ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
E.Nithya1, R.Radhai2,S.Prasanna3, K.Vaideki4 R.Rajendran5, S.Jayakumar6
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Oxygen plasma treatment and/or cellulase enzyme treatment was carried to improve the surface properties of cotton fabrics for medical applications. The influence of Plasma (P), enzyme (E), plasma preceded by enzyme (EP) and enzyme preceded by plasma (PE) treatments on hydrophilicity of the cotton fabrics was studied using standard wicking test. The physical and chemical modification due to plasma and enzymatic treatments were analyzed using SEM and ATR-FTIR spectroscopy. The FTIR results revealed that the oxygen plasma has modified the surface chemically by introducing oxygen containing groups (C=O, C-O, COO-) onto the fabric surface and enzyme have hydrolyzed the cellulose molecule. The physical etching of the fabric surface was observed in SEM photographs and weight loss measurements. The modified fabrics were given antimicrobial finish and tested for its antimicrobial activity against S.aureus and E.coli using AATCC 100 test. The EP treated fabric was found to have 100% bacterial reduction. The wash durability of the finished fabrics was also assessed and the results are discussed.
Keywords |
| Cotton fabric, Hydrophilicity, DC oxygen plasma, Cellulase, UATR-FTIR, Antimicrobial efficacy |
INTRODUCTION |
| Recently, plasma surface modification of textile materials found to replace the wet chemical, thermal and radiative processes [1-4]. Plasma is a partially ionized gas which when hits the surface of the material, modifies the surface property to a depth from 50-500Å without changing the entire material property[5,6].These ionized gas containing excited atoms, free radicals, electrons, ions and molecules interact physically and chemically with the fabric surface[7]. This type of interactions can be exploited to improve surface energy, hydrophilicity, dyeability, adhesion and to impart functional properties [8, 9]. The advantages of plasma surface modification are low cost of operation, highly efficient, rapid processing and a dry process for chemical modification of textile materials rather than using a high cost fiber with inherent built in performance properties [10]. On the other hand, cellulase enzyme hydrolyzes the cellulose fibers by cleaving the glucose units in the molecular chain thereby removing the micro fibrils on the fabric surface [10-12]. This introduces polar groups onto the fabric surface and makes the fabric hydrophilic [13]. Several authors [10, 14, 15] have reported that properties such as wettability, dyeability, adhesion, printability etc. can be improved by enzymatic hydrolysis. Moreover, these properties can be further enhanced by the combination of the above two treatments as reported by several authors [11, 13, 16, 17]. Such improvement of fabric property can be exploited for the enhancement of antimicrobial efficacy [18]. In particular, the improvement in hydrophilicity can increase the uptake of antimicrobial finish onto the fabric. Further, theseantimicrobial textiles can be used in the surgical zones as a first level protection for the doctors and the patients preventing cross infection by disease causing micro-organisms such as bacteria, fungi and virus [19-21]. However, very few