ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
F. O. Faithpraise1*, B. Usibe1,J.Idung1, C. R. Chatwin2, R.Young2 and P.Birch2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The mosquito is a major pest that transmits many dangerous diseases and seriously damages human health. Hence, the reduction of their population by the use of a sustainable control method is a primary objective of this research. This mosquito reduction method utilises biological predators and a fermentation traptosubstantially improve the general environment and provide in-door clearance of mosquitoes. The frequency of capturing the pest mosquitoes by the predators is determined using a Pascal distribution, whilst the insect life cycle mortality is modelled using a Weibull distribution. The results from the model show that by using insect predators, a significant reduction of the larva stage of the mosquito population is possible and the fermentation mosquito trap greatly reduces the adult mosquito population without the application of insecticide spray.
Keywords |
| Fermentation trap, Mosquito eradication, Malaria reduction, Toxorhynchites larvae |
INTRODUCTION |
| The design of an adhesive film trap to catch the egg depositing mosquitoes by Facchinelli, et al [1], and Gama, et al [2]. The gold standard traps with other traps was done by [3], which success and mosquito trapping effect depends on adherence to strict usage or deployment instructions, otherwise the trapping method is rendered ineffective with a corresponding increase in mosquito population density. |
INTRODUCTION |
| Mosquitoes are a unique pest with several species that have a devastating effect on the health of human beings and other mammals. Despite many campaigns to control the mosquito population no favourable results have been reported. For instance the 2010 Nigeria Malaria Indicator Survey preliminary report by NMIS[4] indicates the existence of malaria parasites in almost every household, including in infants, despite the wide distribution of insecticide treated nets [5] and the use of several preventive measures like insecticides sprays, mosquito preventive creams, as enumerated by Faithpraise et al.[6]. Malaria, a major threat to the world and Africa, is an infectious disease caused by the parasite of the genus Plasmodium, transmitted mostly by the bite of an infected female anopheles mosquito. |
ECONOMIC IMPACT OF MOSQUITOES |
| More than 60% of outpatient visits in Nigeria are due to Malaria. The disease has impacted negatively on the nation’s economy with about 132 billion Naira lost to the disease due to the cost of treatment and loss of man-hours as illustrated byNGDTM [7]. The development of resistance to some anti-malarial medicines [8], [9], [10] has continued to threaten the effectiveness of the existing methods of mosquito control. Reduction of human work capacity and productivity and the financial burden of fighting Malaria in sub-Saharan African countries - including Nigeria, results in misery and suffering for the labour force that are necessary to drive the economy; this adversely affects the social and psychological well-being of individuals, families and sabotages the investment efforts of the Government. |
| Our goals are: the reduction of Malaria related morbidity and mortality; abatement of the socio-economic impact of the disease; reduction of the huge financial burden on the government and individuals and finally a decrease in the intake of anti-malarial drugs, which are very expensive for the poor masses. |
| To achieve our goal we propose a model that combines a mosquito trapping system called the Fermentation attractor and a biological control predator as illustrated in the following model. |
MATERIALS AND METHODOLOGY |
THE FERMENTATION ATTRACTOR SYSTEM (FAS) DESIGN |
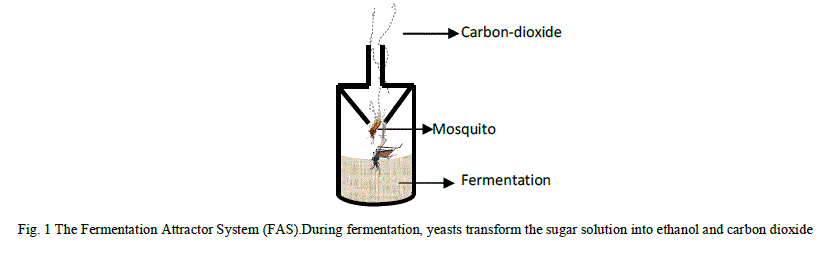
