ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Sopan Mansing Rathod Associate Professor, P. G. & Research Dept. of Physics, Abasaheb Garware College, Karve Road, Pune, Maharashtra, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Nanocrystalline MgxNi0.2Cu0.8-xFe2O4 (x = 0.5 & 0.6) ferrite material has been synthesized by sol-gel method for different compositions. The prepared materials were sintered at 400 0C for four hours, after that it was characterized by XRD, SEM, VSM and IR before laser irradiation. Then the prepared samples was irradiated by He-Ne laser of power 35mW with different time and after it was characterized to study the different properties of the samples by using X-Ray Diffraction (XRD). The Infrared Spectroscopy (IR) confirmed that the synthesized material is ferrite. The ferrite material may be plays vital role in biomedical sensor. The SEM shows surface morphology of the ferrite material. He-Ne laser irradiation on nanocrystalline ferrite materials shows remarkable changes like particle size decrease after irradiation of laser and coercivity of the sample changes. The lattices plane and bond length of the sample also calculated from XRD.
Keywords |
| He-Ne laser, nanocrystalline MgNiCuFe2O4, IR, XRD, UV, SEM. |
INTRODUCTION |
| Spinel ferrites are an important class of compounds having large variety of electronic, magnetic and catalytic properties, an which is depends on the processing condition, sintering temperature ,time, chemical composition and type of additive [1].The Sol-gel auto combustion is simplest and speed up method to prepared nano ferrite[2].The properties of nano ferrite particle are different from bulk material, because of that it is applicable in medical and engineering like targeted drug delivery, biomedical application and data storage devices as well as small size magnet [3- 8]. This technique can be used for enhancing the structural and magnetic properties [9]. Sol-gel combustion route can be investigated nano structure, cation occupancy and magnetic behaviour [10]. In this research work investigated the effect of laser on nano-crystalline Mg Ni Cu Fe2 O4 materials |
II. EXPERIMENTAL PROCEDURE |
| Samples were prepared, MgxNi0.2Cu0.8-xFe2O4 for (x=0.5,0.6) by sol-gel method. Materials are used AR grade pure nitrate of respectively by calculating stoichiometric ratio and mix appropriate amount of material in DI water. Then heat that materials at 75oC on magnetic starrer for 20 min after that add liquid ammonia to maintain pH 7.Then maintain temp 100oC up to material was ignited. Then prepared material was sintered at 400oC for 4 hr and use this sintered powder for different characterization. The material was then irradiated by He-Ne laser with power 35 mW for 1 and 2 hrs. Then the material was again characterized by XRD to determine the grain size after irradiation. This technique can be used for enhancing the structural and magnetic properties |
III. EXPERIMENTAL RESULTS |
1) 1. X-Ray Diffraction |