literatures are available on the synergetic effect of plasma and cellulase enzyme treatment to improve the hydrophilicity which in turn the antimicrobial property of cotton fabrics [18]. We have reported first time the enzyme pretreatment for plasma surface modification which enhances the hydrophilicity and further increases the antimicrobial efficacy. In the present investigation, physical and chemical modifications of cotton fabrics were done using plasma and enzyme treatment to improve the hydrophilicity. The chemical changes on the treated fabrics were analyzed using UATR-FTIR spectroscopy. The surface morphology of the treated fabrics was studied using SEM and the antimicrobial efficacy of the treated fabrics was studied using AATCC 100 test and assessed for its wash durability. |
II. MATERIAL AND METHODS |
Materials |
| 100 % finished cotton fabrics with an EPI/PPI count of 72/54 cut into a dimension of 10 x10 cm were used for all the treatments. Initially the cotton fabrics were immersed in boiling water for 15 minutes and dried to remove the surface impurities and starch. Commercially available acid cellulase Bio-Lish 2.0 L (Avensa chemicals, India) was used for enzyme treatment. Pure oxygen (99.999%) was used for plasma treatment. Plasma Treatment Plasma treatment of cotton fabrics was carried out with oxygen in a 12’’ DC plasma chamber. The schematic diagram of the plasma chamber is shown in the figure 1. |
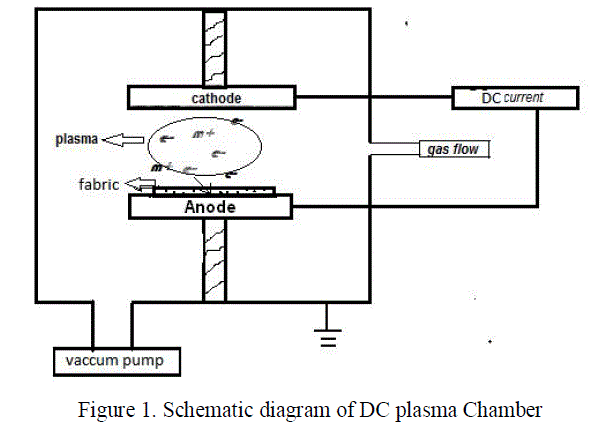 |
| Prior to evacuation, the fabric sample was placed onto the anode and then the chamber was evacuated to a base pressure of 10-3 mbar using a rotary pump. Process parameters such as gas pressure, inter-electrode distance, current and exposure time were optimized for the best treatment conditions of having maximum hydrophilicity. The optimized process parameters in this study are: gas pressure - 0.1 mbar, inter-electrode distance -2 cm, exposure time – 5 mins and current-60 mA. Enzyme Treatment Cellulase hydrolysis of cotton fabrics was performed in an incubator shaker with a material to liquid ratio (MLR) of 1:20 and temperature 55oC. For the enzymatic hydrolysis, the samples were treated for 30 minutes at pH of 5.5 using an enzyme dosage of 2% OWF (on weight of fiber). After the treatment, cellulase enzyme was de-activated by washing it thoroughly with hot water (95oC) for 10 minutes duration and then dried in air. Assessment of Hydrophilicity The hydrophilic behavior of the treated and untreated fabrics was analyzed using wicking test (BS 4554). A cotton fabric strip of dimension of 0.5 cm X 8 cm was suspended vertically with its lower edge having contact with distilled water. A spontaneous rise in the height of water in the fabrics due to capillary forces was observed. The height in the rise of water for a given time is taken as the direct indication of wicking rate of the fabric samples. The rise of water level in the fabric was measured for 20, 40, 60, 120, 180 and 240 seconds .The wicking height measured for 240 seconds was considered for assessment of hydrophilicity of the treated fabric [22].The measurements were made thrice for each sample and the average values are reported with a standard error of ±0.1cm. |
| The mean pore radius on the fabric surface was calculated using Lucas Washburn equation [23].The modified equation is stated as, |
