ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Jaya Mary Jacob1, SoubhikKumar Bardhan2, Raj Mohan B3*
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The concentration of lead and selenium in sea water samples near industrial areas located in Mangalore, Karnataka, India were determined using atomic absorption spectrophotometer. Sea water sample in proximity to petro chemical industries were analysed with high lead and selenium concentrations. Three different genera of fungi were isolated from this lead and selenium contaminated sea water sample. Filamentous fungi isolated belonged to the genera Aspergillus, Fusarium and Penicillium. They were screened for their resistance to lead and selenium. Results reveal that all the isolated strains were tolerant to lead and selenium but the level of tolerance varied in the isolates from the same source. Aspergillus and Penicillium isolates were the most tolerant to lead and selenium and exhibited strong growth, often exceeding the control (isolates grown in agar medium without heavy metals). Tolerance index of Penicillium was reportedly high when compared to the two other isolated fungi, which makes it an attractive potential candidate for further investigations regarding their ability to synthesize Lead Selenide quantum dots with high quantum confinement effects.
Keywords |
| Sea water,Lead, Selenium, Fungi, Tolerance Index. |
INTRODUCTION |
| Industrialization in the current century has pioneered extensive research in the field of environmental impact concerned with heavy metals and metalloids. Of particular interest are the chalcogen group and the related salts with heavy metals. Of these, the salt of Selenium (Atomic number 34) with Lead (Atomic number 82) is of environmental relevance owing to the promising applications of Lead Selenide Quantum Dots bound with remarkable quantum confinement effects as conducive photo catalysts and as leaders in future energy crisis. While the principal lead emission sources into the body come from lead water pipes, lead-based paints and leaded petrol, selenium is a by-product of various electrolytic metal refining, paint and petroleum industries. The effluents from these industries usually find way to water bodies resulting in increased levels of these metals. The contamination of natural sources due to the effluents accompanied by its toxicological and physiological importance, has led to an increasing interest in the determination of these elements [1]. Long term accumulation of heavy metals and metalloids in natural sources of water from the industries not only lead to bio magnification and eutrophication but also turn these water bodies as sources for isolation of heavy metal tolerant microbial species. Prior research reports the isolation of microbes from various contaminated water bodies including lakes, well, sea, industrial effluents and domestic sewer [2, 3]. The bio-sorption of heavy metals using fungal strains has been studied by researchers world-wide. Sastryet.al(2003) studied the bio-synthesis of metal nano particles using fungi and actinomycetes. Because of their tolerance and metal bioaccumulation ability, fungi are taking the centre stage of studies on biological generation of metallic nanoparticles [4]. Review suggests that these biological entities with metal tolerance could serve as agents for the cost effective biosynthesis of quantum dots [5].The present study attempts to determine the concentration of Selenium and Lead from sea water samples near industrial areas. The presence of heavy metal resistant fungus is probable in these heavy metal contaminated sites. The above samples were thus used to isolate heavy metal tolerant fungi and to quantify their tolerance index. |
II. MATERIALS AND METHODS |
| A. Collection of samples |
