ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Shivdas Bajod1, Namdeo Chavhan1, Vijaykumar Markad2, Kisan Kodam2, Jagrut Waghmare3 and Sharad Shelke1*
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
In this study the investigations on the synthesis of N-Acyl Sulfonamide derivatives of pyrazole nucleus are reported. Ethyl-3-pyrazole carboxylate 1 has been used as starting material and it treated with aryl boronic acid in cupric acetate and DMF as a solvent afford N-aryl ethyl-3-pyrazole carboxylate 2 followed by alkaline hydrolysis to get N-aryl ethyl-3-pyrazole carboxylic acid 3. When compound 3 stirred with aryl sulfonylamide in the presence of DCC, DMAP in DCM affords N-pyrazolylcarboxasulfonamides4.
Keywords |
| N-acylsulfonamide, Pyrazole, Boronic acid, DCC, DMAP. |
INTRODUCTION |
| Nitrogen and sulfur containing compounds like sulfonamide is important biological compound. 1, 2Sulfonamides have been associated with antibacterial, 3 anticancer, 4,5and anti-proliferative 6 activities. Biphenyl derivatives of sulfonamides showed excellent angiotensin II receptor antagonist activity. 7 In the last decade, SAR study 8,9of sulfonamoyl compounds has been reported by various researchers. It plays a key role in the field of organometallic chemistry. As an example sandwich cobalt and rhodium complex with combination of N-acyl sulfonamides is applied for lung cancer therapy. 10N-ïÃÂá-ïÃÂâ -unsaturated acyl sulfonamides shows addition properties to organometallic reagents.11 The literature survey revealed that N-acyl sulfonamide derivatives have been used for chemoselectiveNacylation reagent. 12 Recently, application of sulfonamide in asymmetric conjugated addition of Grignard reagents to ïÃÂáïÃâ¬ÃÂïÃÂâ-unsaturated carboxylic acids have been investigated. 13 Due to the sulfonamides shows versatile bioactivity, researcher very much interested in their study of synthetic properties14 and pay attention towards synthesis of their derivatives15-17. Various methods for the synthesis of aryl and heterocycle derivatives of N-acyl sulfonamide18-20 have been reported in the literature. Pyrazole derivatives also exhibited antibacterial, 21 antifungal22 and anti-hyperglycemic23 activity. Pyrazole nucleus shows anti-HCV 24 as well as HIV-1 protease inhibitors25 property. Due to diverse biological activity associated with pyrazole derivatives. Various researchers have been focusing on their synthesis.26-27Newer different routes for the synthesis of pyrazole nucleus has been reported in the literature.28-30 Biological activity associated with N-acyl sulfonamide and in continuation of effort in the study of bioactive heterocycles,31-34 we promoted us to synthesis of N-pyrazolylcarboxasulfonamides and their biological evaluation. |
| In present investigation the synthetic approach was achieved in three steps as shown in Scheme-1. Initially commercially available ethyl-3-pyrazolecarboxylate 1 has been treated with aryl boronic acid with copper acetate afforded N-aryl ethyl-3-pyrazolecarboxylate 2. Compound 2, when subjected to alkaline hydrolysis gave corresponding acid 3, followed by treatment with aryl sulfonamide in presence of DMAP and DCC afforded Npyrazolylcarboxasulfonamides4 (Scheme-I). |
SCHEME-I |
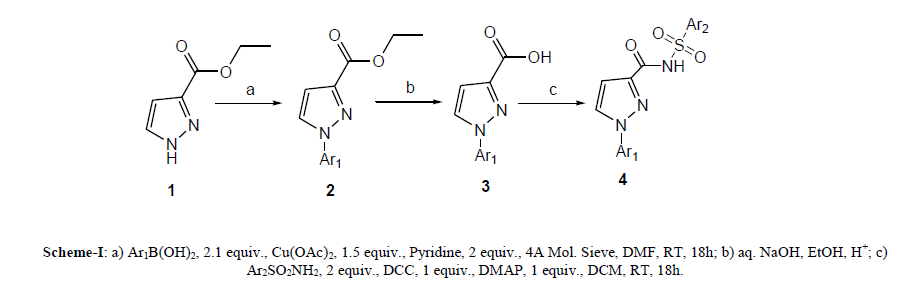 |
| In each step, progress of the reaction was monitored by TLC (Thin layer chromatograpy). Formation of the intermediate compound 2 was confirmed by TLC as well as M.P. and mixed M.P. Formation of acid 3 was confirmed by routine sodium bicarbonate test, TLC and M.P. and LCMS. Characterization data of synthesized compound has been summarized in Table 1. |