 |
| where H - wicking height, r - Pore radius, η - Coefficient of viscosity of the liquid, γ - Surface tension of the liquid, t - Time taken for wicking. The slope of the plot between H2 and t gives the mean pore radius of fabrics. Weight Loss The weight loss of the fabric samples was calculated by measuring the dry weight of fabrics before and after the treatments. The percentage weight loss was calculated using the following equation: |
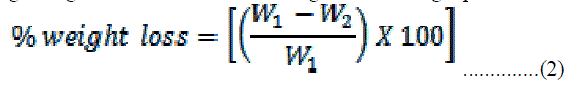 |
UATR- FTIR analysis |
| The fabric sample surface was examined using a UATR-FTIR (Universal Attenuated Total Internal Reflection Fourier Transformation Infrared) spectrophotometer. The spectra were recorded using a Perkin Elmer (Spectrum100) FTIR spectrometer in the range of 4000-650 cm-1 with a resolution of 4 cm-1. UATR consists of Zn-Se single crystal that collects the spectra from the cotton fabrics of dimensions 10 mm X 10 mm placed onto it. Pressure was applied to the samples to ensure a good contact between the sample and crystal to prevent the loss of IR incident radiation. The spectra were obtained for each sample at a resolution of 4 cm-1 with 32 scans. Surface Morphology Studies-SEM The surface morphology of untreated and treated cotton samples was studied using SEM (JEOL-JSAL 6360). Platinum was sputtered on to the fabric samples, as a conducting material to analyze the samples. Anti-microbial finish treatment The methanolic extract of neem leaf prepared as an anti-microbial finish for the cotton fabric was applied onto the fabrics using pad-dry cure method. The fabric samples were immersed in the extract with a material to liquid ratio of 1:20 for 10 mins. 8% citric acid was added as a cross linking agent to fix and increase the durability of the finish. Subsequently the fabric samples were passed through a padding mangle running at a speed of 10 m/min with a pressure 1 kgf /cm2 to remove excess solution. After padding, the fabrics were air-dried and then cured at 140°C for 3 min. Quantitative assessment of anti-microbial activity (AATCC 100) The anti-microbial activity of the fabric samples was ascertained quantitatively by AATCC 100 method [24]. 1.0 ml of the test inocula (Staphylococcus aureus, Escherichia coli) were loaded on the swatches (treated and untreated) of 4.8±0.1cm diameter. They were then transferred to the sterile AATCC bacteriostasis broth. After an incubation of 24 hours, up to 10-7 serial dilutions were made for all the samples. 0.1ml sample from each dilution were spread plated onto the sterile AATCC bacteriostasis agar plates and incubated at 37 oC for 24 hours. The percentage reduction of bacteria by the treatment was then calculated using the following formula: |
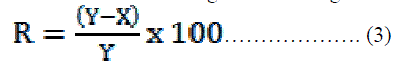 |
| Where R is the % reduction and X and Y are the number of bacteria recovered from the inoculated treated and untreated swatch respectively. Wash durability test (IS: 687-1979) Washing test was carried out as per IS: 687-1979 by using a neutral soap (5gpl) at 40oC ïÃâñ 2oC for 30 minutes, keeping the material to liquid ratio at 1:50, followed by rinsing, washing and drying. After drying, the test samples were assessed for anti-microbial activity using AATCC 100 procedure up to 30 washing cycles. |
III. RESULTS AND DISCUSSION |
Effect of Plasma and Enzyme treatment on the Hydrophilicity of the cotton fabric |