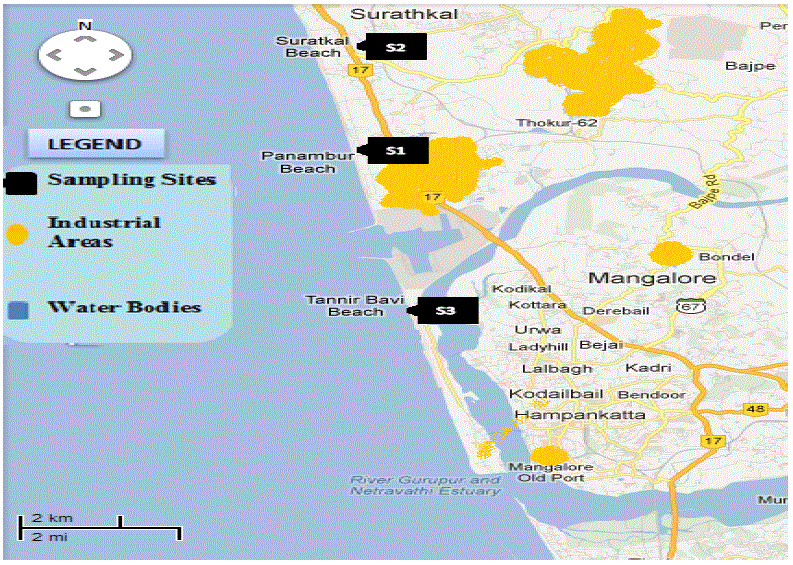
| Sea water and sediment samples were collected in clean sampling containers from beaches near Mangalore Refineries and Petrochemicals Pvt. Ltd, Mangalore (Fig.1). These samples were brought to laboratory and kept in refrigerator at 4oC for further processing. |
 |
| B. Lead and Selenium analysis in samples Water and sediment samples were analyzed for their total content of lead and selenium. The sediment samples were oven dried at 105°C. A sample of 1 g was digested with 5 ml HNO3 and 10ml HCl. Digestion was carried out on a hot plate until dense fumes evolved and a clear solution was obtained. The clear solution was filtered through a Millipore filter (0.45 μm) and diluted to 50 ml with distilled water prior to analysis. As for liquid samples, 50 ml of sea water was directly used for analysis using double beam GBC932 plus Atomic Absorption Spectrometer with selenium and lead lamps. Stock Solutions of Lead and Selenium for AAS were purchased from HiMedia Laboratories Pvt Ltd. Working standard solutions of Selenium and Lead were made by appropriate dilution of the respective 1000ppm standard stock solutions. C. Isolation of micro-organisms from polluted sites The water sample with highest contamination was enumerated for micro-organisms employing a serial dilution technique using Potato Dextrose Agar (Hi-Media, Mumbai, India) containing 20 ppm of Pb and Se. The 1000 ppm stock solutions of Pb and Se were made in double distilled water using Lead Nitrate and Sodium Selenite. The stock solution of heavy metals was sterilized separately and added to sterilized potato dextrose agar (PDA) medium under sterile conditions. Sea water samples were serially diluted up to 10-8 and 1ml of diluted sample (10-8 and 10-6)was added in sterilized petri plates. 20 ml of PDA medium containing 20ppm of the heavy metals was poured in these petri plates and incubated for least 3 days at 25°C. Developed colonies were randomly picked and purified isolates were by streaking the colonies repeatedly in PDA medium. Pure cultures of isolated micro-organisms were identified using the keys of Pitt (1979) and Domschet.al. (1980) [6, 7]. The cultures were characterized to the genus level on the basis of macroscopic characteristics like colonial morphology (color, appearance of colony, shape). |
| D. Growth Response Study Heavy metal tolerant (20 ppm) fungal isolates were further screened for growth response towards Pb, Se at concentrations of 5, 10, 15 and 20 ppm on PDA. All the fungal spores were seeded on PDA medium containing the respective concentrations of the metals in combination. Streaking of fungal isolates on normal PDA medium served as control (normal growth). Observations on growth of fungal isolate were made by measuring the colony diameter every day after 72 h of incubation. The tolerance index of the different fungi was estimated by measuring the radius of the colony extension (mm) in presence of 20ppm of metal against the control (medium without metal) after at least 7 days of incubation. The index is defined as the ratio of the extension radius of the treated colony to that of the untreated colony. The fungal strain with maximum tolerance index was chosen for further studies. E. Uptake of Pb and Se by tolerant fungal isolate from liquid media The more tolerant fungal isolate was subjected to broth studies in potato dextrose broth medium containing Pb and Se in a concentration range of 25-125 ppm.Potato dextrose broth containing the respective concentrations of the metals was dispensed in 100 ml lots to 250 ml conical flasks and sterilized at 15 lbs/psi for 15 min. These flasks were inoculated with 1 ml of freshly prepared spore suspension of the fungal isolate and put on shaker at 150 rpm at 280C for 5 days. Fungal growth was harvested after 5 days through filtration using Whatman filter No. 42. The harvested fungal biomass was rinsed with double distilled water 3–4 times and dried in hot air oven at 80oC for 18h. The dry weight of the biomass was calculated. |