| The Fermentation Attractor Model is a simple mosquito attractor system, which is designed by reacting glucose with yeast to produce carbon dioxide (CO2) and ethanol: eqn. 1, the carbon dioxide attracts the adult mosquito from up to a mile away, see Fig. 1.The fermentation attractor system (FAS) is made up of yeast, warm water and dissolved glucose. Yeast is a microscopic fungus consisting of single oval cells that reproduce by budding, and are capable of converting sugar into alcohol and carbon dioxide + energy. |
| The elephant mosquito (Toxorhynchites) larvae is a predator of mosquito larvae. The Toxorhynchites are unique in that none feed on blood and, unlike many other mosquitoes, they are harmless to mankind. The larvae of all Toxorhynchites are predaceous on other mosquito larvae or small aquatic arthropods; they are therefore beneficial to mankind. They have the ability to consume 10 to 20 mosquito larva in a day and up to 5000 during their whole larval stage [11], [12], [13]. |
EXPERIMENTAL SET UP OF THE MODEL |
| • Fill the stainless steel vessel one-half full with warm water and dissolve glucose into the warm water . |
| • Add dry yeast into the solution. |
| Seal the entrance of the stainless steel vessel and allow a little opening wide enough to allow the passage of CO2 out, so attracting the mosquitoes into the fermenting fluid as shown in Fig.1. |
REACTION: |
| The reaction between the yeast and glucose is partially broken down to produce alcohol and carbon dioxide (CO2) as bye products, the carbon dioxide attracts the adult mosquito into the trap. |
RELIABILITY: |
| The reliability of this fermentation trap in attracting the adult mosquitoes stems from the fact that mosquito, as a blood sucking bug, is attracted to human beings and mammals via the exhaled carbon dioxide, octenol, odour from bacteria on the skin, a person’s secretor status and blood type [14], [15]. The mosquito is hyper-sensitive to carbon dioxide exhaled from mammals, it has poor eyesight and is very sensitive to mammal sweat scent from a half mile distance, [16], [17], [18]. |
 |
THE FERMENTATION ATTRACTOR & PREDATOR MODEL |
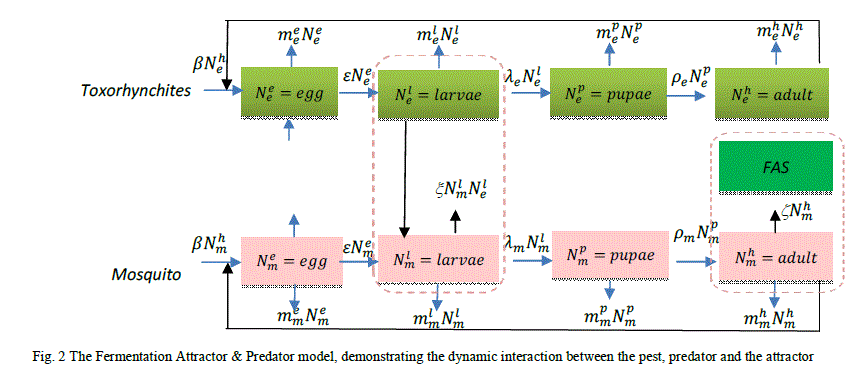
| A model of the interaction between the pest mosquito species and the predatorToxorhynchites adult and its life cycle stages and the fermentation attractor is illustrated in Fig. 2, which was created in order to observe the effect on the mosquito population. The model is illustrated by the following non-linear simultaneous ordinary differential equations 2-9 |
 |
 |
| The proposed model consists of eight simultaneous non-linear, ordinary differential equations (3) to (10), which are solved using a 4th order Runge–Kutta method as described by Klassische [19], [20], [21], [22] and using the average life span of all the insect life cycle stages and their mortality rates as described in the previous works of Faithpraise et al. [23]. The Weibull probability distribution function as described [24], [25] in their work on the Weibull distribution. Hence, the mortality rate can be modelled using the Weibull distribution function as in [26] to determine the various mortality rates of the pests and predators. |
| The negative binominal distribution or Pascal distribution as shown in equation 10-12, was used to determine the frequency with which predators capture prey and the probability of the FAS trapping an adult mosquito as shown previously in [27]. |
 |
| This models the scenario for the successive random trials that the predator undertakes, with each predation attempt having a probability of success ‘p’. The number of attempts that the predator must make in order to capture a given number of prey r has a negative binomial distribution where ‘I’ is the indicator function, which ensures that ‘r’ only adopts integer values and n is the mean. |
EXPERIMENTAL RESULTS |
| The results obtained in Fig. 3 illustrate the reproductive capability of a typical pest mosquito, which has had its blood meal. Since there were no control measures in place to manage the mosquito growth rate in the environment, within the space of 80 days the mosquito population density reaches its carrying capacity of 2.0 *107 per square kilometre |
