ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Kavitha Vijayaraghavan1, S.Mohamed Ali 2,4, R.Maruthi 3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The aim of this study was to investigate the phytochemical composition and antioxidant activity of Chromolaena odorata and Annona squamosa collected from Ooty hills and Ealagiri hills respectively. The leaf extract of Chromolaena odorata and seed extract of Annona squamosa with different solvents were investigated. The phytochemical composition was carried on the leaf extracts of Chromolaena odorata and seed extract of Annona squamosa, revealed the presence of active ingredients such as glycosides, steroids, Saponins, Phenols, Flavonoids, Terpenoids and Tanins. The leaf extracts and seed extracts were evaluated for antioxidant activities by DPPH radical scavenging assay. Among three accessions with different solvents used, maximum antioxidant activity found ethanolic leaf extract from Chromolaena odorata and seed extract of Annona squamosa followed by others. The present study reveals that these plants are of therapeutic potential due to their high free radical scavenging activity.
Key words |
| Chromolaena odorata, Annona squamosa, antioxidant activity, phytochemicals. |
I.INTRODUCTION |
| Plants are the richest resource of drugs of traditional systems of medicine, modern medicines, nutraceuticals, food supplements, folk medicines, pharmaceutical intermediates and chemical entities for synthetic drugs (Hammer et al., 1999). Medicinal plants have been used as traditional treatments for numerous human diseases for thousands of years. Medicinal plants are valuable natural resources and regarded as potentially safe drugs and have been tested for biological, antimicrobial and hypoglycemic activity also play an important role in the modern medicine [Hassawi and Kharma, 2006; Bhatet al., 2009]. |
| Therapeutic benefits can be traced to specific plant compounds; many herbs contain dozens of active constituents that, together, combine to give the plant its therapeutic value (Jeeva et al., 2012). Phytochemical screening of various plants has been reported by many workers (Mojab et al., 2003; Parekh and Chanda 2008). These studies have revealed the presence of numerous chemicals including alkaloids, flavonoids, steroids, phenols, glycosides and saponins. The phenolic compounds are one of the largest and most ubiquitous groups of plant metabolites (Hagerman et al., 2008). A number of studies have focused on the biological activities of phenolic compounds which are antioxidants and free radical scavengers (Evans et al., 1995; Cespedes 2008; Reddy et al., 2008). |
| Antioxidants are known to inhibit oxidational stress13. Reactive Oxygen species (ROS), including free radicals such as superoxide anion radical (O-2), hydroxyl radical species (OH.), singlet oxygen(1O2) and hydrogen peroxide(H2O2) are active oxygen species that are often generated by biological oxidation reactions of exogenous factors (Crutti 1990; Halliwell and Gutteridge 1990; Motlhanka et al., 2008; Motlhanka and Nthoiwa, 2012). These oxidative mediators can lead to the damage of biological structures such as nucleic acids, proteins and lipids (Gutteridge and Halliwell, 1994). Many free radicals have been implicated in the causation of several diseases such as liver cirrhosis, atherosclerosis, cancer, diabetes, ageing and Alzheimer’s disease (Patel et al., 2010). |
| Besides, phenolic compounds and flavonoids are also widely distributed in plants which have been reported to exert multiple biological effects, including antioxidant, free radical scavenging abilities, anti- inflammatory, anticarcinogenic etc. [Miller, 1996]. While, flavonoids are a group of polyphenolic compounds with known properties, which include free compounds with known properties, which include free enzymes and anti-inflammatory action [Chu et al., 2000]. |
| Chromolaena odorata is being used traditionally for its many medicinal properties, especially for external uses as in wounds, skin infections, inflammation etc. Studies have demonstrated that the leaf extract has antioxidant, antiinflammatory, analgesic, anti- microbial, cytoprotective and many other medicinally significant properties. The ability of wound healing is attributed to the antioxidant property of the drug which helps in conserving the fibroblast and keratinocyte proliferation on the site (M.N.Vaisakh. et al., 2011). |
