ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Umar Muhd Sani Associate Professor, Department of Chemistry, Nigeria Police Academy, Wudil, Kano State |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Peasant farmers in northern Nigeria indigenously use many plants to protect cereals and legumes against pest damage during storage. Phytochemical screening of the seed extracts of Parkia biglobosa indicated the presence of alkaloids, flavonoids, cardiac glycosides, polyphenols, tannins, and terpenoids in ethanol extract and alkaloids, steroids, terpenoids, flavonoids and saponins in petroleum extract. In this study the test insects (Callosobrucus maculatus) for the antifeedant efficacy were cultured and the first generation progenies that emerged were used. Beans were dressed with different doses (0.04, 0.08, 0.12, 0.16, and 0.20g) of the extracts and the seed damage data were used to estimate the weevil perforation index (WPI). WPI was found to have decreased as concentration of extracts increased. Compared to the control which has WPI of 50%, both extracts were found to be highly effective antifeedants against the cowpea weevil storage pest for the time allowed for the study. The ability to protect the seeds from damage has been clearly shown by the percent protectant ability (PPA) whose value at 0.20g dose for ethanol extract was 94.74% while for petroleum ether extract it was 81.82%. The ethanol extract was therefore more effective than the petroleum ether extract at the highest dose. The results of this study therefore showed that Parkia biglobosa seed extracts possessed some antifeedant activity which may be associated with the presence of the secondary metabolites detected by the phytochemical screening.
Keywords |
| Antifeedant, secondary metabolites, cowpea beans, Parkia biglobosa, Callosobruchus maculatus, weevilperforation- index, percent-protection-ability |
INTRODUCTION |
| The cowpea beans, Vigna unguiculata (L.) Walp., is an important legume in West Africa and it is widely cultivated and eaten in Nigeria [1]. Nigeria accounts for 70% of the world’s production of cowpea beans [2]. It is a major food crop in tropical countries and popularly used as protein suppliment for meat and fish; moreover, it contains digestible carbohydrates and lysine [3]. The cowpea beans, however, are vulnerable to insect pests of which the cowpea weevil, Callosobruhus maculatus (Coleoptera: Bruchidae) is the most important [4]. Callosobruchus maculatus is a major field to storage pest of cowpea with initial infestation starting in the field and expanding rapidly during seed storage. It is a cosmopolitan pest ranked as the principal post-harvest pest of cowpea in the tropics [5,6]. It causes substantial quantitative and qualitative losses manifested by seed perforation and reduction in weight, market value and germinability of seeds [7]. Under traditional storage conditions, 100% infestation of cowpea occurring within 6 months or more often within 3 to 5 months of storage is common [8,9]. The occurrence of this cowpea pest therefore constitutes a major problem contributing to huge food shortage in tropical and subtropical countries of the world. At least 4% of the total annual production valued at over 30 million US dollars is lost annually in Nigeria alone to this bruchid [10]. |
| Attempts made to reduce the damage by C. muculatus led to the acquisition of improved traditional storage methods. Conventionally, method of protecting stored cowpeas is known to depend on the use of synthetic chemical insecticides [11]. Synthetic insecticides have proved very effective in the control of the weevil [12]. In Nigeria, fumigants like aluminum phosphide, dusts like Gammalin “A” dust, Lindane dust and Pirimiphos-methy1 (Actellic) have been extensively used to control these pests [8] Caswell and Akibu, 1981). Control of cowpea pests using chemical insecticides is, however, being discouraged because of health hazards to humans and environmental concerns amongst others [13]. Moreover, there is a distinction between antifeedants and insecticides. Insect antifeedants are compounds that temporarily or permanently reduce or prevent insects from feeding and therefore antifeedants differ from insecticides by their indirect, rather than direct, action. Thus an antifeedant should not kill the insect although death can be caused by starvation due to inhibited feeding. An alternative approach for the reduction of the attack by the weevil is therefore the use of natural products of plants origin. Peasant farmers in northern Nigeria indigenously use various plants to protect cereals and legumes against pest damage during storage. The low cost and safety of botanical extract is