ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Rati Sinha1, H R Singh2, S K Jha2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
L-Asparaginase is a well known enzyme for its chemotherapeutic properties since it catalyzes hydrolysis of the amino acid asparagine into aspartate and ammonia which is essential for tumour cells. It has been effectively used in the treatment of acute lymphoblastic leukemia and lymphosarcoma from decades. This article comprises a brief introduction about the enzyme, its sources, mechanism of action as well as a detailed information about its production and purification.This article also provide some recent updated information about its structure, physiochemical and kinetic properties of the enzyme as well as about modifications such as cloning to get the enzyme in recombinant form, formulation and immobilization of the enzyme.
Keywords |
| Asparaginase, chemotherapeutic, physiochemical, kinetic, immobilization |
I. INTRODUCTION |
| Recombinant therapeutic proteins have made a revolutionary impact in the area of healthcare as these are disease specific, safe and more effective drugs. They have a broad variety of specific uses: as oncolytics, as anticoagulants or thrombolytics and as replacements for metabolic deficiencies. At present about 50 recombinant thereapeutic products have been globally approved for their commercial use of which more than one fourth are already produced into India and have received government approval for their marketing in the country [1]. A major potential application of therapeutic enzyme is in the treatment of cancer.One such therapeutic recombinant enzyme is L-Asparaginase is which has proved to be promising for the treatment of acute lymphocytic leukemia, a childhood cancer which results from the transformation of a clone of cells from the bone marrow where early lymphoid precursors proliferate and replace the normal cells of the bone marrow. |
II. MECHANISM OF ACTION |
| L-Asparaginase catalyzes the hydrolysis of a non-essential aminoacid L-Asparagine into L-Aspartate and ammonia. Lasparagine is an essential aminoacid for the growth of tumour cells whereas the growth of normal cells is independent of its requirement. Most normal tissues synthesize L-asparagine in amounts sufficient for their metabolic needs with their own enzyme, L-asparagine synthetase but the malignant cells requires an external source of L-Asparagine for growth and multiplication. In the presence of L-Asparaginase, the tumour cells get deprived of an important growth factor and cannot survive. This fact suggested the development of this enzyme as a potent anti-tumour or anti-leukemic drug [3]. |
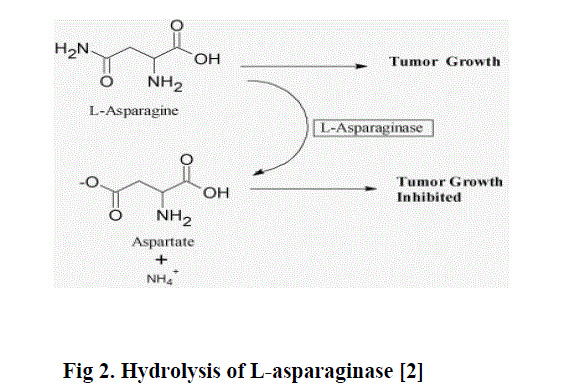 |
III. APPLICATIONS |
| As an antitumour drug - L-asparaginase is a therapeutically important protein in combination with the other drugs in the treatment of various types of blood cancer such as acute lymphocytic leukemia (ALL,mainly in children), Hodgkin disease, acute myelocytic leukemia, acute myelomonocytic leukemia, chronic lymphocytic leukemia, lymphosarcoma treatment, reticulosarcoma, and melanosarcoma surrounding tissue. The first studies showed that intraperitoneal injection of guinea-pig serum-which has a high content of asparaginase produced obvious regression of the Gardner lymphosarcoma in mice [4];[5]. Later on a potent source of L-Asparaginase was found in Escherichia coli [6]. Since then L-Aparaginase derived from E.coli E.C.2 has been used for treating human malignant disease. |
| Role in aminoacid metabolism - L-Asparaginase also play a very critical role in biosynthesis of the aspartic family of aminoacids. Corynebacteria- producing aminoacids are of great industrial interest as they excrete large amounts of various aminoacids [7]. Lysine, threonine and methionine, commercially important aminoacids produced by C.glutamicum, are derived from the aspartic acid, which under normal physiological conditions, might be limiting for lysine and/or threonine biosynthesis. Apart from Kreb’s cycle (using glutamic acid as aminoacid donor), aspartic acid is formed from asparagine by the action of asparaginase. L-Asparaginase was produced constitutively and its role may be that of an overflow enzyme, converting excess asparagines into aspartic acid, the direct precursor of lysine and threonine. A very active L-asparagine was found in C. glutamicum under lysine producing fermentation conditions [8]. |
| Making biosensor - L- Asparaginase has also been used for making a diagnostic biosensor as the amount of ammonia produced by the action of the enzyme directly correlates to the level of L- asparagine in a patient’s blood [9]. |
| Role in food processing - Moreover it is also used in food processing. Recent advances in food technology have demonstrated that fried and baked food (particularly fried potato) contains a significant amount of acrylamide (a carcinogenic toxicant)[10]; [11], formed by the reaction of asparagine with reducing sugars [12]. A pretreatment of potato slices and bread dough with asparaginase before frying or baking prevents acrylamide formation [13] [14]. In this regard, asparaginases from Aspergillus oryzae and A. niger are used in baking industries [15]. These enzymes work optimally at 40-60ºC and pH 6.0-7.0. Since baking temperatures often go up to 120ºC, it is desirable to have enzymes that are stable and active over a wide range of temperature and pH. Therefore, L-asparaginases from various sources (bacterial, fungal, plant, and animal) have been investigated. So, L-asparaginases have both therapeutic and industrial applications. |
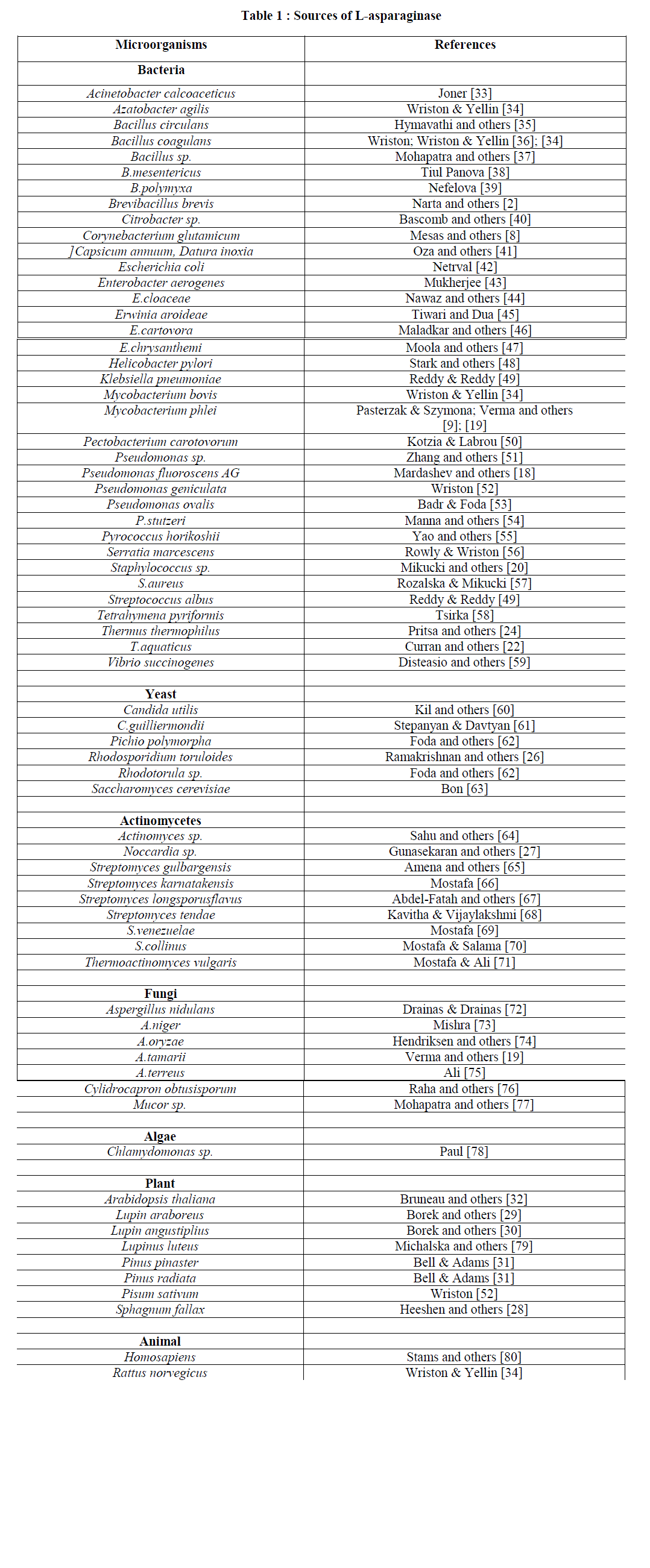
IV. SOURCES OF L-ASPARAGINASE |