| Chromolaena odorata is locally used for its medicinal properties in Vietnam for wound healing traditionally, the aqueous extract and decoction from the leaves of this plant have been used for the treatment of soft-tissue wounds, burns, and skin infections (Phan et al., 1993; 1998; 2000; 2001 and Thang et al., 2001). |
| Annona squamosa, sugar apple is native to the tropic America and is widely grown throughout the tropics in India (Abdalbasit et al., 2012). Phytochemical studies on A.squamosa showed many active compounds which are having many pharmacological activities such as anti-inflammation, anti-tumour activities (Pardhasaradhi et al., 2004) |
II.PROPOSED METHODS |
Collection of Chromolaena odorata and Annona squamosa |
| The healthy plants of Chromolaena odorata were collected from Ooty hills. Annona squamosa fruits were collected from two different geological area - Ealagiri hills & Andhra Pradesh. Seeds were collected from the fruits and both were dried and used for all experiments. |
Preparation of the plant extract |
| Preparation of the extracts was done according to a combination of the methods used by Pizzale et al., (2002) and Lu and Foo, (2001). About 15g of dried leaf fine powder of Chromolaena odorata and seed powder of Annona squamosa plant materials were extracted with 150 mL methanol (80%), ethanol (75%), ethyl acetate (Merck, extra pure), petroleum ether and aqueous extract for 1 min using an Ultra Turax mixer (13,000 rpm) and soaked overnight at room temperature. The samples were then filtered through Whatman No. 1 paper in a Buchner funnel. The filtered solution was evaporated under vacuum in a rota-evator at 40 °C to a constant weight and then dissolved in respective solvents. The concentrated extracts were stored in airtight container in refrigerator below 10ºC. |
Phytochemical Screening of Chromolaena odorata and Annona squamosa |
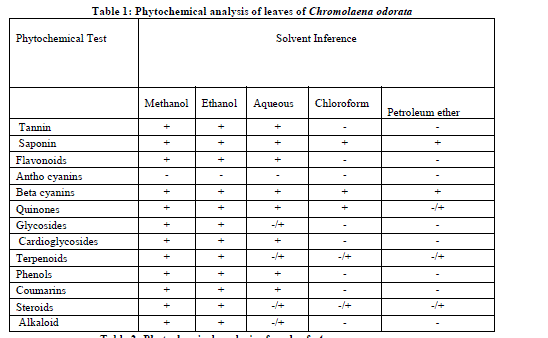
| The phytochemical screening of leaf and seed extracts was assessed by standard phytochemical methods (Brinda et al., 1981; Siddiqui and Ali, 1997; Savithramma et al., 2011). Phytochemical screening was carried out the qualitative chemical composition of leaf and seed extracts using different solvents and coloration to identify the major natural chemical groups such as tannins, saponins, flavonoids, phenols, terpenoids, alkaloids, glycosides, cardiac glycosides, coumarins and steroids. General reactions in this analysis revealed the presence or absence of these compounds in the leaf and seed extracts tested. |
Determination of Anti oxidant activity of Chromolaena odorata and Annona squamosa |
| Antioxidant activity in the ethanolic leaf and seed extracts were estimated for their free radical scavenging activity by using DPPH (1, 1-Diphenyl-2, Picryl-Hydrazyl) free radicals (George et al., 1996). |
Antioxidant activity: Qualitative analysis of Chromolaena odorata and Annona squamosa |
| 50μL of leaf extracts of Chromolaena odorata and seed extract of Annona squamosa were taken in the microtiter plate. 100μL of 0.1% methanolic DPPH was added over the samples and incubated for 30 minutes in dark condition. The samples were then observed for discoloration; from purple to yellow and pale pink were considered as strong and weak positive respectively. The antioxidant positive samples were subjected for further quantitative analysis. |
Antioxidant activity: Quantitative assay of Chromolaena odorata and Annona squamosa |
| Leaf and seed extracts of 100μl were mixed with 2.7ml of methanol and then 200μl of 0.1 % methanolic DPPH was added. The suspension was incubated for 30 minutes in dark condition. Subsequently, at every 5 min interval, the absorption maxima of the solution were measured using a UV double beam spectra scan (Chemito, India) at 517nm. The antioxidant activity of the sample was compared with known synthetic standard of (0.16%) of Butylated Hydroxy Toluene (BHT). |
| Radical scavenging activity was calculated by the following equation: |
| DPPH radical scavenging activity (%) = [(Absorbance of control - Absorbance of test sample) / (Absorbance of control)] x 100 |
III.EXPERIMENT AND RESULT |
| Scavenging activity for free radicals of DPPH (1,1-Diphenyl-2-picrylhydrazyl) has widely used to evaluate the antioxidant activity of natural products from plant and natural sources. Free radicals have aroused significant interest among scientists in the past decade. Their broad range of effects in biological systems has drawn the attention of many experimental works. It has been proved that these mechanisms may be important in the pathogenesis of certain diseases and ageing. Many synthetic antioxidant components have shown toxic and/or mutagenic effects, which have shifted the attention towards the naturally occurring antioxidants (Galvez and Cordero, 2005; Tepe, et al., 2005; Mammadov et al., 2011). |
