ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Ibrahim M. El- Deen, Jehan A. Hasanen ?and Maha El-Ashery
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
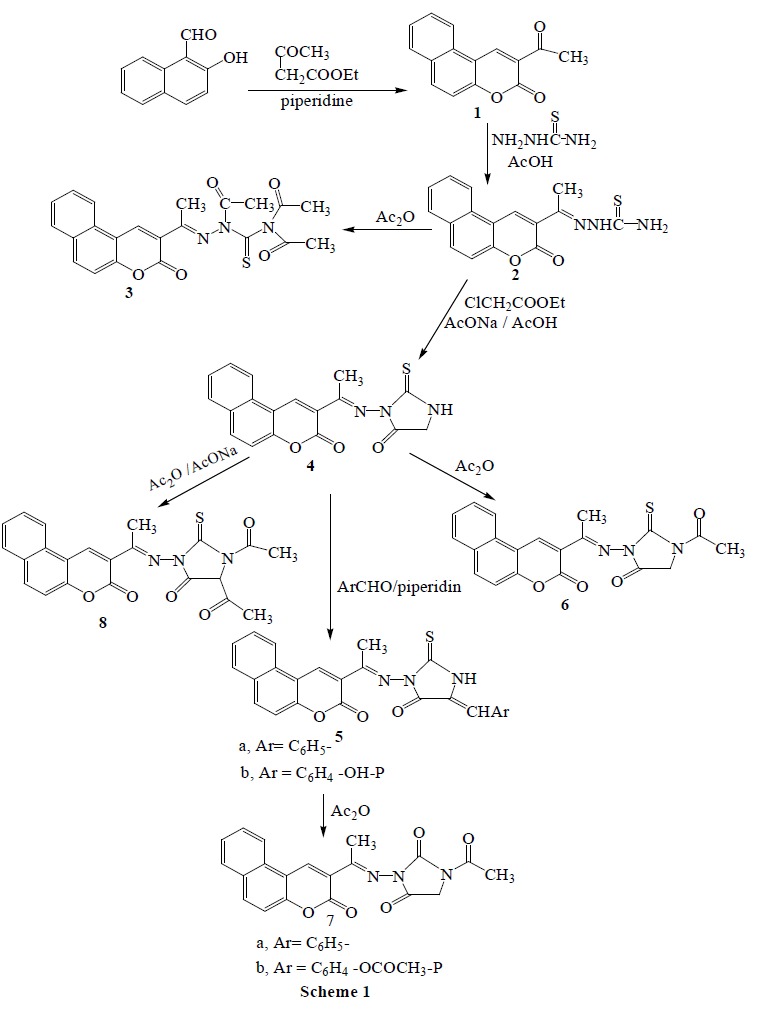
1-[(Bezocoumarin-3-ylethylidene)amino]-thiourea (2) was prepared via condensation of 3-acetyl benzocoumarin (1) with thiosemicarbazide, Reaction of compound 2 with acetic anhydride and ethyl chloroacetate in the presense of fused sodium acetate gives N-triacetyl derivatives (3) and 3-[(bezocoumarin-3-ylethylidene)amino]-4- oxo-imidazolidin-2-thione (4).Treatment of 4 with aromatic aldehydes yielding the corresponding 3-substituted-4-oxo- 5-arylidene-imidazolidin-2-thiones (5a, b). Acetylation of compounds 4 and 5 with acetic anhydride afforded the corresponding 1-acetyl-3-substituted-imidazolidin-2-thione (6) and 1-acetyl-3-substituted-5-arylidene-imidazolidine-2- thiones (7a, b), respectively. Acetylation of 4 with acetic anhydride in presence of fused sodium acetate gives 1, 5- diacetyl-3-substituted-imidazolidin-2-thione (8). The mass spectral fragmentation patterns of some prepared heterocyclic benzocoumarin derivatives have investigated in order to elucidate the structure of the synthesized compounds. The biological activity studies of heterocyclic benzocoumarins were carried out against antioxidant and antitumor activities.