| The importance of microorganisms as L-asparaginase sources has been considered since the time it was first discovered from E.coli and its antineoplastic activity demonstrated in guinea pig serum. [5]; [6]; [16]; [17]. Microorganisms such as bacteria, fungi, yeast, Actinomycetes and algae have proven to be proficient sources of this enzyme. Although it was also been isolated from plant and some animals sources. But microbes are a better source of L-asparaginase because of the following advantages: |
| ïÃâ÷ Bulk production capacity |
| ïÃâ÷ Economical |
| ïÃâ÷ Microbes are easy to manipulate to obtain enzymes with desired characteristics. |
| ïÃâ÷ Easy to extract the enzyme and purify. |
| Bacterial sources- Bacterial sources proved to be an abundant source of L-asparaginase as they are easy to manipulate. These include E.coli, Erwinia cartovora, Pseudomonas flouroscens AG [18], Mycobacterium phlei [19], Staphylococci [20], Tetrahymena pyriformis [21], Thermus aquaticus [22], a marine luminous bacteria [23] and Thermus thermophilus which was reported as not hydrolyzing L-glutamine [24] and a new Erwinia sp. [25] |
| Yeast sources- Yeast sources include Rhodotorula sp., red imperfect yeast, Rhodosporoidum toruloides which produces a homodimer enzyme has been reported by Ramakrishnan [26]. |
| Actinomycetes sources- L-Asparaginase production from Nocardia sp. [27] and Streptomyces sp. has been reported. |
| Plant sources-Among plants L-Asparaginase has been reported to be produced from the Bryophyta Sphagnum fallax [28], Lupin araboreus & Lupin angustiplius [29] and Lupinus leuteus [30]. L-Asparaginase activity has also been reportedly found in soil of the roots of Pinus pinaster and Pinus radiata due to ectomycorhizal fungi in the wheat belt of Western Australia [31]. A K+-dependent L-asparaginase from Arabidopsis, At3g16150 has been characterized [32]. |
 |
 |
 |
V. PRODUCTION OF L-ASPARAGINASE FROM VARIOUS FERMENTATION METHODS |
| L-Asparaginase have been reported from various microbial sources. Production of L-asparaginase II from E.coli has been reported by Cedar and others [81]. Higher yield of L-asparaginase II was obtained when bacteria were grown under anaerobic condition in a shake flask culture with high concentrations of various amino acids but show poor yield in presence of glucose. Bilimoria [82] reported about the culture conditions for L-asparaginase2 production by Coliform bacteria. Of 28 coliforms, 5 strains of E.coli produced L-asparaginase2. It was found that gentle aeration proved suitable for good growth as well as high enzyme content while vigourous aeration gave good growth but low enzyme content of the cells and the stationary cultures gave poor growth. L-asparaginase production in Enterobacter aerogenes expressing Vitreoscilla hemoglobin for efficient oxygen uptake has been described by Geckil and Gencer [83] which showed lower L-asparaginase activity than wild type. As aeration and agitation were the determining factor for the enzyme production, low aeration and agitation showed the higher yield while fully aerobic and anaerobic conditions reduce the enzyme yield. Also the effect of different concentration of glucose showed catabolic repression on enzyme production. Glucose at 1% completely inhibit the enzyme activity while at 0.1 %, it showed slightly stimulatory effect on enzyme production. Kumar and others [84] reported the localization and production of novel Lasparaginase from Pectobacterium carotovorum MTCC1428. The cell fractionation technique showed the enzyme to be localized in the cytoplasm. The enzyme activity was found to be 85 and 77% grown on a medium containing Lasparagine and combination of L-asparagine and glucose respectively which were also found to be most suitable carbon sources to maximize L-asparaginase production. The maximum enzyme production was observed to be 14.56IU/ml at 4 and 2g/l of L-asparagine and glucose respectively. Enhanced production of novel glutaminase free L-asparaginase from Pectobacterium carotovorum MTCC 1428 as reported by Kumar and others [85] by the optimization of the physical process parameters (pH, temperature, rpm of the shaking incubator and inoculums size) using central composite design technique which enhanced the enzyme production and productivity by 26.39% and 10.19% respectively. Maximum production was achieved at 12h under optimum level of physical process parameters in shake flask level. Moharam and others [86] described the production of intracellular L-asparaginase from Bacillus sp. R36 and its immobilization. Optimum culture condition were found to be 9:1 air:medium ratio, 55x105 CFU/ml inoculum size, 24 hr incubation period with shaking growth conditions and an initial pH value of 5.6.Addition of 1% lactose or 1% raffinose doubled the enzyme productivity. Immobilization is done by covalent binding with the activated carbon which greatly enhance the thermal stability of the enzyme. Ebrahiminezhad and others [87] reported L-asparaginase production by moderate halophilic bacteria isolated from Maharloo Salt lake. The halophilic bacteria was screened for L-asparaginase producing ability and found to produce intra and extracellular L-asparaginase. Bacillus sp BCCS 034 was found to produce the highest L-asparaginase (1.64 IU/ml supernatant) extracellularly. |