| Wild accessions of leaves of Chromolaena odorata and seeds of Annona squamosa were used for antioxidant studies. Analysis on different extraction of methanol (80%), ethanol (75%), petroleum ether, ethyl acetate and aqueous extract showed the presence of antioxidants. |
| 50μl of leaf extracts (methanol, ethanol and petroleum ether, chloroform and aqueous extracts of leaves of Chromolaena odorata and seeds of Annona squamosa were estimated for free radical scavenging activity using Diphenyl-2-picryl hydrazyl (DPPH) assay. The samples observed for its bleaching from purple to yellow and pale pink were considered as strong positive and weak positive respectively. |
| Among the five different extracts of Chromolaena odorata and Annona squamosa, the ethanolic leaf extract of Chromolaena odorata and ethanolic seed extract of Annona squamosa recorded the most effective DPPH radical scavenging activity (94.29 %) and (77.14%) values being close to synthetic antioxidant (BHT) as positive control (76.3%). In each case, ethanolic leaf extracts recorded higher percentage of free radical scavenging activity than methanolic extractions followed by aqueous, petroleum ether, chloroform extract. |
| In the present study, phytochemical screening was performed with methanol, ethanol, chloroform, petroleum ether and aqueous extracts of the leaf of plant name. The ethanolic leaf extract of Chromolaena odorata and seed extract of Annona squamosa were rich in Glycosides, alkaloids, flavonoids, steroids, phenols, tannins and saponins than methanolic and aqueous extracts (Table 1&2). |
| Phytochemical constituents such as tannins, flavonoids, alkaloids and several other aromatic compounds or secondary metabolites of plants serve as defense mechanism against predation by many microorganism, insects and herbivores. The curative properties of medicinal plants are perhaps due to the presence of various secondary metabolites such as alkaloids, flavonoids, glycosides, phenols, saponins, steroids, etc (Britto and Sebastiana, 2011). Thus the preliminary screening test may be useful in the detection of the bioactive principles and subsequently may lead to the drug discovery and development (Doss 2009). Tannin compounds are present in plant name. The growth of many fungi, yeasts, bacteria and viruses was inhibited by tannins (Chung et al., 1998).The presence of alkaloids and saponins in leaf extract, The biological function of alkaloids and their derivatives are very important and are used in analgesic, antispasmodic and bactericidal activities (Stary et al., 1998). Saponins have properties of precipitating and coagulating red blood cells and they also have cholesterol binding properties, formation of foams in aqueous solutions and haemolytic activity (Sodipo et al., 2000) and traditionally saponins have been extensively used as detergents, as pesticides and molluscicides, in addition to their industrial applications as foaming and surface active agents and also have beneficial health effects [Shi et al., 2004]. Plant steroids are known to important for their cardio tonic activities and also used in nutrition, herbal medicine and cosmetics. Thus the preliminary screening tests may be useful in the detection of the bioactive principles and subsequently may lead to the drug discovery and development. Further, these tests facilitate their quantitative estimation and qualitative separation of pharmacologically active chemical compounds. |
 |
 |
 |
IV.CONCLUSSION |
| In the conclusions, phytochemical screening and antioxidant activity of medicinal plants is very important in identifying new sources of therapeutically and industrially important compounds. It is imperative to initiate a critical step for screening of plants for secondary metabolites. The present communication attempt was to assess the status of phytochemical properties and antioxidant activity in leaves of medicinal plants to improve the health status of people and also to use the same in nutraceutical products of commercial importance. The results indicate that the plant material may become an important source of compounds with a high potential to protect health. |
References |
|