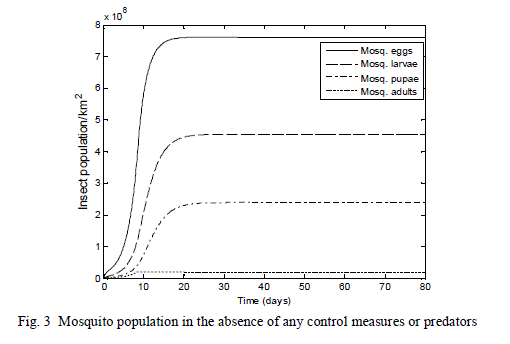 |
| The result of Fig. 3 shows a great increase in the population of the mosquito: eggs, larvae, pupae - from an assumed initial starting population of 3,200,000 to the peak of 7.60*108 eggs; 4.53*108 larvae, 2.38*108 pupae and the adult population reaches the carrying capacity of 2.0 *107 in the environment within 12 days. |
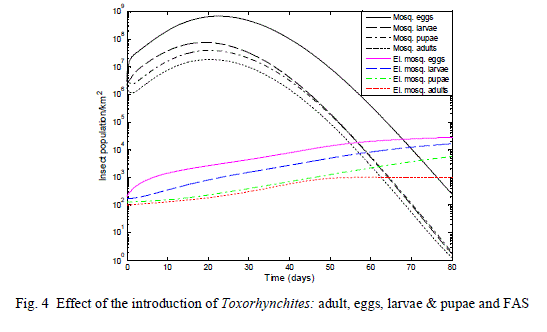
| A control measure was introduced by deploying 4 fermentation attractor units to different strategic locations within the one km2 area and aToxorhynchites population density of 200 eggs, 170 larvae, 150 pupae and 120 adults, were deployed as shown in the result of Fig. 4 |
| In Fig. 4, with the introduction of the Fermentation attractors, most of the mosquito adult population was attracted into the trap illustrated in Fig.1. The Toxorhynchites larvae were able to control the population of the larvae stage of the mosquito thereby controlling the overall population density of the mosquito. The effective control of the fermentation attractor and the predator was observed when the population density of the mosquito dropped from a peak of 6.69*108 to 249 eggs, 7.42*107 to 3 larvae, 3.84*107 to 4 pupae and 1.825*107 to 2 adults in 80 days. |
 |
DISCUSSION OF RESULTS |
| The results of Fig. 3 show that mosquito have the ability to multiple uncontrolably expecially if a blood meal is obtained. |
| The result of Fig. 4, illustrate a successful reduction of the population of mosquitoes as the predator established their population in the environment with the deployments of 4 fermentation attractors within a one square kilometre area. |
| The result demonstrates the possibility of obtaining a healthier environment with the deployment of fermentation attractors (FAS) and the Toxorhynchites species into the mosquito infested environments. |
| The combination of the FAS and predator to control the mosquito population demontrates the great potential of this strategy to significantly reduce the mosquito life cycle stages in a reasonable number of days either short term or long term. This duplex approach requires fewer preditors to be deployed, reducing the resources required to achieve the desired result. |
CONCLUSIONS |
| It is observed that as long as an environment is left uncared for, it will definitely become a breeding ground providing many mosquito hatcheries. When an environment is occupied with millions of mosquitoes, no preventive measures can cure or manage the mosquito pest. Therefore it is recommended that in addition to managing the environment and preventing it from becoming a breeding zone, a permanent control measure with a combination of Toxorhynchites predators and the FAS should be employed. |
| The deployment of FAS should be encouraged in the household and outdoor environment to reduce mosquito population in the surrounding environment. |
| Breeding, then deploying beneficial insect predators like the Toxorhynchites will transform a hostile environment into a zone habitable by humans, as these predators have a great ability to control mosquitoes at low cost and in a short amount of time. We also advise the restoration of natural habitats that will attract the visits of important naturally beneficial insects. |
| Breeding, then deploying beneficial insect predators like the Toxorhynchites will transform a hostile environment into a zone habitable by humans, as these predators have a great ability to control mosquitoes at low cost and in a short amount of time. We also advise the restoration of natural habitats that will attract the visits of important naturally beneficial insects. |
RECOMMENDATION |
| To minimize the mosquito-attraction into your home and your skin during the period of this experiment |
| • wash regularly to reduce bacteria and propanoic acid build-up on your skin. |
| • use mosquito repellents and mosquito treated nets until it is certified that the environment and homes are clear of mosquitoes. |
References |
|