| An intracellular L-asparaginase have been reported from a strain of Citrobacter [40]. The enzyme produced in a fermentor on scales upto 2700 1 with highest yield in corn-steep liquor medium (9.2% w/v) at 37°C. L-Asparaginase produced from Serratia marcescens by submerged fermentation as reported by Heinemann and others [88] produced highest yield with autolyzed yeast extract medium, an incubation temperature of 26°C and limited aeration with zero level of dissolved oxygen during the fermentation. Highest enzyme yields were obtained when the pH of the fermentation cycles rose to 8.5. Yields of 4 IU of asparaginase/ml of cell suspension have been obtained consistently in 40 to 42 hr from 10-liter volumes (500 ml/4-liter bottle) produced on a reciprocating shaker. Scale-up to a 60-liter fermenter yielded 3.1 IU/ml in 35 hr. Peterson and others [89] reported L-asparaginase production by Erwinia aroideae which gave maximum yield of 1250 IU/g. Liuand and others [90] reported maximum enzyme activity from Erwinia aroideae was obtained with 1% lactose as carbon source and 1.5 % yeast extract as nitrogen source, while glucose showed the inhibitory effect. Both batch and continuous fermentation were employed for large scale production of enzyme. Batch fermentation gave a higher yield (4IU/ml) as compared to continuous fermentation except at dilution rate of 0.1hr -1. The optimum temperature was 24°C for both. L-Asparaginase production by isolated Staphylococcus sp.-6A has been reported by Prakasham and others [91]. Evaluation of various fermentation process parameters were done to optimize nutritional (carbon and nitrogen sources), physiological (incubation temperature, medium pH, aeration and agitation) and microbial (inoculum level) fermentation factors. They reported that incubation temperature, inoculun concentration, pH and nutritional sources (glucose and ammonium chloride) had major impact at individual level and contributed to more than 60% of L-asparaginase production, while aeration, agitation and incubation time showed influence at interactive level. Murthy and others [92] reported production of extracellular L-asparaginase from a soil isolate of Bacillus sp. by the method of submerged fermentation using two different carbon sources, glucose and maltose, of which glucose was found to be the better carbon source. Sarquis and others [93] reported the production of L-asparaginase by Filamentous fungi under nitrogen regulation. The two fungi Aspergillus tamarii and Aspergillus terreus were cultivated in a medium containing different nitrogen sources. Aspergillus terreus showed the highest Lasparaginase production in 2% proline medium, while lowest enzyme production levels in both fungi were found in glutamine and urea as nitrogen source. Narayana and others [94] reported L-asparaginase production by Streptomyces albidoflavus which were optimized under submerged fermentations. Enhanced level of L-asparaginase was obtained with maltose and yeast extract as carbon and nitrogen source. The optimum pH and temperature were found to be 7.5 and 35°C. Amena and others [65] reported the production of extracellular L-asparaginase by Streptomyces gulbargensis using groundnut cake extract medium with maltose (0.5%) and L-asparagine as the best carbon and nitrogen source respectively. The optimum conditions for enzyme production was found to be as follows- pH 8.5, temperature 40°C, inoculum size 1x108 spores/ml and agitation speed 200 rev/min. After purification the optimum pH and temperature for enzyme production were 9 and 40°C respectively. The enzyme was more stable at alkaline pH than acidic one and retained 55% of activity at 80°C for 60 min. L-asparaginase production by strain improvement (i.e. mutation) and whole-cell immobilization of Streptomyces gulbargensis was described by Amena and others [95]. The strain Streptomyces gulbargensis was subjected to mutagenesis in order to isolate strains capable of producing higher yields of L-asparaginase. S.gulbargensis mu24 was isolated and found to be most potent and showed the maximum Lasparaginase activity (44.7IU) at 120 h of fermentation using ground cake extract medium. Thus mutation enhanced the enzyme activity by 1.49 fold. Also the whole-cell immobilization of the mutant and the prototype strain in gelatin enhanced enzyme production by 1.10 fold with fermentation carried out at 40°C at 200 rev/min. Dharmaraj [96] studied L-asparaginase production by Streptomyces noursei MTCC 10469 isolated from marine sponge Callyspongia diffusa. This marine actinomycete was isolated using specific ISP medium. Production was carried out by submerged fermentation using Tryptone Glucose Yeast extract (TGY) broth. The enzyme was purified 98.23 fold and showed a final specific activity of 78.88 IU/mg with 2.14% yield. The optimum pH, temperature and incubation time was found to be 8, 50°C and 35 min respectively. |
| Abdel-Fateh has reported L-asparaginase production by Pseudomonas aeruginosa 50071 in solid state culture. Culture conditions were optimized using Plackett-Burman factorial design. pH, casein hydrolysate and corn-steep liquor were found to the most significant factors for improving enzyme production and their optimal values as determined by Box- Behnken design were found to be pH 7.9, casein hydrolysate, 3.11% and corn-steep liquor, 3.6%. After optimization enzyme activity (142.81IU) was enhanced by more than five-fold than that of the basal medium. Asparaginase production from Pseudomonas aeruginosa 50071 by solid-state fermentation has also been described by Ashraf and others [97]. They purified (106 fold) the enzyme to give a specific activity of 1900IU/mg with a 43% yield and with its maximum activity at pH 9 when incubated at 37âÃÂðC for 30 min. Maximum L-asparaginase production was obtained from Bacillus circulans by solid state fermentation using red gram husk (an agricultural waste) [35]. Optimization of a wide range of SSF parameters were done using fractional factorial central composite design (FFCCD).Venil and others [98] reported L-asparaginase production by Serratia marcescens SB08 in rice bran as it is cheap and easily available source for enzyme production in solid state cultivation. A maximum production (79.84 IU/g) was achieved with 50% initial moisture content in rice bran and supplemented with yeast extract (0.5% w/v) and at 36h incubation time but rice bran combined with other