gaining more importance in controlling cowpea pests [14,15,16,17] and it has been concluded that plant-derived extracts are environmentally safe, degradable, and target specific insecticides [18]. Plant powders’ for antifeedant activity tests are easy to prepare and apply and many plants have been used as powders in pilot tests. Among some five edible plant products powders investigated by Ajayi and Wintola [2], the African locust bean (Parkia biglobosa) was found to be the least potent in the ability to suppress oviposition, inhibit progeny emergence or to cause egg mortalities of Callosobruchus maculatus. However, extracts of fresh or dry plants materials are usually more effective than powders [19]. Mostly the extracts are effective against adult beetles, being repellents or toxicants to the beetles. The need to study the effects of some solvent extracts of the Parkia biglobosa seeds has been realized. The present research, therefore, has evaluated the antifeedant efficacies of the petroleum ether (60-80 oC) and ethanol extracts of the seeds of Parkia biglobosa. |
II. MATERIALS AND METHODS |
2.1 Experimental Cowpea |
| The black-eyed cowpea (Vigna unguiculata), contained in their pods, used for this experiment were purchased from a farmer in Wudil market, Kano State in Northern Nigeria. The beans were left in the pods and only removed when needed for the insect culture and the antifeedant study. |
2.2 Insect Culture |
| The test insects (Callosobrucus maculatus) were obtained from previously infested black-eyed cowpeas and placed in three plastic petri dishes (150 x 25 mm) covered with fine nylon netting that permits the passage of air only. Twenty fresh blackeyed peas (Vigna unguiculata L. Walp.) removed from bean pods were placed each in two different plastic petri dishes and in each twenty insects (10 males and 10 females) were introduced to allow for mating and oviposition. The petri dishes were covered with nylon netting held in place by means of rubber bands to prevent the escape of the insects. The culture was maintained on a laboratory bench at room temperature (22-25°C) with indirect outdoor window lighting and ambient humidity (ranging from 45%-55% RH) for 7 weeks. The first generation progenies that emerged were used for the experiment. |
2.3 Preparation of Plant Material |
| Five hundred grams (500g) of sun-dried seeds of Parkia biglobosa (African locust bean), were secured and grounded into powder. The powder was placed in transparent polyethylene bag and stored in a refrigerator. |
2.4 Extraction of Plant Material |
| 200g of the plant material were exhaustively extracted by maceration in a 2000ml volumetric flask using 1000ml of petroleum ether (60-80 °C) for three days at room temperature. The petroleum ether seed extract was filtered and another 500ml of the solvent was again poured onto the residue and extracted for another two days; the extract again was filtered into the previous one. The solvent was removed by distillation under reduced pressure using a rotary evaporator at 40 °C. The residue of the seeds after petroleum ether extraction was dried and next extracted with ethanol using the same procedure as above to afford the ethanol extract. |
2.5 Phytochemical Screening |
| The phytochemical screening for terpenoids, steroids, alkaloids, tannins, saponins, polyphenols, flavonoids and cardiac glycosides on the ethanol and petroleum ether extracts were carried out using standard methods. |
2.6 Antifeedant Bioassay |
| Each of the two extracts were weighed at different levels (0.04, 0.08, 0.12, 0.16, and 0.20g) and dissolved in 2ml of methanol. 0.25ml of the solution was transferred into a vial and allowed to dry overnight. The dried sample was redissolved in 5ml of acetone. For each extract, 20 cowpea beans were counted out and placed in a beaker. The beans were then dressed with the prepared acetone solutions. Another 20 beans for the control were dressed with acetone only. Five males and five females of newly emerged cowpea weevils were then introduced into the eleven beakers of the test samples plus that of control. All the beakers were covered with a net that permits the passage of air and prevent the escape of the insect. The set up was allowed to stay for five weeks and the numbers of infested and un-infested beans were counted in all the beakers including the control. Weevil Perforation Index (WPI) has been calculated according to the methods of [20] Rotimi and Ekperusi, 2012). Subtracting the value of WPI from 100 gives the Percent Protectant Ability (PPA). |
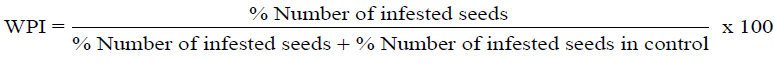 |
II. RESULTS AND DISCUSSION |
3.1 Phytochemical Screening |
