ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Divya Pal1, Madhulika Singh2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
In this study, tomato (Solanum lycopersicum) and Chilli (Capsicum) F1 hybrids and their parents were studied for identification and genetic purity testing using 22 and 15 random10-base oligonucleotide RAPD primers respectively. The present study analyzed suitability of RAPD markers for characterization of chilli(capsicum) hybrids and their parents. Out of 15 decamer random primers used for analysis, three primes, OPC-08, OPL-12 and OPV-14 yielded scorable results in the form of amplified products. This study also analyzed Solanum lycopersicum F1 hybrids and its parents (male and female) with 22 different decamer primers to identify the band pattern which could differentiate between the parents and hybrids. Out of these 22 RAPD primers only 5 primers, OPA-05, OPA-09, OPN- 14, OPG-04 and OPK-02 showed polymorphic bands between male, female and hybrids. Since a lot of chances are there for contamination by selfing of female parental lines and out crossing with other plants during hybrid development programs. This seriously affects the hybrid production quality. So molecular marker tools can be effectively used to find out such contaminations in DNA polymorphism of respective hybrids
Keywords |
| Solanum lycopersicum, Capsicum, RAPD markers, F1 hybrids, Genetic purity. |
INTRODUCTION |
| Cultivated tomato, Lycopersicum esculentum Miller (Solanaceae), is a very important crop due to high value of its fruits for fresh consumption. The tomato is a herbaceous, usually sprawling plant in the Solanaceae or nightshade family that is typically cultivated for the purpose of harvesting its fruit for human consumption. The taxonomy keys, interior of ripe fruit red, fruit diameter, leaves subdivided, internodes short, densely pubescent, and large accresant calyx are a revision to that formulated by Rick et al. [1] and published in the Tomato Genetics Cooperative. Genotypic differences within tomato cultivars detected by molecular markers can also be used for cultivar identification and protection of the plant breeder’s intellectual property rights (plant breeders’ rights, PBRs). Data reported in all the studies dealing with the application of DNA markers in tomato cultivar identification are useful and provide important background information to address the issue of PBRs [2, 3]. Chili (Capsicum spp.) is one of the most popular and widely grown vegetable in India. Chili rich in vitamins A, C and E is being used as spice, condiment, vegetable, culinary supplement, medicine and as ornamental plant. The fruit of Capsicum [4] plants have a variety of names with reference to place and type. They are commonly known as chilli pepper, capsicum, red or green pepper, or sweet pepper in Britain, and typically just capsicum in Australia and India. Molecular markers are important tool for genotype identification and studying the organization and evaluation of plant genome [5]. With the advent of molecular biology technique, large number of highly useful DNA markers has been developed for the identification of genetic polymorphism. RAPD markers are amplification product of anonymous DNA sequence. |
| The analysis of genetic diversity and relatedness between or within different species, populations and individuals is a prerequisite towards effective utilization and protection of plant genetic resources [6]. Molecular markers have many advantages such as abundance in polymorphism, no pleiotropic effect, less affected by environment and subjected to rapid detection [7].Among the molecular markers, RAPD has been widely used for the identification of genetic relationship between cultivars [8,9,10] for species identification [11]. The advantages of using RAPDs in gentic studies are, it requires small amount of DNA, short primers of arbitrary sequences, the rapidity to screen for polymorphism, easy, fast and also cost effective [11]. |
MATERIALS AND METHOD |