starchy material considerably reduced the enzyme yield. Optimization of L-asparaginase production by Serratia marcescens SB08 reported by Venil and others [99] was done by a combined method of Plackett-Burman design and response surface method in which 4 factors - Sucrose, peptone, KH2PO4 and incubation time were screened as best medium factors and their optimal values as determined by central composite design were found to be 12.50g/l, 4.5g/l, 4.0g/l and 51h respectively. Siddalingeshwara and others [100] reported the production of L-asparaginase from Aspergillus terreus KLS2 by SSF using carob pod as a substrate and showed 6.05IU enzyme activity after optimizing the fermentation parameters. Hosamani and others [101] reported L-asparaginase production by Fusarium equiseti using soya bean meal under solid state fermentation. Optimized fermentation parameters for maximum enzyme yield (8.51IU) were as follows – incubation period (48 hrs), initial moisture content (70% v/w), particle size (3mm), inoculum volume (20%), supplemented with glucose (0.5% w/v), ammonium sulphate (0.5 % w/v) and yeast extract (0.5 % w/v). |
| An extracellular L-asparaginase enzyme from marine Actionmycetes has been reported by Basha and others [102]. The enzyme production was done by solid-state and submerged fermentation. The enzyme production was carried out in three different media, namely solid-state media, Tryptone Glucose Yeast extract (TGY) broth and Tryptone Fructose Yeast extract (TFY) broth. The isolates S3, S4 and K8 showed the potential for L-asparaginase activity and produced the enzyme with yields ranging from 24.6 to 49.2IU/ml. S3 showed productivity of 49.2 IU/ml with a protein content of 65 μg/ml and optimum activity at pH 7.5 and temperature at 50âÃÂðC. Rani and others [103] described isolation and screening of L-asparaginase producing fungi from soil sample using Czapek’s agar media. Aspergillus sp (KUFS20) was isolated on the basis of pink colour zone formation. Enzyme production was done by both submerged and solid state fermentation using 20 different substrates of which orange peel showed maximum enzyme production. |
VI. CLONING OF L-ASPARAGINASE |
| L-Asparaginase is an important therapeutic enzyme used in the treatment of leukemia. The clinical trials indicated that the two bacterium E.coli and Erwinia sp. as the potential source of the enzyme L-asparaginase and has been used as an antitumor drug. But various side effects arises as a consequence of their intrinsic glutaminase activity, which leads to lglutamine depletion in the blood and also the cost of this antitumour drug is quite high. Consequently, new asparaginases with low glutaminase activity, fewer side effects and high activity towards l-asparagine with reduced treatment cost are highly desirable as better alternatives in cancer therapy. Thus there is need to implement the recombinant techniques to enhance the properties of enzyme and reduce the cost of the final product. The recombinant enzyme is produced by the molecular cloning techniques and genetic engineering methods. |
| Khushoo and others [104] reported the extracellular expression of L-Asparaginase II from E.coli in the recombinant form by fusing the gene coding for asparaginase to an efficient pelB leader sequence and an N-terminal 6x histidine tag cloned under the T7lac promoter. The recombinant protein after purification gave an overall yield of 95 mg/L of purified protein, with a recovery of 86% which is approximately 8-fold higher to the previously reported data in literature. The purified protein has the similar fluorescence spectra, analytical size and the specific activity as the native protein which signified that the protein had folded properly and was present in its active tetramer form. |
| Kotzia and others [105] reported the cloning and expression of L-asparaginase from E.carotovora via performing PCR of its gene sequence, inserting into suitable expression vector and transforming the expression host E.coli BL21 (DE3) pLysS. The resulting recombinant enzyme showed the high asparaginase activity with the specific activity of 0.72U/mg protein. In 2006 they also reported the cloning and expression of L-asparaginase from Erwinia chrysanthemi 3937 (ErL-ASNase) in Escherichia coli BL21 (DE3) pLysS. The enzyme after purification and characterization was efficiently immobilized on epoxy-activated Sepharose CL-6B which retains most of its activity (60%) and shows high stability at 4 degrees C. The approach offers the possibility of designing an ErL-ASNase bioreactor that can be operated over a long period of time with high efficiency, which can be used in leukaemia therapy. |
| In order to reduce the antigenicity of Escherichia coli L-asparaginase (L-ASP) a strategy known as alanine-scanning mutagenesis was applied which identify the amino acid residues responsible for the antigenicity. Three continuous alkaline residues, 195RKH197, were mutated to Ala selectively. Four mutant recombinant L-ASPs were constructed and expressed in E.coli, and then purified. By the competition ELISA method it was found that the antigenicity of the enzyme was greatly reduced [106]. |
| The recombinant asparaginase of Escherichia coli, consisting of asparaginase, a tetanus toxin peptide (TTP) spacer (831-854 fragment), and the foreign cholesteryl ester transfer protein C-terminal fragment (CETPC) was expressed and targeted to the periplasm of E. coli. After purification it exhibited approximately 83% activity of the native enzyme, allowing the rapid screening of recombinant clones while an asparaginase-CETPC fusion protein without the TTP spacer produced only about 23% activity of the native enzyme. This chimeric protein when inoculated in rat cells, generated CETP-specific immunoresponse. In contrast, rats immunized with the cells expressing asparaginase only did not induce specific anti-CETP antibodies. This showed that asparaginase of E. coli was an effective carrier for displaying foreign peptides or epitopes and the TTP spacer appeared to play a critical role in directing the foreign CETPC peptide to the surface of the enzyme. Thus the chimeric enzyme constructs fusing asparaginase with foreign peptides via a TTP spacer could be utilized as a rapid pepscan technique for antigen epitope mapping [107]. |