| The effects of oxygen plasma and enzyme treatments on the hydrophilicity of the cotton fabrics were determined by measuring the wicking height as shown in the figure 2. From the figure it is observed that the wicking height considerably improved for the oxygen plasma treated fabrics when compared with the enzyme treated and untreated fabrics. This indicates that the oxygen plasma treatment has enhanced the formation of polar species on the fabric surface due to surface oxidation and etching the waxy layers in the primary wall of cotton fabric which is evident from SEM analysis. When the fabrics are treated with cellulase enzyme, it gains access to the cellulose and removes the noncellulosic compounds on the fabric surface making it hydrophilic. However, more significant improvement in hydrophilicity takes place when a combination of plasma and enzymatic treatments (PE and EP) are done. This is attributed to the synergetic effect of enzymatic and plasma etching which leads to physical and chemical modification of the fabric samples. When plasma treatment is done prior to enzyme treatment (PE), oxygen plasma etches the cotton surface inducing more reactive sites which may hinder the action of cellulase on cotton fabric [17], as evident from the ATR-FTIR results. But when enzyme treatment is done prior to plasma treatment, the fabric surface is made smoother after the enzyme hydrolysis, which increases the accessibility of plasma species with the cellulose. This may be the reason for the slight increase in the hydrophilicity of EP treated fabrics than PE treated fabrics. |
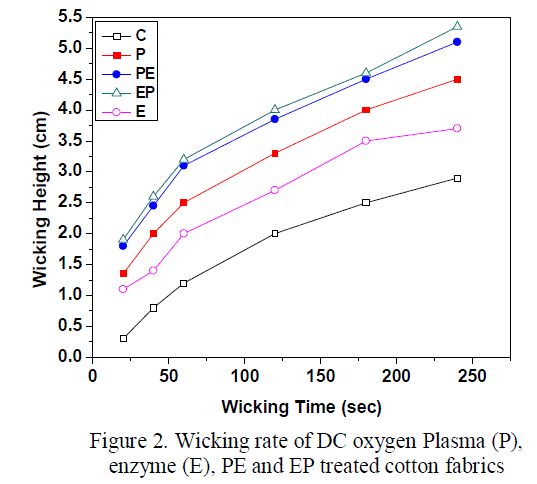 |
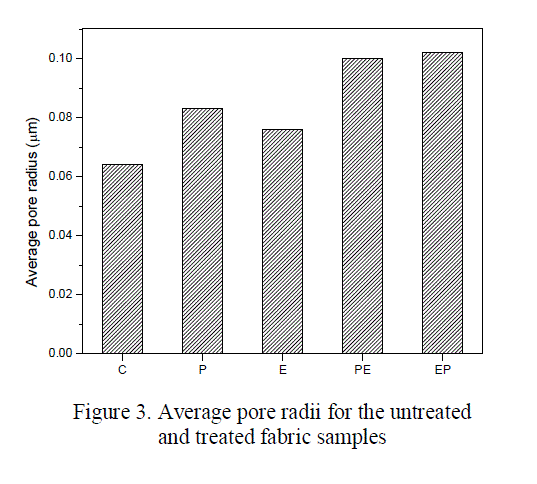 |
| The average pore radii found using Lucas Washburn’s equation for the untreated and treated fabrics is shown in the figure 3. From the figure it is evident that, the pore radius of oxygen plasma treated fabric (0.083 μm) is found to be higher than enzyme treated fabric (0.076μm). This increase in the pore radius results in the increase of hydrophilicity, which in turn increases the water holding capacity of the fabric. The pore radius is found to be maximum for EP treated samples (0.102 μm) and it is almost same for PE treated samples (0.100μm) which might be due to the etching effect of enzyme as well as plasma. |
UATR-FTIR Analysis |
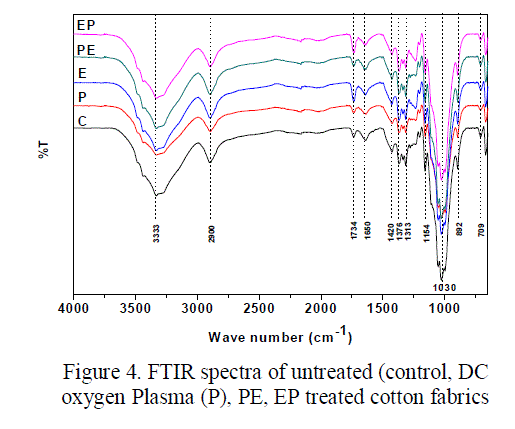
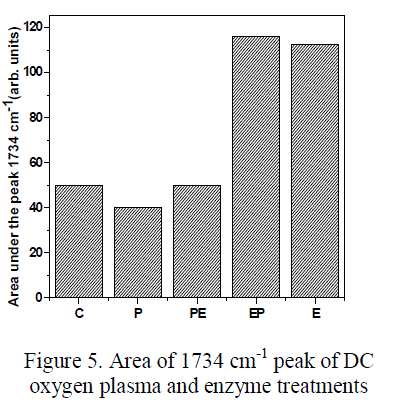
| UATR-FTIR, a surface sensitive technique was used to study the chemical modification on the surface of treated fabrics. The spectra of the untreated and treated cotton fabrics are illustrated in figure 4. The characteristic bands of the untreated and treated cotton fabrics are presented in the table 1[25]. The peak corresponding to 1734 cm-1 represents the carbonyl group in aldehyde or carboxylic acid. The area under 1734 cm-1 peak of the treated and untreated fabric is shown in the figure 5. From the figure 5 it is evident that the intensity for the oxygen plasma treated fabrics was found to be less when compared to the control fabric. This decrease in carbonyl group (1734 cm-1) in oxygen plasma and PE treated fabric may be due to two reasons, first the removal of pectin and wax which has –COOH group in it, followed by the formation of carboxylic acid group by oxidation of primary alcohol