| 10 samples of tomato ( Lycopersicum esculentum) and chilli (Capsicum) were studied in this investigation. Fresh leaf tissues of all the samples were used for DNA extraction.DNA was extracted by cetyltrimethyl ammonium bromide (CTAB) method. A total of 22 primers of Operon Technologies were used for tomato samples and 15 primers of the same were screened in the case of chilli. The list of random primers used in this study is given as follows: |
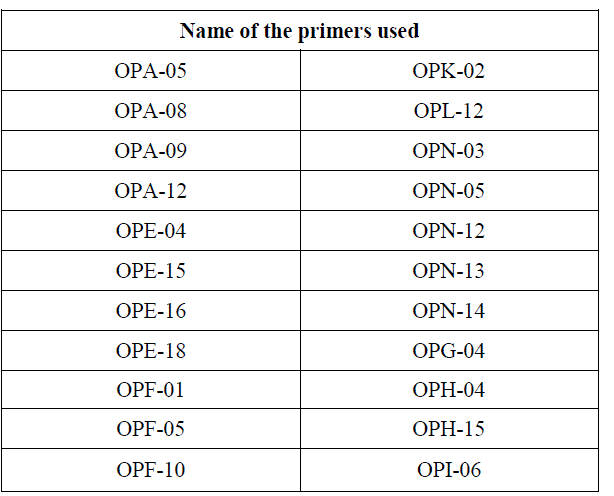 |
| The PCR reactions were performed using a 15μl reaction mixture containing 10X Taq DNA Polymerase buffer with 2mM MgCl2, 2.5 Mm dNTPs, 5μM primers, 4 μl Taq DNA polymerase, 30 to 40 ng template DNA and sterile distilled water. For DNA amplification, the DNA thermal cycler was programmed as follows: Denaturation step was for 5 minutes at 94ºC, Annealing for 45 seconds at 55 ºC and Extension for 1 minute at 72 ºC. The number of cycles repeated was 35. After completion samples were stored at 4 ºC. This PCR amplified products were separated by electrophoresis on 1.5% agarose gel with 1X TBE buffer containing Ethidium Bromide (EtBr) and then visualized in Gel documentation system. |
RESULT AND DISCUSSION |
| Molecular markers have been successfully used in the genus Capsicum to select parents for hybrid production, for intra-specific or inter-specific classification, and for the analysis of variation. However, commercial pepper varieties possess a low level of molecular polymorphism [12]. In our study, 15 primers (Operon technologies) were analysed. Out of these 15 primers, 3 primers (OPC-08, OPL-12, and OPV-14) showed polymorphic bands for the hybrid. In case of tomato (Lycopersicum esculentum), 22 primers were screened and out of these 22 primers only 5 primers (OPA-09, OPA-05, OPN-14, OPG-04 and OPK-02) showed polymorphic bands between male, female and hybrid samples. For tomato (Lycopersicum esculentum), when band pattern of hybrids were compared with their parents, primers OPA- 09, OPA-05, OPN-14, OPG-04 and OPK-02 showed polymorphic bands which can be used to distinguish between hybrid and its parents. These primers can also be used to confirm that the hybrid is of its respective parents. Among all the five primers, OPN-14 showed maximum number of polymorphic bands and thus can be used efficiently for genetic purity testing. |
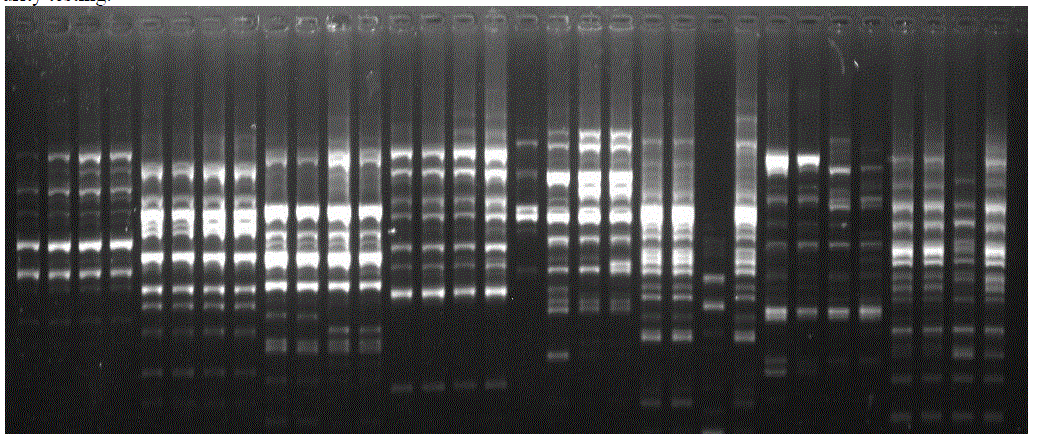 |
| Similarly, 15 primers were screened in the case of Chilli (Capsicum).Out of these 15 primers only 3 primers showed polymorphic bands. The three primers which showed reproducible results were OPV-14, OPC-08 and OPL-12. OPV- 14 primer showed maximum polymorphism with highest number of polymorphic bands between F1 hybrid and parents of chilli (Capsicum) samples. Thus 0PV-14 primer can be further used to analyse the difference between hybrid and parent sample of tomato. It can also be used to check the genetic purity of tomato. |
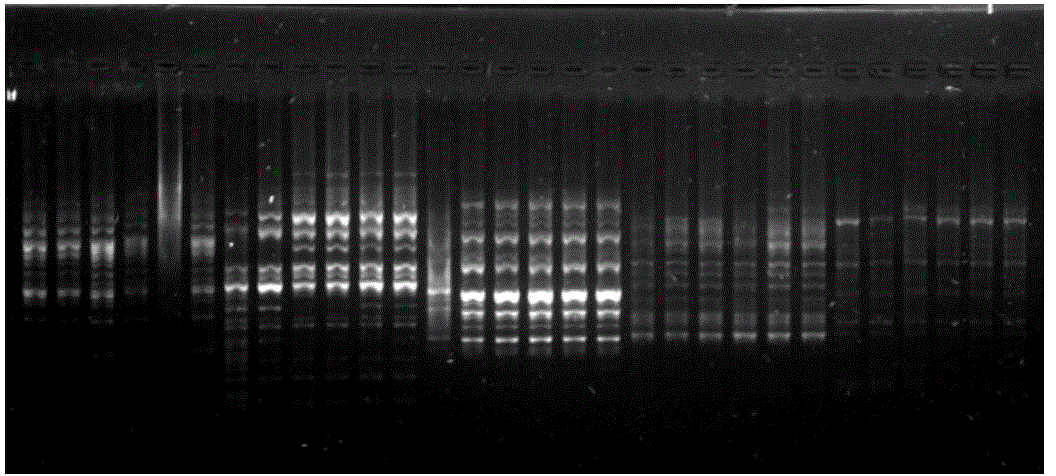 |
CONCLUSION |
| RAPD markers have found a wide range of applications in gene mapping, population genetics, molecular evolutionary genetics and plant and animal breeding. This is mainly due to the speed, cost and efficiency of the RAPD technique to generate large numbers of markers in a short period compared with previous methods. Therefore, RAPD technique can be performed in a moderate laboratory for most of its applications. Despite the reproducibility problem, the RAPD method will probably be important as long as other DNA-based techniques remain unavailable in terms of cost, time and labor. [13] The molecular fingerprinting of a plant variety is of the utmost importance for protecting a plant breeders’ rights (PBR).[14][15]Thus the RAPD profile of tomato and chilli from the present study shows that RAPD can be proved very useful for identification of hybrids from their respective parents. The method to distinguish the hybrids from parental lines using molecular markers is much more efficient and accurate. Study suggests that the primer which produces polymorphic bands can be further used as polymorphic marker which will prove promising in identification and genetic purity testing in case of tomato and chilli. |
ACKNOWLEDGMENT |
| We are thankful to the CytoGene Research & Development, Lucknow for providing the facilities and kind support throughout the research work. We are also thankful to the Almighty. |
References |
|