| L-Asparaginase from Helicobacter pylori was cloned and overexpressed in E. coli and purified [108]. |
| Cappalletti and others [109] cloned and expressed recombinant L-asparaginase from the pathogenic strain Helicobacter pylori CCUG 17874 which showed a low glutaminase activity and exhibited a sigmoidal behavior towards Lglutamine. |
| Krastokina and others [110] cloned and expressed ECAR-LANS, the recombinant L-asparaginase from Erwinia carotovora in Escherichia coli which was found to have a relatively low glutaminase activity. |
| L-asparaginase from Pyrococcus furiosus was cloned and expressed in Escherichia coli. Kinetic studies revealed that the enzyme was thermostable, natively dimeric, and glutaminase-free [111]. |
VII. PURIFICATION OF L-ASPARAGINASE |
| L-Asparaginases from different sources have been purified by various methods. Wei and others [112] have constructed various mutant enzymes from Escherichia coli in order to reduce the antigenicity and to effectively purify these enzyme mutants, developed a native preparative polyacrylamide gel electrophoresis to further the perform immunological and biological studies which gave 400U/mg activity. This new method was efficient and cost-effective compared with other methods, such as column chromatography and affinity chromatography [112] |
| Purification of L-Asparaginase from Acinetobacter calcoaceticus was done by precipitation with streptomycin, chromatography on DEAE-cellulose and CM-cellulose, gel filtration on agarose and chromatography on phosphocellulose. The enzyme catalyzed the deamination of L-glutamine to about the same extent as L-asparagine and showed a weak tumor inhibitory power [33]. |
| Bacterial L-asparaginases (L-ASNases) catalyze the conversion of L-asparagine to L-aspartate and ammonia. Kotzia and others [105] reported the cloning and expression of L-asparaginase from Erwinia chrysanthemi 3937 (ErLASNase) in Escherichia coli BL21(DE3) and purification of the enzyme was done by a single-step procedure involving cation exchange chromatography on an S-Sepharose FF column which showed comparatively high activity. |
| To test the asparaginase of Escherichia coli as a vector to display linear peptides on the surface of each enzyme subunit, a chimeric protein was constructed and targeted to the periplasm of E. coli. Both native wild type enzyme and recombinant mutant chimeric enzyme was purified from periplasm preparation through osmotic shock and anion exchange chromatography. The purified chimeric enzyme exhibited approximately 83% activity of the native enzyme [107]. |
| L-Asparaginase (isozyme II) from Escherichia coli was cloned and expressed extracellularly. The resulting recombinant protein was purified by a single step using Ni-NTA affinity chromatography which gave an overall yield of 95 mg/L of purified protein, with a recovery of 86%. This is approximately 8-fold higher to the previously reported data in literature [104]. |
| Gladilina and others [113] reported the cloning and expression of recombinant protein from Helicobacter pylori J99 in E.coli (BL21) which was first purified upta 1.8 fold, then sonicated for the preparation of cell free extract and the enzyme was purified from the soluble fraction of cell free extract by chromatography on SP-Sepharose which gave more than 60% yield. |
| Statistically based experimental design to assess the physical process parameter was applied to maximize the production of glutaminase-free L-asparaginase from Pectobacterium carotovorum MTCC 1428 which after purification via a three step process enhanced the production and productivity of L-asparaginase by 26.39% (specific activity) and 10.19%, respectively [85]. |
| Purification of the L-asparaginase enzyme to homogeneity from Pseudomonas aeruginosa 50071 cells that were grown on solid-state fermentation was done by applying different purification steps including ammonium sulfate fractionation followed by separation on Sephadex G-100 gel filtration and CM-Sephadex C50crude culture filtrate. The enzyme was purified 106-fold and showed a final specific activity of 1900 IU/mg with a 43% yield [114]. |
| The recombinant L-asparaginase enzyme of Pyrococcus koshii was expressed in E.coli (BL21) and purified by anion exchange chromatography and gel filtration followed by hydrophobic interaction chromatography and ultrafiltration [55]. L-asparaginase of Streptomyces tendae isolated from laterite soil samples of Guntur region and the crude enzyme was purified to homogeneity by ammonium sulfate precipitation, Sephadex G-100 and CM-Sephadex G-50 gel filtration [68]. |
VIII.STRUCTURAL PARAMETERS |
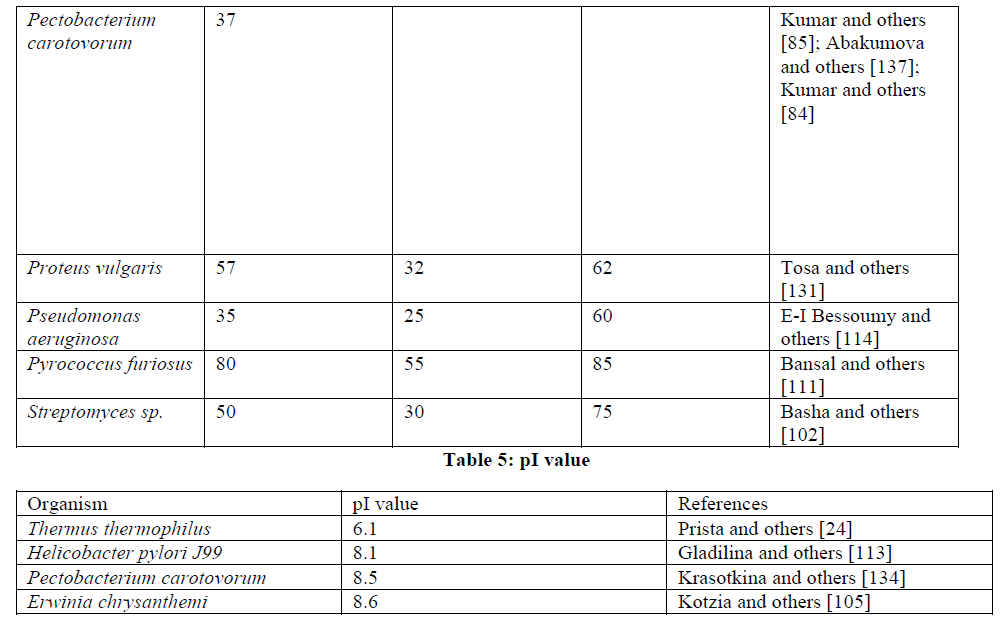