present in the cellulose fabric. And conversion of few carboxyl (COOH) to carboxylate (COO-) group through deprotonation [26, 27, 28]. The existence of carboxylate group can be confirmed by the presence of two peaks at 1650cm-1 and 1420 cm-1 for asymmetric and symmetric stretching of COO- ion respectively. However when the fabrics are treated with cellulase enzyme the concentration of carbonyl group increases [13]. Furthermore, when the fabrics are treated with oxygen plasma and enzyme combination, it is observed that for EP treated fabrics the concentration of carbonyl group increases, whereas for PE treated fabrics the concentration of carbonyl group decreases. |
 |
 |
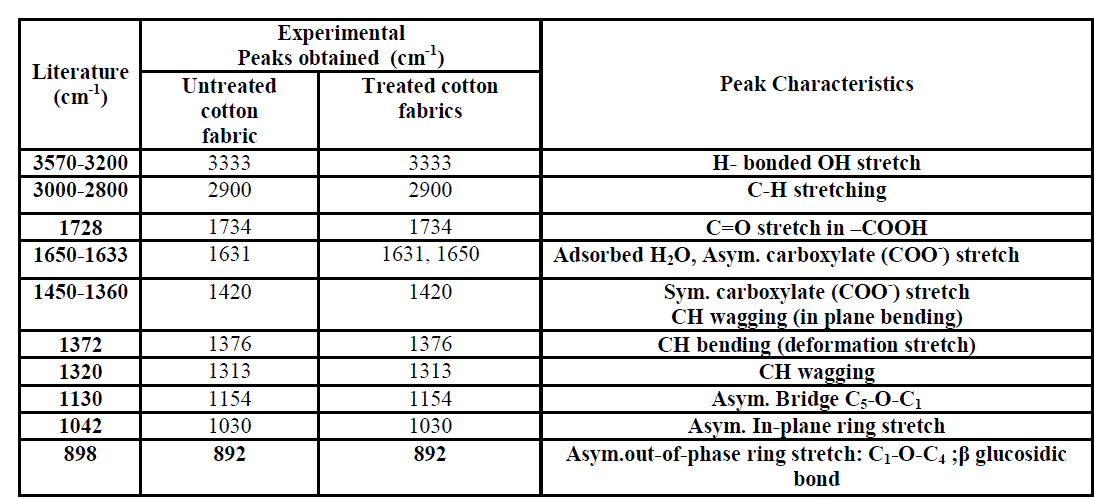 |
| But the hydrophilicity of the PE and EP treated fabrics were found to be more or less equal. The reason could be, for PE treated fabrics, when the fabrics are first exposed to oxygen plasma, more carboxylate groups (COO-) are formed, further, when it is treated with enzyme there is no drastic increase in the carbonyl group due to the enzyme hydrolysis, since the carboxylate groups are more stable. Table. 1 Infrared absorption frequencies of untreated and treated cotton fabrics (cellulose) For EP treated fabrics, the intensity of carbonyl group increases significantly due to enzyme hydrolysis and subsequent oxidation [13]. When |
| enzyme treated fabrics are exposed to oxygen plasma, there is no significant increase in COO- groups which in turn does not affect the hydrophilicity of the fabrics considerably. Thus the ATR FTIR results provide an insight into the formation of COO- groups in oxygen plasma and PE treated fabrics which also increases the hydrophilicity similar to EP treated fabrics. Percentage weight loss and SEM analysis The physical changes that occurred during the treatments were studied by weight loss and SEM results. In order to study the morphological changes due to plasma and enzyme treatments SEM measurements were performed. |
 |
 |
| Figure 6a shows the SEM images of untreated cotton fabric with protruding fibrils, groves, cracks and non cellulosic compounds. When cotton fabrics are exposed to oxygen plasma (figure 6b), the surface fibrils and non cellulosic compounds are etched by the bombardment of plasma species. |
 |
 |
| The cellulase enzyme treated cotton fabric (fig 6c), shows a smoother surface than the untreated fabrics. This is due to attack of enzyme on the cotton fabric which peels of the protruding fibers making the surface smoother. This removal has led to the weight loss in enzyme treated fabrics to a percentage of 1.5 as observed in figure 7. The surface of PE treated fabrics (fig 6d) is found to have micro cracks and pores and the fibers are almost smooth when compared to untreated fabrics. |
 |
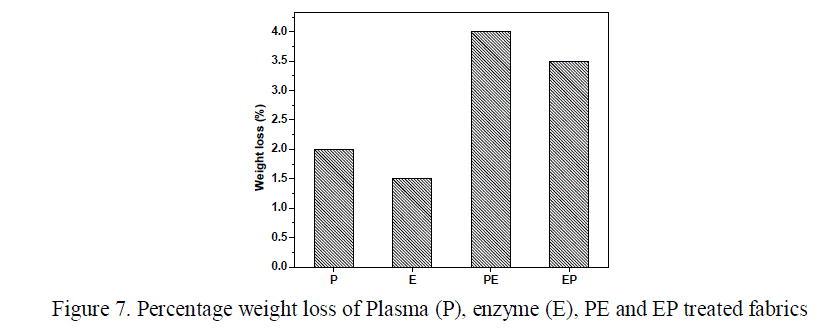 |
| The weight loss of PE treated fabrics was found to be higher (4%) due to the higher etching tendency of enzymes by penetrating into the pores introduced by plasma species. The surface appearance of the EP treated fabrics was examined from figure 6e and was found to have cracks and grooves formed by the etching effect of post plasma treatment which lead to a weight loss of about 3.5%. Although the images do not provide complete information about the fabric surface, we are able to observe the etching effect of