Keywords |
| Synthesis, evalution, antitumor, heterocyclic, benzocoumarin. |
INTRODUCTION |
| The coumarins that were studied have divers biological properties1-5 and various effects on the different cellular system. A lot of biological parameters should be evaluated to increase our understanding of mechanisms by which these coumarins act. Coumarins have important effect in plant biochemistry and physiology, acting as antioxidant. Enzyme inhibitors and precursor of toxic substances. The coumarins have long been recognized to possess antiinflammatory, antioxidant, antiallergic, antithrombotic, antiviral, and anticarcinogentic activities. This paper describes the synthesis of some heterocyclic benzocoumarin derivatives from condensation of 3-acetyl benzocoumarin with thiosemicarbazide in acetic acid. The electron impact (EI) ionization mass spectral fragmentation of the prepared compounds is also described. These benzocoumarinyl heterocyclic compounds were investigated for antioxidant and antitumor activity. |
II-RESULTS AND DISCUSSIONS |
II-1)-CHEMISTRY |
| 3-Acetylbenzocoumarin (1) was prepared from 2-hydroxy-1-naphthaldehyde and ethyl acetoacetate according to a literature method. Condensation of compound 1 with thiosemicarbazide under reflux in acetic acid produced the 1- [(bezocoumarin-3-ylethylidene)amino]-thiourea (2). Acetylation of thiourea derivatives (2) with acetic anhydride under reflux led to the formation 1-acetyl, 1-[(benzocoumarin-3-ylethylidene)amino]-3, 3-diacetyl thiourea (3, Scheme 1). Treatment of thiourea derivatives (2) with ethyl chloroacetate in presence of fused sodium acetate in acetic acid under reflux, yielded the corresponding 3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-imidazolidin-2-thione(4). Condensation of imidazolidin-2-thione derivatives (4) with aromatic aldehyde (namely, benzaldehyde and 4- hydroxybenzaldehyde) in presence of piperidine under fusion led to the formation of 3-[(benzocoumarinylethylidene) amino]-4-oxo-5-arylidene-imidazolidin-2-thiones (5a, b). Acetylation of substituted imidazolidin-2-thione derivatives (4 and 5) with acetic anhydride under reflux led to the formation 1-acetyl-3-[(bezocoumarin-3-ylethylidene)amino]-4-oxo-imidazolidin-2-thione (6) and 1-acetyl-3- [(benzocoumarin-3-ylethylidene)amino]-4-oxo-5-arylidene-imidazolidin-2-thiones (7a, b), respectively. 