| Enzyme structure: L-Asparaginases are mostly found in tetrameric form but when isolated from various sources also exist in dimer, hexamer or in monomer form. The recombinant enzyme from Escherichia coli was found in active tetramer form with each subunit having the molecular weight of 37kD as revealed by SDS-PAGE while gel filtration of the purified enzyme showed its molecular weight to be 150kD [104]. Zhang and others [115] reported Escherichia coli L-asparaginase exists in homotetrameric form with one active centre having a molecular weight of 138kD. On performing the SDS-PAGE of L-Asparaginase derived from Helicobacter pylori, it was found that the enzyme exist in homotetramer form with 37kD molecular weight for each subunit while gel filtration revealed the molecular weight to be 140kD [109]. The molecular weight of L-asparaginase isolated from Pseudomonas aeruginosa was found to be 160kD and it exists in monomeric form as determined by gel filtration SDS-PAGE [114]. The molecular weight and its no. of subunits enzyme from Pectobacterium carotovorum were determined by native PAGE, gel filtration and MALDI-TOF mass spectrometry and it was found to be homotetramer with 144kD molecular weight [85]. The enzyme from Pyrococcus furiosus was found in dimer form as determined by SDS-PAGE and mass spectrometry and its molecular weight was found to be 75.45kD as determined by gel filtration and Superdex column [111]. Thermus thermolphilus enzyme existed in homohexameric form with molecular weight 33kD for each subunit as revealed by SDS-PAGE and has a molecular weight of 200kD as determined by gel filtration [24]. Verma and others [19] reported that the L-asparaginase of Erwinia sp. have hexameric structure. Liu and others [116] reported that the enzyme from Erwinia aroidea have a molecular weight of 108kD. |
 |
| Crystallization of the enzyme had also been done by many scientists. Elucidations of crystal structure provide insight into the active site of the enzyme and a better understanding of the structure-activity relationship in L-asparaginase. Aghaiypour and others [117] reported the high resolution crystal structure of complexes of L-asparaginase with Lglutamic acid, D-aspartic acid and succinic acid. Comparison of the four independent active sites within each complex indicates unique and specific binding of the ligand molecules; the mode of binding is also similar between complexes. The lack of the alpha-NH3(+) group in Suc, compared to L-Asp, does not affect the binding mode. The side chain of LGlu, larger than that of L-Asp, causes several structural distortions in the ErA active side. A comparison of the amino acid sequence and the crystal structure of ErA with those of other bacterial L-asparaginases shows that the presence of two active-site residues, Glu63 (ErA) and Ser254 (ErA), may correlate with significant glutaminase activity, while their substitution by Gln and Asn, respectively, may lead to minimal L-glutaminase activity. Sanches and others [118] reported the crystallization of E.coli L-asparaginase by hanging drop vapour diffusion method. The X-ray structure of the enzyme, crystallized in a new form (space group C2) and refined to 1.95 A resolution is compared with that of previously determined crystal form (space group P2(1)). L-asparaginase dimer was found as he asymmetric unit of the new crystal form instead of the tetramer found in the previous crystal form. Major conformational differences are confined to the lid loop (residues 14-27). Dhavala and others [108] reported the crystal structure of Helicobacter pylori L-aparaginase in the presence of L-aspartate which was determined to 1.4 A resolution. Major differences were found in the active-site flexible loop and in the 286-297 loop from the second subunit, which is involved in active-site formation. Accordingly, Glu289, Asn255 and Gln63 appeared to play roles in modulating the accessibility of the active site. |
Physiochemical parameters: |
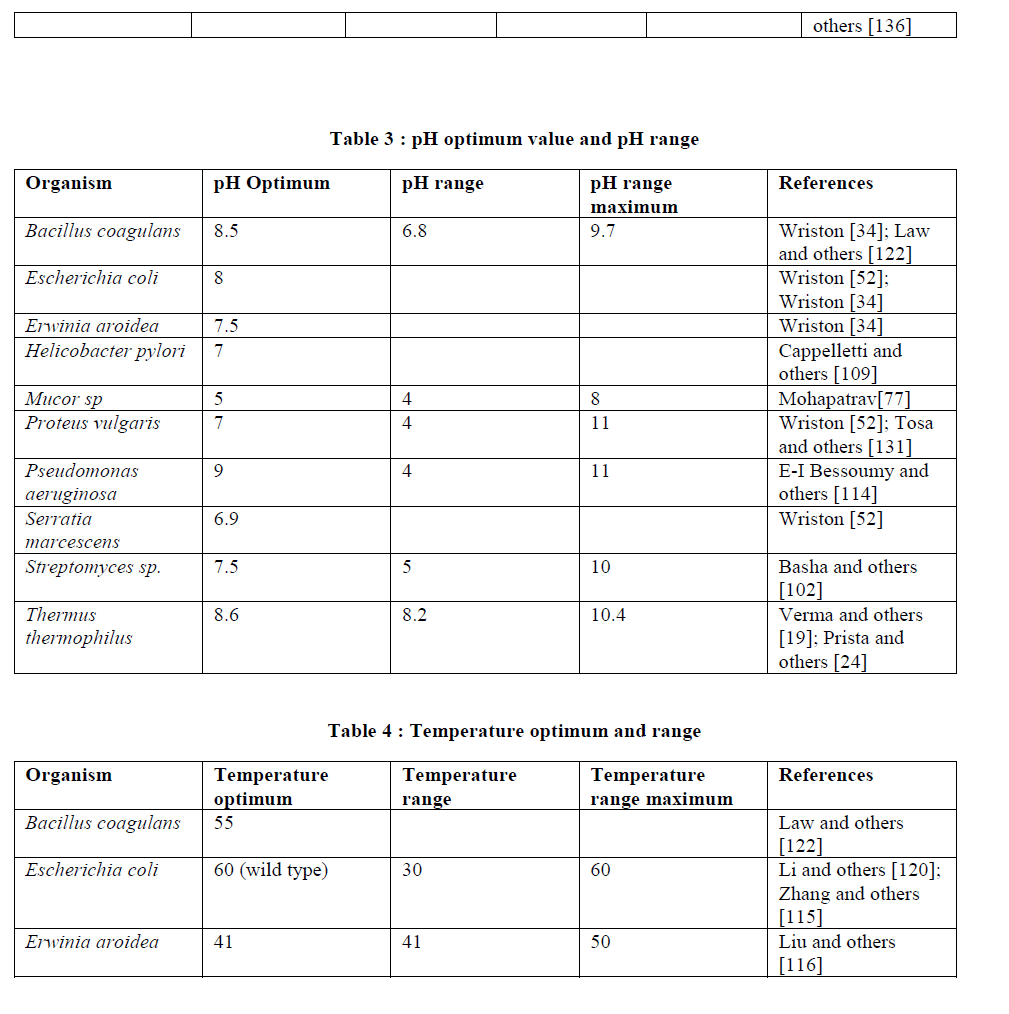
| pH stability: L-Asparaginase from different sources shows different pH stability. Some are stable at acidic pH, some at neutral while some have stability at alkaline pH. The extracellular asparaginase from Cyberlindnera jadinii were quite stable at pH 4 for about 10 min at 50âÃÂðC and upto 20h at 37âÃÂðC. However it remain stable upto a maximum of pH 10 [119]. The purified enzyme from Pseudomonas stutzeri MB 405 having the optimum activity at pH 9 and 37âÃÂðC showed maximum stability in the range of pH 7.5 to 9.5 [54]. The enzyme derived from Helicobaterium pylori J99 remain stable near pH 10 but rapidly loses its activity irreversibly at pH 10 or above [113]. Amena and others [65] reported the enzyme from Streptomyces gulbargensis to be more stable at alkaline pH and retained its 80% of activity in the pH range of 7-10. |
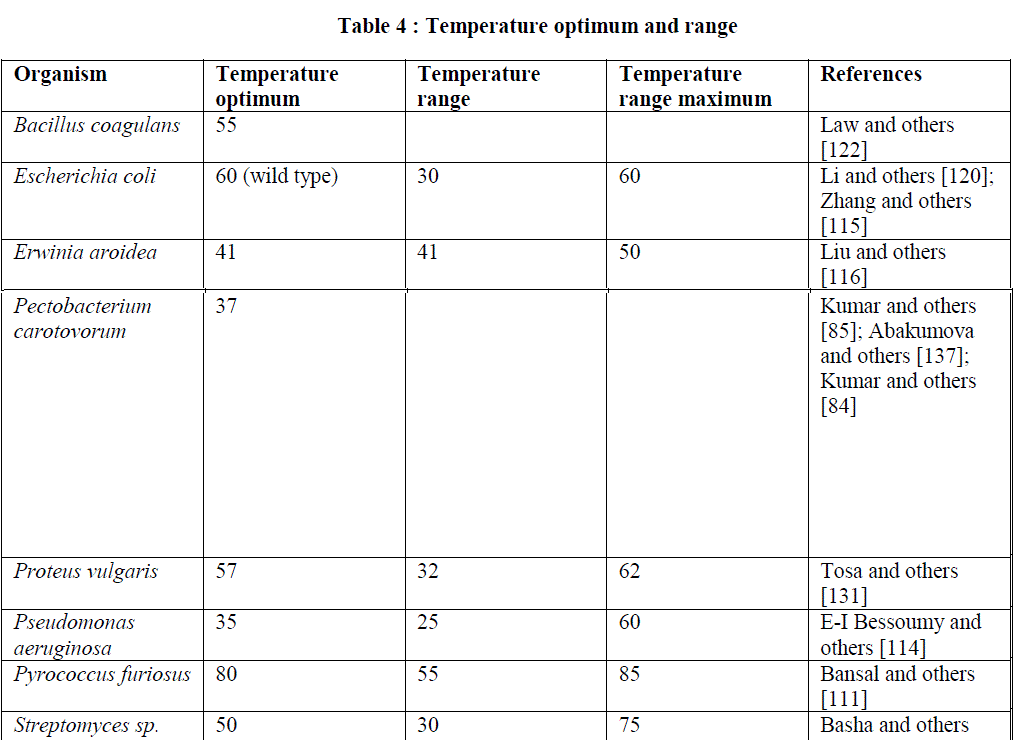
| Temperature stability: The L-asparaginase enzymes from various sources have different temperature stabilities ranging from 37âÃÂðC to upto 80âÃÂðC due to differences in their amino acid sequences and structures. Both the wild type and mutant L-asparaginase enzyme D178P from Escherichia coli remain stable at 37âÃÂðC upto 4 h but after that the wild type enzyme rapidly loses its residual activity as compared to mutant one. After 6 h incubation, wild type shows 35% while mutant enzyme shows 19 % loss of activity. With incubation time of 1h, 97% of the mutant and 79% of the wild type enzyme activity remained at 45âÃÂðC while upto 50âÃÂðC, 90% of mutant and 71 % of the wild type enzyme activity remained [120]. Zhang and others [115] reported that the immobilized L-asparaginase enzyme showed significantly higher stability upto 40-50âÃÂðC while loses 70 % of activity at 60âÃÂðC for 30 min and become completely inactivated when incubated at 80âÃÂðC for 30 min. The enzyme from Cylindrocarpon obtusisporum remains stable at 37âÃÂðC for 30 min while loses 20% of its activity at 40âÃÂðC [76]. The enzyme from Pseudomonas stutzeri remain stable at 40âÃÂðC for 1h with no significant loss of activity while have a half-life of 25 min and 10 min at 60âÃÂðC and 65âÃÂðC respectively [54]. Kotzia and others [50] reported that the Tm of wild type enzyme of Erwinia chrysanthemi and its thermostable variant containing a single point mutation (Asp133Val) were 46.4âÃÂðC and 55.8âÃÂðC and the half-life of both wild type and mutant enzyme were found to be 2.7 h and 159.7 h respectively. The enzyme isolated from Helicobacter pylori remained absolutely stable upto a temperature of 50âÃÂðC for 10 min. But at 53âÃÂðC, after 10 min loses 50% of its activity [109]. L-Asparaginase obtained from Azatobacter vinelandii remained stable up to a temperature of 50âÃÂðC but get inactivated at 65âÃÂðC [121]. The enzyme of Streptomyces gulbargensis retained its 55% of activity at 80âÃÂðC for 1h at alkaline pH [65]. |