plasma and smoothening effect of enzymes. Quantitative assessment of Anti-microbial Activity The oxygen plasma, enzyme, PE and EP treated fabrics were processed with methanolic neem leaf extract to impart anti-microbial property. The percentage bacterial reduction of Staphylococcus aureus and Escherichia coli for the treated fabrics post-treated with neem leaf extract were confirmed quantitatively using AATCC 100 method. The results obtained were tabulated (Table 2). It is observed that, the activity against the S.aureus organism was greater when compared to E.coli which may be due to the difference in the cell wall characteristics between Gram positive and Gram negative bacteria . |
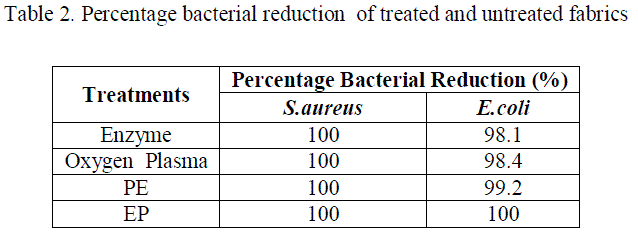 |
| The percentage reduction of S.aureus organism was 100% for all the treated samples, but there was a decrement in the activity against E.coli in the order of enzyme < plasma < oxygen plasma < PE < EP treated samples. The enhancement in the antimicrobial activity may be attributed to the increase in the uptake of the neem leaf extract as a result of plasma and enzyme modifications. The results suggest that the combination of treatments especially EP treatment is a promising approach for the preparation of antimicrobial textiles with excellent antimicrobial activity. Wash Durability Although the fabrics were found to possess excellent antimicrobial activity, it has to be tested for its wash durability. To assess the durability of the antimicrobial finish, the samples were subjected to wash durability test as per IS: 687-1979 procedure. |
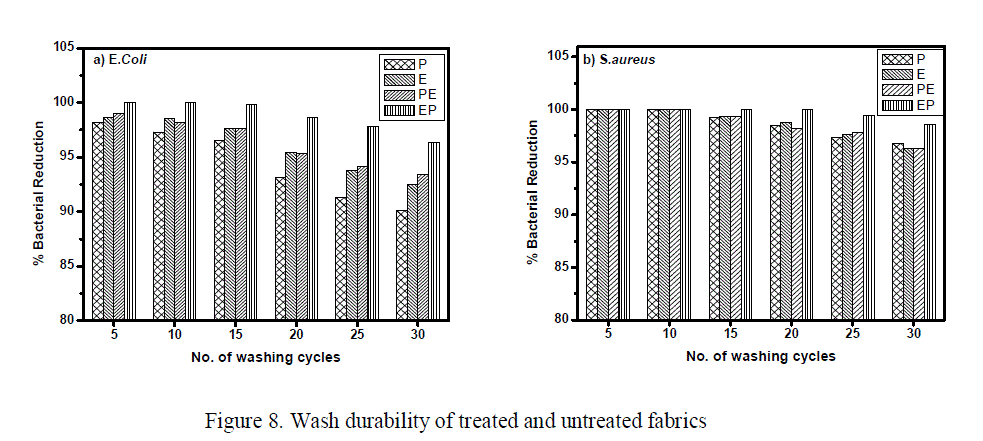 |
| The activity after 5, 10, 15, 20, 25 and 30 washing cycles was assessed using AATCC 100 test against S.aureus and E.coli and the results are plotted (fig 8a &b) . From the figure it is observed that the antimicrobial activity was higher for EP treated samples up to 30 washing cycles for both the organisms. |
IV.CONCLUSIONS |
| Cotton fabrics were treated with DC oxygen plasma or cellulase enzyme and their combinations to study their effect on the hydrophilicity and in turn anti microbial activity. All the treated samples showed significant increase in hydrophilicity and it was found to be highest for EP treated fabrics. The increase in hydrophilicity is due to the physical and chemical modification of fabric surface induced by the synergetic effect of enzyme and plasma. Oxygen plasma treatment was found to be effective in removing the non cellulosic compounds and introducing more number of polar groups like COO-, C=O onto the fabric surface. Enzyme treatment was effective in smoothening the fabric surface and formation of COOH groups by enzymatic hydrolysis followed by oxidation. Weight loss measurements indicated that PE treated fabrics was found to have more weight loss than EP treated fabrics. Thus the plasma and enzyme surface modifications have enhanced the hydrophilicity of cotton fabrics increasing the antimicrobial efficacy. It can be concluded that the cotton fabrics can be suitably modified by EP treatment to improve the antimicrobial activity and durability in an environmental friendly way. |
ACKNOWLEDGEMENTS |
| The authors would like to thank the Principal and Management PSG college of Technology for providing the necessary facilities for carrying this research work. |
References |
|