| The phase formation behavior of spinel ferrites of was studied by XRD. Fig.1 shows the X-ray diffraction patterns of MgxNi0.2Cu0.3Fe2O4 & Mg0.6Ni0.2Cu0.2Fe2O4 samples before and after irradiation by laser. From X-ray analysis the structure is spinel cubic8. After laser irradiation, the diffractogram indicates the formation of a distorted cubic structure beside the spinel cubic. This is reflected in the partial dissociation of the single phase cubic structure.Using the XRD data calculate lattice constant of all samples values are listed in Table1. From Table 1 lattice constant increases with increases in irradiation time. The crystallite size was calculated from Full Width at Half Maximum (FWHM) for all the peaks using Scherer formula: |
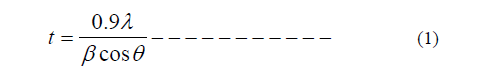 |
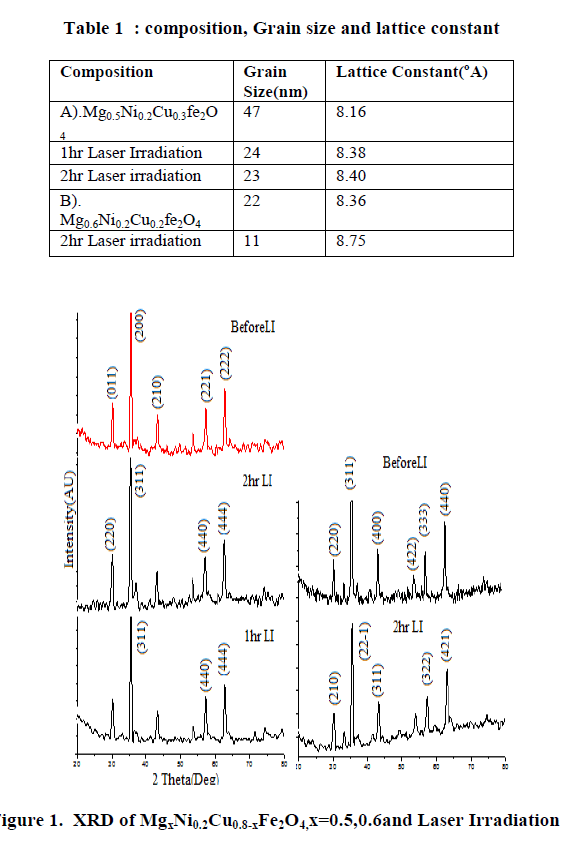
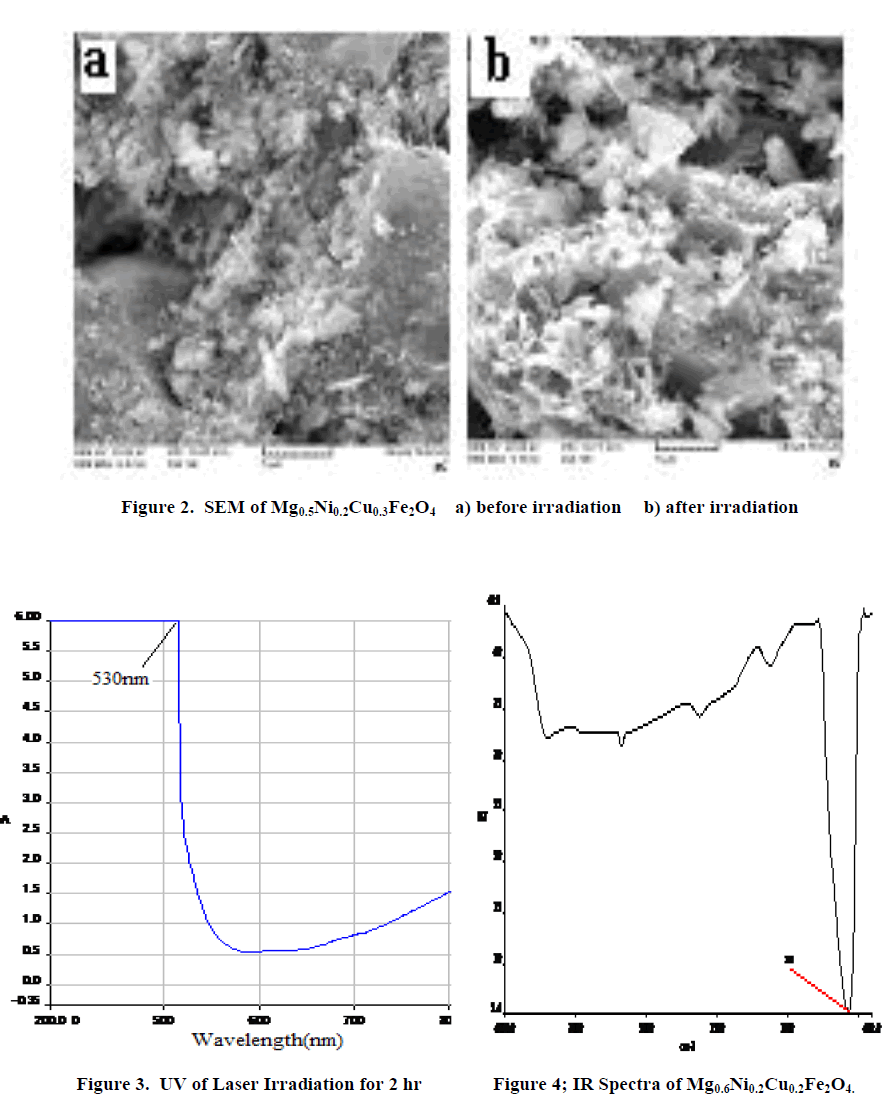
| β=full width at half maximum, θ= Bragg angle for the actual peak. The value of crystallite size is present in Table 1. Crystalline size decreases with increases in irradiation time. Fig 2 shows the typical scanning electron micrograph samples. From the SEM micrographs of the powders (Figure 2), the average grain size was ~47-11nm. The change in morphology can be related to the structure of samples. The UV-Vis spectrum show that the ferrite powder is identified by the charge transfer band at γmax = 530nm. Figure3. The absorption coefficient, α of the nanoparticles has been calculated using the fundamental relations9. To estimate the energy band gap for the samples, the graph of (αhυ)2 versus hυ has been plotted. The intercept of the line at α = 0 gives the value of energy band gap. The values of energy band gap 2hr irradiation samples is 3.5eV. Fig.4 illustrates the infrared absorption spectrum for (x=0.6) after laser irradiation. The first absorption band υ1 is related to intrinsic vibrations of octahedral groups. |
 |
 |
IV. CONCLUSION |
| From the above results we conclude that in MgxNi0.2Cu0.8-xFe2O4 where x=0.5,0.6 after laser irradiation average grain size of the material decreases as the percentage of the composition changes simultaneously there is change in the lattice constant of the material. From SEM result due to less resolution specific morphology was not determined. Infrared gives the sharp peak at 560cm-1 gives information about formation of ferrite material because it is characteristic band of ferrite. From UV spectroscopy the band gap energy of ferrite material is 3.5eV.The energy band gap shows the material is semiconductor. |
VI. ACKNOWLEDGMENTS |
| I thanks to the Department of Chemistry of Abasaheb Garware College, Pune, for providing their lab facilities for completion of research work. I thank the Department of C-MET Pune, IIT Powai, Mumbai and the Department of Physics, University of Pune for their kind co-operation and providing their instruments for the purpose of characterization also I would like thanks to UGC for providing the fund of this work. |
References |
|