III. RESULTS AND DISCUSSION |
| A. Sediment and water analysis: Common sources of lead and selenium include discharge from industries such as electroplating, plastics manufacturing, fertilizer producing plants and wastes left after mining and metallurgical processes [8]. The effluents from these industries are consequently discharged into the environment. Introduction of metals in various forms into the environment can produce numerous modifications of microbial communities and affect their activities [9, 10] In the present study, different fungi were isolated from sea water near industrial areas after analyzing the lead and selenium content in the same. The selenium and lead content in the collected sea water and sludge samples is listed in Table I. Results indicate higher concentrations of lead as well as Selenium in sea water samples in proximity to industrial areas. Although there is no significant levels of Selenium in the samples, the evidence by researchers that health risks may occur at exposures below the current European Union and World Health Organization upper limit of 10μg/l, draws relevance. Analysis of lead content in the water and sludge samples show a similar trend with the sea water showing relatively higher concentration of the heavy metal. According to EPA (2011), the permissible limit of lead in water samples in 15ppb [11]. The samples analyzed in the current study manifest higher levels of lead and selenium owing to the proximity of the sampling sites to petrochemical and refinery industries. |
 |
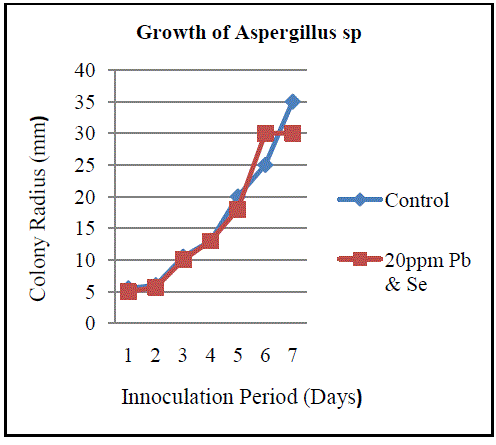
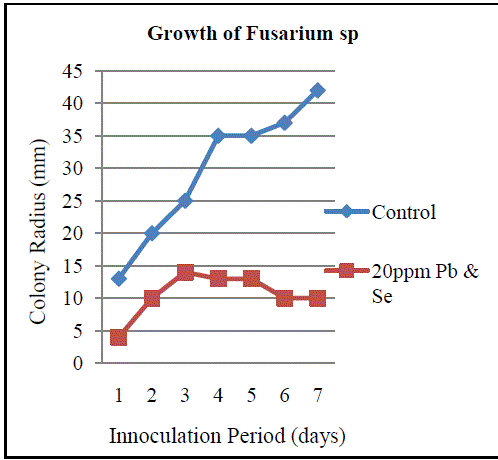
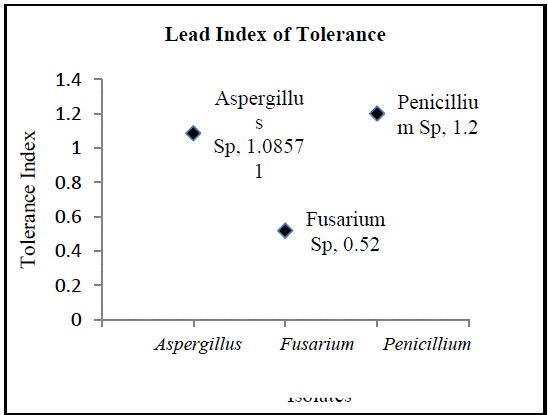
| B. Isolation of Heavy Metal Tolerant Fungi: Fungi and yeast biomasses are known to tolerate heavy metals [12]. In the present study, three fungal isolates tolerant to heavy metals were isolated from sea water sample contaminated with lead and selenium. All three isolated fungi exhibited tolerance to these metals individually and in combination. Fungi isolated belonged to the genera Aspergillus, Penicillium and Fusarium (Fig.3, Fig. 5 andFig.7). The isolates could tolerate 20 mg/l each of lead and selenium. Although prior research reports the isolation of fungi from natural sources with tolerance limits up to 25ppm, the fungi isolated in the current study are of relevance as the source of isolation is relatively less polluted compared to the sources reported in prior studies [13]. Observations on colony diameter of the fungi illustrate an inverse relationship between colony diameter and metal concentrations. The growth of Aspergillusspin the presence and absence of lead and selenium is shown in Figure 2. There is a linear growth phase in first 5 days of cultivation, followed by decline phase. On the 6th day the colonyradius of Aspergillus reached its maximum. Surprisingly, the decline phase in presence of selenium and lead is not so evident comparing to the control. Maximum colony diameter was observed on the 6th day for both control and the growth of Aspergillus in presence of the metals. The observed colony diameter of Aspergillus grown on PDA amended with 20 mg/l of Se and Pb is depicted in Figure 3. Similar growth patterns were exhibited by Fusarium and Penicillium. Although Fusarium reached a maximum diameter in 4 days, the colonies grown on amended PDA showed inhibited growth on the 7th day of incubation (Fig.4). Of the three fungal isolates, Penicilliumsp was found to fit better in the growth curve with a maximum colony diameter within 6 days of