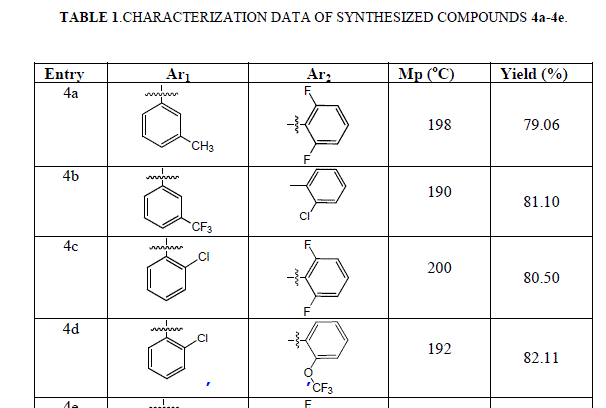 |
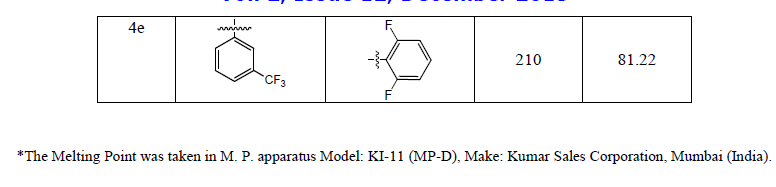 |
| The structure of the titled compounds 4a-4e was confirmed by spectral studies such as IR, 1HNMR, LCMS. |
III.EXPERIMENTAL |
| All the recorded melting points were determined on M. P. apparatus Model: KI-11 (MP-D), Make: Kumar Sales Corporation, Mumbai (India) and are uncorrected.FTIR spectra were recorded on a Perkin-Elmer FTIR spectrophotometer in KBr pellets. The NMR spectra were recorded on a Bruker 400 MHz instrument using CDCl3 (deutrated chloroform) as a solvent and TMS (tetramethylsilane) as an internal standard; the chemical shifts (ïÃÂä) are reported in ppm (part(s) per million) and coupling constants (J) are given in Hertz. Signal multiplicities are represented by s (singlet), d (doublet), t (triplet), dd (double doublet), and m (multiplet). Mass spectra were recorded on a Finnigan mass spectrometer. TLC was performed on pre-coated silica gel glass plates (Kieselgel 60, 254, E. Merck, Germany) Synthesis of N-Pyrazolylcarboxasulfonamides4a-4e A solution of N-aryl 1H-pyrazole-3-carboxylic acid 3 (1 equiv.) and aryl sulfonamide (2 equiv.) in 10 mL of DCM (dichloromenthane) was added drop wise, at R.T., to a stirred solution containing DCC (1 equiv.) and DMAP (1 equiv.). The reaction mixture was stirred at R.T. for 18 hrs. The progress of the reaction was monitored by TLC (Thin layer Chromatography). After completion of the reaction, the mixture was extracted with DCM, washed with 10 % aqueous NaHCO3 and dried over anhydrous sodium sulfate. The solvent was removed under vacuum and the crude residue was purified by column chromatography (20% ethyl acetate/n-hexane) to afforded titled compound 4a-4e. This typical experimental procedure was followed to prepare other analogues of this series. The compounds synthesized by above procedures have been listed in Table 1 with their characterization data. |
| 4a: IR (KBr) v/cm-1: 3243 (-NH), 1709 (–C=O), 1606 (C=N), 765 (C-F); 1H-NMR (400 MHz, CDCl3, δ, ppm):2.47 (s, 3H, CH3) 6.97-7.92 (m, 9H, HAr), 9.68 (s, 1H, NH); MS (m/z): 378 (M++1). |
| 4b: IR (KBr) v/cm-1: 3248 (-NH), 2948 (–CH3), 1705 (–C=O), 1660 (C=N), 757 (C-Cl); 1H-NMR (400 MHz, CDCl3, δ, ppm): 6.79-8.39 (m, 10H, HAr), 9.60 (s, 1H, NH); MS (m/z): 428 (M++1). |
| 4c: IR (KBr) v/cm-1: 3242 (-NH), 1709 (–C=O), 1663 (C=N), 760 (C-Cl); 1H-NMR (400 MHz, CDCl3, δ, ppm): 6.63- 7.87 (m, 9H, HAr), 9.63 (s, 1H, NH); MS (m/z): 398 (M++1). |
| 4d: IR (KBr) v/cm-1: 3250 (-NH), 1710 (–C=O), 1661 (C=N), 760 (C-Cl); 1H-NMR (400 MHz, CDCl3, δ, ppm): 6.93- 8.34 (m, 10H, HAr), 9.52 (s, 1H, NH); MS (m/z): 445.9 (M++1). |
| 4e: IR (KBr) v/cm-1: 3198 (-NH), 1715 (–C=O), 1671 (C=N), 765 (C-F); 1H-NMR (400 MHz, CDCl3, δ, ppm): 7.03- 8.00 (m, 9H, HAr), 9.65 (s, 1H, NH); MS (m/z): 431.8 (M++1) |
IV. CONCLUSION |
| In conclusion, various carboxasulfonamides4 having pyrazole nucleus were synthesized successfully by newer route. It was found that yield of titled compound is good. |
ACKNOWLEDGMENT |
| The authors are thankful to the Director of SAIF, Punjab University (Chandigarh, India) for the spectral analysis. We are also grateful to the Principal Dr. K. H. Shinde and Dr. A. B. Nikumbh, (HOD), S.S.G.M. College, Kopargaon, Ahmednagar (MH) for providing research facilities and constant encouragement. |
References |
|