| The results of the phytochemical screening, as shown in table 1.0 below, revealed that alkaloids, flavonoids, cardiac glycosides, Polyphenols, tannins and terpenoids were present in the ethanol extract of the seeds of Parkia biglobosa. The steroids and saponins were only detected in the petroleum ether extract which also contained terpenoids. |
 |
| Secondary metabolites such as phenolic compounds, alkaloids, flavonoids and terpenoids have been identified to exhibit feeding deterrent activity [21,22]. Duke [23] further reported that there is growing evidence that most secondary products of plants are involved in the interaction of plants with other species, primarily the defense of the plant from plant pest. In this regard flavonoids have been considered [24] as one of the plant's defensive systems against phytophagous insects. A research paper on flavonoids and their related compounds have indicated that these compounds possessed antifeedant activities against the subterranean termite Coptotermes formosanus Shiraki [25]. Structure-activity relationship study of these compounds showed that the pyran ring (A-ring) was responsible for the antifeedant activity whereas the absence of this ring increased feeding-preference activity. Saponins have been reported to exhibit clear insecticidal properties in several pest insects [26,27]. Some of the reported observed effects of saponins are increased mortality, lowered food intake, weight reduction, retardation in development and decreased reproduction [27]. A tricylic sesquiterpenoid isolated from Senecio palmensis has been shown to have high antifeedant potency on the Colorado potato beetle, Leptinotarsa decemlineata [28]. The alkaloids isolated from the seeds, seed pods and flowers of Erythrina latissima were reported [29] to have antifeedant activities against Spodoptera littoralis. Terpenoids, among other secondary metabolites, have been reported to be the most successful pesticides [23]. Some of these secondary metabolites could be responsible for the antifeedant property observed in Parkia biglobosa seed extracts. In fact complex mixtures of secondary compounds in plant extracts has been reported to contribute to a great deal for synergism, which enhances the joint action of active compounds against insect and reduces the rate of resistance development [30]. |
3.2 Bioactivity testing |
| From table 2.0 below, the results obtained for the bioactivity testing of the petroleum ether extract, ethanol extract and the control revealed that the Weevil Perforation Index (WPI) decreases as the concentration of the extract increases. This is to say that both the petroleum ether and ethanol extracts of the Parkia biglobosa seeds were effective at high dosage rate, that is higher concentration is required for higher effectivity. |
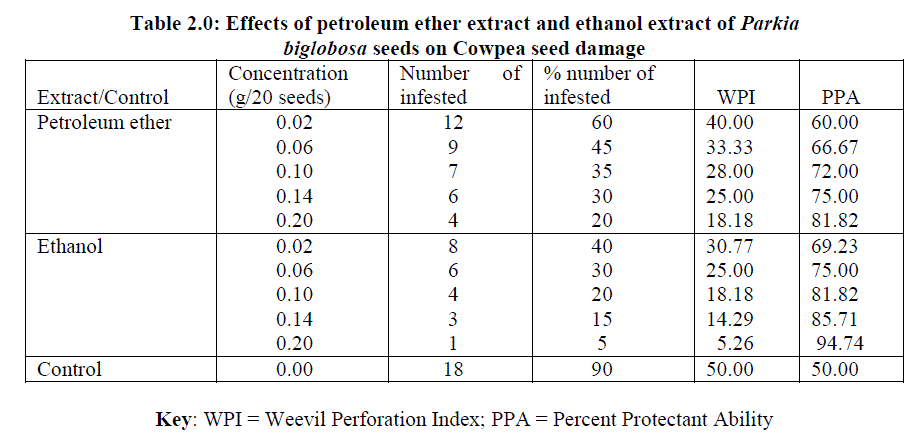 |
| From table 2.0, the Weevil Perforation Index for petroleum ether extract decreases from 40.00% to 18.18% as the dosage level increases from 0.02g to 0.20g. The same was also for the ethanol extract whose Weevil Perforation Index decreases from 30.77% to 5.26% as concentration increases from 0.02g to 0.20g, indicating ethanol extract to be more effective than the petroleum ether extract at the highest dose. The ability to protect the seeds from damage has been clearly shown by the Percent Protectant Ability whose value at 0.20g dose for ethanol extract was 94.74% while for petroleum ether extract it was 81.82%. The two extracts are effective in controlling the infestation of or serving as antifeedants against the cowpea weevil storage pest when compared to the control which has the highest Weevil Perforation Index of 50% for the time allowed for the study. Overall, the results have shown that both extracts have similar effect. The Weevil Perforation Index indicates the ability of the extract in protecting the cowpea seeds from damage and values of this index exceeding 50 % is regarded as enhancement of infestation by the weevil or negative ability of the plant material or insecticides tested [31]. |
VI. CONCLUSION |
| The results of bioactivity screening showed that Parkia biglobosa seed extracts possesses some antifeedant activity and is effective at higher concentration. The extracts of the Parkia biglobosa seeds contain alkaloids, flavonoids, steroids, cardiac glycosides, terpenoids, tannins, polyphenols and saponins. The presence of these phytochemical compounds in the extracts may be responsible for their antifeedant bioactivities. Moreover, complex mixtures of secondary compounds in plant extracts has been reported to contribute to a great deal for synergism, which enhances the joint action of active compounds against insect and reduces the rate of resistance development. |
References |
|