| Metals and ions: Metals and ions play a great role in the regulation of enzyme activities. L-asparaginase isolated from various organisms respond differently to different metals and ions. L-Asparaginase obtained from the bacteria Bacillus coagulans showed a rapid and reversible activation in presence of metal ions such as K+, Li+, Na+ [122]. In case of Pseudomonas stutzeri, K+, Mg2+ and Na+ causes activation of L-asparaginase [54]. When 1mM, 3mM and 10mM of enzyme obtained from Streptomyces sp. were tested, the enzyme activity was inhibited in presence of Cu2+, EDTA, Zn2+ while activated in presence of Mn2+ [102]. In case of Mucor sp., the enzyme showed the maximum activity in presence of Na+ at 2% NaCl [77]. The L-asparaginase obtained from Tetrahymena thermophila showed 40-45% of activation in presence of Ca2+ in the concentration of 5mM while Mg2+ causes 5-10% of activation [123]. The enzyme obtained from Acinetobacter calcoaceticus get stimulated in presence of Mg2+ [124]. The ions such as CN-, I- and PO4 3 enhances the activity of L-asparaginase from the fungi Cylindrocarpon obtusisporum [76]. |
| Inhibitors: There are several compounds which inhibits the L-asparaginase activity. One of the inhibitors of Lasparaginase obtained from Escherichia coli is 5-Diazo-4-oxo-L-norvaline [125]. Other inhibitors of L-asparaginase include Dimethylsulfoxide, Glutaraldehyde which inhibits the enzyme during immobilization at concentration above 0.2%, L-Asp which is indirectly responsible for the enzyme inhibition by preventing the hydrolysis of diazo-4-oxo-Lnorvaline, NH4 + which causes the product inhibition at pH 8.5n and trypsin which completely inactivate the free enzyme within 10 min. while the immobilized enzyme remain stable for 240 min. with loss of 30% activity. In case of Helicobacter pylori, 5-Diazo-4-oxo-L-norvaline inhibits L-asparaginase activity and affects broth culture filtrate inhibition of the cell cycle [126]. In case of Pseudomonas stutzeri, 5-Diazo-4-oxo-L-norvaline, Ca2+, Cd2+, Cu2+, Fe3+, Hg2+, iodoacetamide, Mg2+, Mn2+, Ni2+, PCMB, SDS, urea, Zn2+ were the inhibitors of the enzyme L-asparaginase [54]. |
| In Mucor sp. EDTA happens to be a strong inhibitors of L-asparaginase while Ca2+ causes 26% loss of activity in 25mM concentration, Cu2+ causes 44% inhibition in 20mM concentration, Zn2+ in 25mM concentration causes 25% inhibition [77]. Some of the inhibitors of L-asparaginase obtained from Azatobacter vinelandii were atabrin, Cu2+, Fe2+, Hg2+, hydrazine, hydroxylamine, iodoacetate, KCN, p-hydroxymercuribenzoate, potassium ferricyanide and Zn2+ [121]. For the L-asparaginase from Tetrahymena pyriformis, D-Asn acts as competitive inhibitor which reverses the antiproliferating effect on breast cancer cells [127]. In case of Thermus thermophilus, D-Asn and L-Asp act as competitive inhibitor of the enzyme while diethylbicarbonate, Mg2+, Mn2+, Ca2+ and p-hydroxymercuribenzoate are some of the other inhibitors of L-asparaginase. PMSF causes the partial inactivation of the enzyme and there is 40% loss of enzyme activity in presence of Zn2+ in 1.0mM concentration [24]. L-Asp and PCMB are the inhibitors the enzyme from Bacillus coagulans [122]. In case of Pyrococcus furiosus, L-asparagine in above 20mM concentration causes the substrate inhibition of the enzyme [111]. |
| Activating compounds: Various compounds are responsible for the enhancement in the activity of L-asparaginase. In case of Pseudomonas stutzeri, the compounds such as 2-mercaptoethanol, dithiothreitol, EDTA, glutathione, GSH, LCys, L-His enhances the enzyme activity [54] while 1-amino-6-methyl-4-phenyl-2-thioxy-1, 2, 3, 4- tetrahydropyrimidine-5-carboxylic acid methyl ester enhances the activity of enzyme obtained from Pectobacterium carotovorum [128]. In case of Tetrahymena thermophila, albumin in quantity as little as 0.1 mg/ml does the 3-fold activation of L-asparaginase II while sorbitol in concentration of 30% w/v causes more than10-fold activation [123]. The enzyme obtained from Acinetobacter calcoaceticus get stimulated in presence of citrate [33] while in case of Brevibacillus brevis, n-dodecane and silicone oil exhibit a stimulatory effect in L-asparaginase activity; 104% for the Ist and 108% for the IInd compared to control while liquid paraffin in concentration of 6% (w/v) results in 34% increase in L-asparaginase activity accompanied by a 48% increase in the production of cell mass at a 10L scale [2]. |
| Storage stability: Storage of the enzyme L-asparaginase is one of the major concerns. Scientists have tried various method and temperatures to store the L-asparaginase from different sources. The enzyme from different microbial sources shows variable storage stabilities. Joner and others [124] reported L-asparaginase B from Acenetobacter calcoaceticus can remain stable at -20ºC with little loss of activity even after prolonged storage. Law and others [122] reported that the Bacillus coagulans L-asparaginase can remain stable in frozen state for 6 months or longer. Robert and others [129] reported that E.coli L-asparaginase in lyphilyzed form can be stored at 5ºC and remain stable for at least several weeks while as a sterile enzyme solution with high specific activity can also be stored at 5ºC with no loss of activity. E.coli L-asparaginase enzyme can also be stored as 1000U/ml in plasma solution at -40ºC for 1 year with no decline in activity [130]. L-asparginase from Proteus vulgaris as reported by Tosa and others [131] can remain stable for 1 month when stored at -20ºC while when stored at 5ºC as suspension in 3.5M ammonium sulphate can remain stable for 1 year. Ferrara and others [132] reported that the crude asparaginase preparation from Saccharomyces cerevisiae is stable upon storage at -18ºC for several months. |