inoculation and morphologically healthier colonies (Fig.6, Fig.7). Our observations are seconded by the findings of Sun and Shao (2007) who demonstrated that both intracellular bioaccumulation and extracellular bio-sorption contributed to the high resistance of Penicilliumsp to heavy metals. To compare the resistance levels of our isolates to individual metals, the tolerance index of the metal tolerant strains for lead and selenium and was calculated. The index of tolerance showed significant difference between isolates from the same source. Our results are in agreement to the findings by Shaziaet.al (2012) who confirmed that the response of isolates to heavy metals depended on the metal tested, its concentration in the medium and on the isolate under consideration [14]. Although prior researchers highlight AspergillusSp isolated from industrial waste water as promising biological entities with metal tolerance, the current study showed the significantly higher Pb and Se tolerance indices in Penicilliumsp as compared to Aspergillussp isolated from sea water. Results of the present study affirm the statement that the level of resistance depended on the isolate tested, as well as the site of its isolation. Furthermore, findings by NatarajanVelmuruganet.al (2010) on the high level of lead resistance in Penicillium isolates from heavy metal contaminated mine soil signifies the importance of this fungi as an important bio sorbent of heavy metals [15]. Though the tolerance to selenium did not vary significantly between Aspergillussp and Penicilliumsp, the Se as well as Pb tolerance indices was remarkably lower in Fusarium sp. The Pb and Se tolerance index was pertinently higher in Penicilliumspcompared to the other isolates (Fig.8, Fig.9). The variation in the metal tolerance may be due to the presence of one or more strategies of tolerance or resistance mechanisms exhibited by fungi. The higher tolerance levels in Penicillium may probably be due to the production of organic chelators, acids and anti-oxidants by the heavy metal tolerant fungi during growth [16]. |
 |
 |
 |
 |
 |
 |
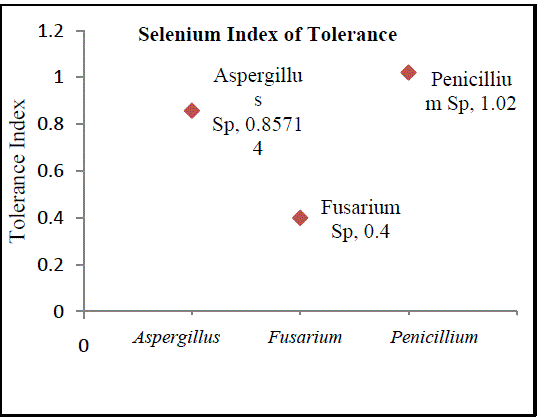 |
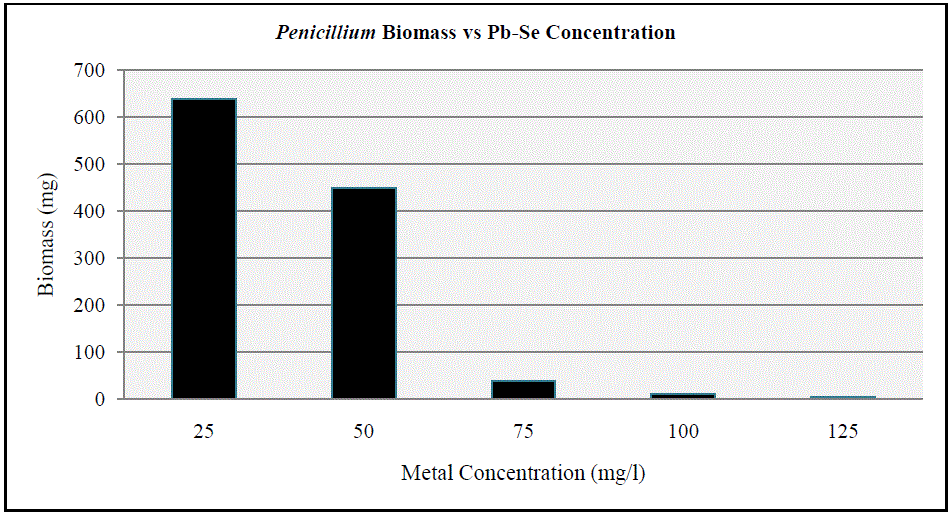
| Based on the high index of tolerance showed by Penicillium, the metal tolerant strain was subjected to higher concentrations of the metals in typical broth studies using PDB. As the concentration of the metals increased from 25 ppm to 50 ppm, there was a slight reduction in biomass of the fungi as shown in Figure 10. Results clearly indicate that the fungi could not tolerate metal concentrations beyond 75 mg/l and that the fungal dry weight significantly dropped at metal concentrations of 100 mg/l and 125 mg/l. |
 |
IV. CONCLUSION |
| The present study concludes that sea water near industrial areas contain lead and selenium salts in concentrations beyond the tolerable limits. Abundance of these metals in sea water makes it potential candidates for the isolation of lead and selenium tolerant fungal species. Three fungi were isolated namely; Aspergillus sp., Fusarium sp., Penicillium sp. Growth studies in the presence and absence of metals and tolerance studies revealed that Penicillium is more physiologically adapted to higher concentrations up to 50ppm of Pb and Se. Hence Penicilliumsp isolated from Pb-Se contaminated sea water can serve as promising biological agents for the synthesis of Lead Selenide based nano materials that is likely to lead the future solar energy sector. |
ACKNOWLEDGEMENT |
| The authors are thankful to the Department of Chemical Engineering, National Institute of Technology Karnataka, Surathkal for providing all the laboratory facilities for this work.Special thanks to all our friends and family members for the moral support and encouragement. |
References |
|