for the rapid synthesis of MOFs, LAG (Liquid Assisted Grinding) is used. In Liquid Assisted grinding process a small amount of solvent was added into a reaction mixture for e.g., different solvent was added to a mixture of fumaric acid and ZnO, and they form 1D, 2D, and 3D coordination polymers. The liquid can also act as structure directing agent. For the synthesis of pillared MOFs there was expansion of LAG (liquid assisted grinding) to ILAG (ion- and liquid assisted grinding). The advantage of this method is over other method such as sonochemical and microwave assisted synthesis is that mechanochemical synthesis is solvent free while sonochemical and microwave methods were still depend on organic solvent. However, mechanochemical synthesis is limited to specific MOF types only and large amount of product is difficult to obtain and one drawback about this method is that it was limited for particular types of MOFs and immense amount of outcome was hard to acquire [41].
Klimakow et al. carried out mechanochemical synthesis in a conventional ball mill via the liquid-assisted grinding of fine powders of copper acetate monohydrate and 1,3,5-benzenetricarbocylic acid (H3BTC) and copper acetate monohydrate and 4, 40, 400- benzenetribenzoic acid (H3BTB), respectively, in a molar ratio of 3:2 for 25 min. The color of the powder changed during the reaction from deep green to blue, accompanied by the strong odor of the byproduct, acetic acid [42].
Sonochemical Synthesis
Sonochemistry is a process in which molecules undergo chemical change due to the application of intensive ultrasound radiation between 20 kHz and10 MHz. As compared to conventional solvothermal methods this method create homogeneous and accelerated nucleation which can also give a minimal crystallization time and appreciably using sonochemical irradiation in 1- methyl-2-pyrrolidinone (NMP) can produce 5–25 mm crystals in 30 min, which is similar to MOF-5 synthesized by solvothermal or microwave methods [43].
Tahmasian et al. synthesized {[Mg(HIDC)(H2O)2].1.5H2O}n nanostructure by using Sonochemical Method. To prepare nano-sized {[Mg(HIDC)(H2O)2].1.5H2O}n, 20 mL of an aqueous solution of the ligand H3IDC (0.05 M) and potassium hydroxide (0.1 M). Into the solution of H3IDC and potassium hydroxide, add 20 mL of an aqueous solution of magnesium nitrate (0.05 M) was added dropwise. The obtained precipitates were filtered off, washed with water and ethanol, and air-dried [44].
Ionothermal Synthesis
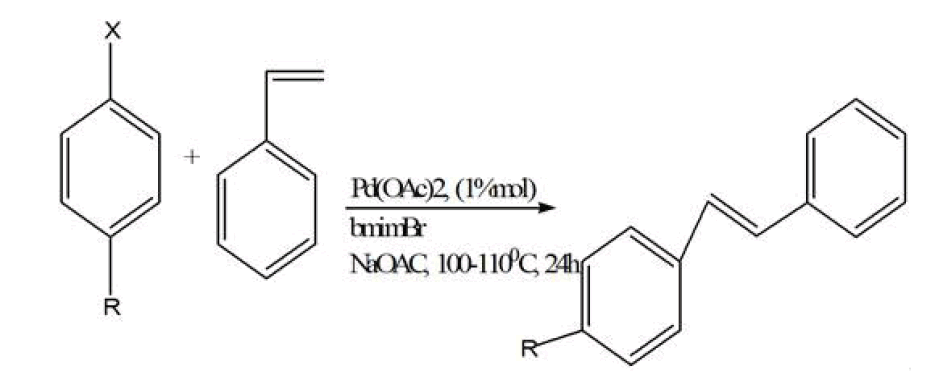
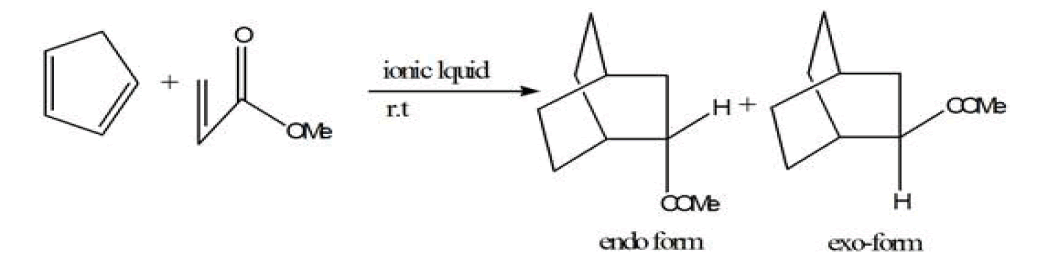
Metal organic frameworks were synthesized in ionic liquid rather than organic solvent is known as ionothermal reactions. Ionic liquids are defined as materials that are composed entirely of ions and that have a melting point below 373 K [45]. Ionic liquids have salient features like low vapor pressure at room temperature,[46] nonvolatile,[47] nonflammable,[48] recyclable, high thermal stability, varying polarity, high conductivity. Ionic liquid are called ‘liquids’ because the cations are asymmetrically substituted with different bulky groups which weaken the ionic interactions and they also prevents packing of the anions/cations into a crystal lattice. The high concentration of both organic cations (e.g., imidazolium, pyrrolidinium, pyridinium,) and inorganic or organic anions (e.g., tetrafluoroborate, hexafluorophosphate, and bromide) in an ionic liquid allows for the formation of new MOF structures by incorporating both anions and cations into the open cavities of MOFs. In this method of synthesis both cations and -anions were trapped or fused into MOFs. In most of the cases cationic part of ionic liquid was included in the pores of MOF, whereas anionic charge of MOFs act as a counter ion and MOF structure forms. Ionic liquid act as solvent and template or structure directing agent. It is synthesized in ionic liquid at room temperature by the starting precursors of MOFs with the help of modulator which initiate the reaction then this mixture is sealed inside Teflon-lined stainless-steel autoclave then it is placed into the furnace by providing particular temperature for some days. Eventually, it is cool down at standard temperature and crystals are collected. Likewise the formation of MOFs by Ionic liquids, ionic liquids also have great importance in the synthesis of organic reactions like Diels-Alder reaction, Heck reaction, etc.
Diels-Alder reaction
Heck reaction