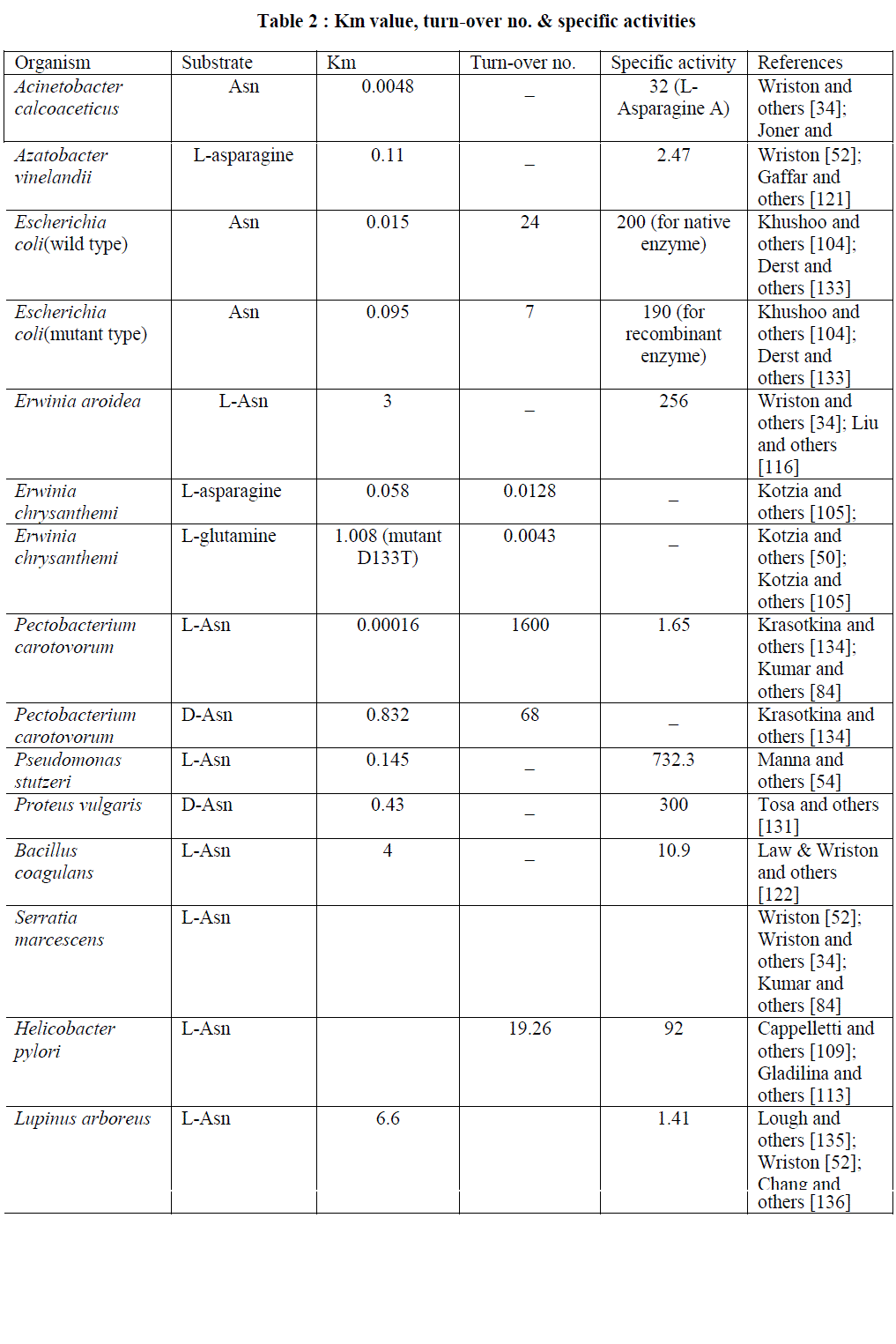
| Kinetic parameters: The steady state kinetics measurement of the enzyme L-asparaginase was perfermed by varying the concentration of substrate. The non-linear regression analysis of the steady state data were usually employed to calculate various kinetic constants such as Km, Vmax, Kcat etc. Turn over no.were calculated on the basis of one active site per subunit. A no. of computer programs were available for analysing the kinetic data (such as GraFit, Erithacus Software Ltd., Leatherbarrow, 1998; OriginLab version 8.5, OriginLab Corporation Northampton, MA, USA). LAsparaginase from different sources showed differences in their kinetic properties and hence in the values of Km, turn over no. etc. Some of the kinetic parameters of L-asparaginase from different sources are as follows: |
 |
 |
 |
 |
VIII. FORMULATION AND IMMOBILISATION |
| Some of the major limitations in the use of the enzyme are its severe immunological reactions and a very short serum half-life. Modifications like formulation and immobilization of the enzyme onto a suitable matrix can greately reduce the immunogenicity of the enzyme; increase its half-life and its therapeutic potential. |
| Gasper and others [138] reported the formulation of poly (lactide-co-glycolide) (PLG) nanoparticles containing the enzyme L-asparaginase. They encapsulated the enzyme in PLG nanoparticles using a water-in-oil-in-water solvent evaporation technique which resulted in the delivery of L-asparaginase for an extended period of time. |
| The immobilization of E.coli L-asparaginase into a hydrogel matrix made of poly (ethylene glycol) PEG and BSA showed a 200 fold increase in its Km value and a wider pH range for the optimal activity of the enzyme with 90% of activity at physiological pH of 7.3 as compared to 43% activity for the native form. Also the half life of the immobilized enzyme enhanced to 50 days with 90% activity at 37ºC as compared with the half-life of 2 days for the native enzyme [139]. In an another experiment, they (1997) coupled E.coli asparaginase in a biocompatible hydrogel made of rat serum albumin and PEG and assessed for its effectiveness to deplete the serum L-asparagine in vivo. It was found that 85-90% of serum asparagines got depleted in 2 days with 5 units/rat and also 80% activity of the enzyme was still retained even after 10 days [140]. |
| Vina and others [141] reported the immobilization of L-asparaginase from Erwinia carotovora on a biologically active fructose polymer levan of different molecular mass (75 & 2000 kDa) obtained from Zymomonas mobilis. They employed the method of periodate oxidation of the polysaccharide followed by reductive alkylation which retained greater than 55% of activity, a higher Km value and a wide optimum pH range of the enzyme. The immobilization of the enzyme also resulted in reduced electrophoretic mobility, higher thermostability and prolonged storage (1 month) in aqueous solution as compared to native enzyme. |
| Another method of immobilization was tried by Zhang and others [115]. They covalently conjugated the enzyme on the microparticles of natural silk sericin protein followed by cross-linking with glutaraldehyde which maintained 62.5% of the original activity of the enzyme. |
| A new strategy based upon multisubunit covalent immobilization of L-asparaginase from E.coli onto the activated supports (agarose-glutaraldehyde) had been employed by Balacao and others [142] followed by solid-phase intersubunit cross-linking with aldehyde-dextran. This resulted in a longer half-life and a nil risk of subunit release into the blood circulation |
| Bacillus sp. L-asparaginase was efficiently immobilized by covalent binding with activated carbon as reported by Moharam and others [86]. This enhanced the enzyme yield (73.6%) and showed the activity of 33U/g carrier.The immobilization process greatly enhanced the thermostability of the enzyme showing 100% activity upto 80ºC as compared to 50ºC for that of the native enzyme [86]. |
IX. CONCLUSION |
| From the past 30 years, a lot of research had been done for the role of L-asparaginase as a potent antitumour drug. It was first found that the guinea-pig serum exhibit an anti-cancer properties. Later on this drug was isolated from E.coli and Erwinia sp. which have been used in the treatment of acute lymphocytic leukemia since then. But the restricting factors in the use of this drug are several side effects due to its antigenicity and a very short half-life of this enzyme in the blood. Its repeated doses can lead to the development of allergic reactions in the patients. Moreover the lower yield of the enzyme is also one of the major concerns. Not only various mode of productions and optimization conditions were adopted to isolate the enzyme from different sources all over the world to enhance its production but also a various kind of modifications of this enzyme had been tried by scientists to reduce its immunogenicity and increase its half-life. The use of recombinant technology and its formulation and immobilization is still in progress. Still there is a long way to go. Moreover application of L-asparaginase in the food industry for the elimination of cancer-causing acrylamide from baked food has been one of the eminent discoveries of mordern time. Thus a lot more is needed to explore about this amazing enzyme. |
References |
|