1, 5-Diacetyl- 3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-imidazolidin-2-thion (8) was prepared via acetylation of imidazolidin-2-thione derivative (4) with acetic anhydride in presence of fused sodium acetate under reflux. |
 |
II-2)- MASS Spectroscopy |
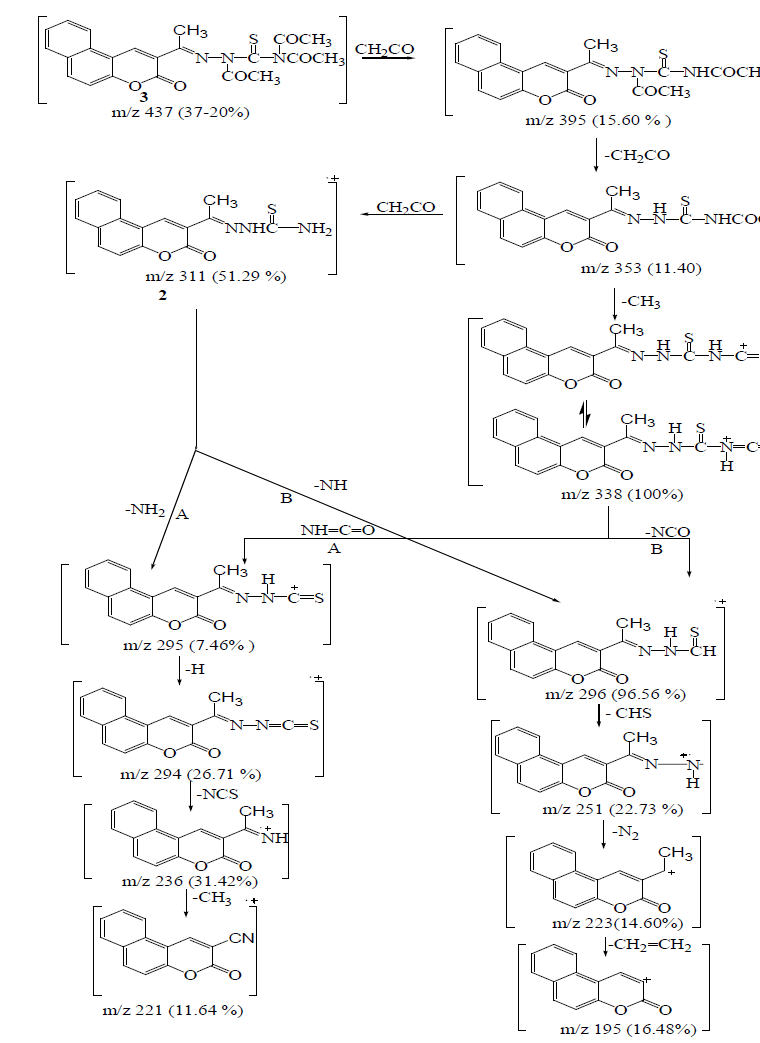
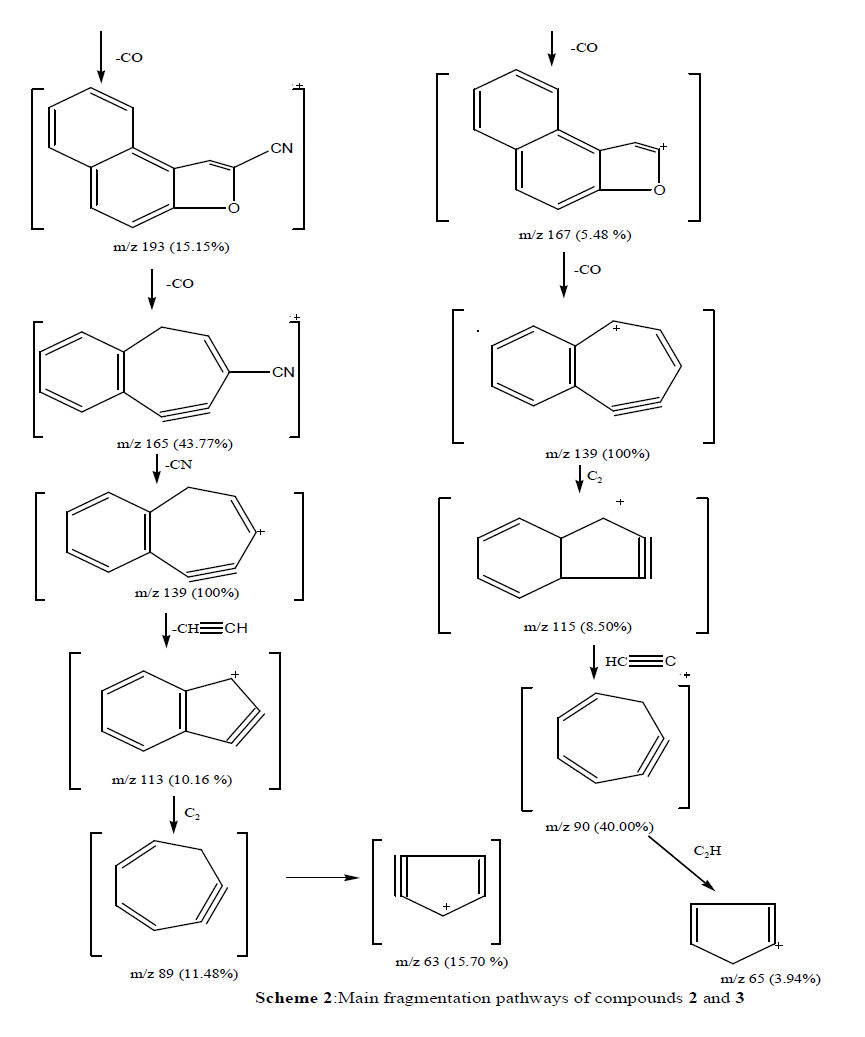
| The mass spectral decomposition modes8-10 of various heterocyclic compounds containing benzocoumarin substitutuents have been investigated and fragmentation pathways have been suggested. Compounds 2 and 3 The mass spectrum of compounds 2 and 3 showed intense molecular peaks at m/z 311 and 437, corresponding to the molecular formula C16H13N3O2S and C22H19N3O5S, respectively. The molecular ion peak of compound 2 (Scheme 2, Figure1) underwent fragmentation to produce a peak at m/z 296 by losing NH group. The loss of thioformyl group (CHS) from the ion with m/z 296 resulted in an ion at m/z 251. The ion at m/z 251 underwent loss of nitrogen and ethylene molecules to give peaks at m/z 223 and 195, respectively. Also, the molecular ion at m/z 311 underwent loss of amino group (NH2) to give peak at m/z 295, which further broke to give an ion at m/z 294. The ion of m/z 294 beoke to give an ion at m/z 236 which lost isothiocyanate group (NCS). Ion of m/z 236 fragmented to give an ion of m/z 221 which lost an methyl group (CH3). From the mass spectrum of compound 3 (Figure 2), it was concluded that the molecular ion at m/z 437. The ion of m/z 437 underwent fragmentation to produce a peak at m/z 395 by losing ketene molecule (CH2CO). The loss of ketene molecule (CH2CO) from the ion with m/z 395 gave an ion at m/z 353. The common peak at m/z 338 was also observed in this case which is attributed to an ion obtained by the loss methyl group (CH3) from the ion of m/z 353. The stable ion of m/z 338 underwent loss of isocyanate (NCO) and iminocarbonyl (NHCO) to give peaks at m/z 296 and 295, respectively. The fragment ions of m/z 296 and 295 further broke via pathway similar to compound 2 (Scheme 2) |
 |
 |
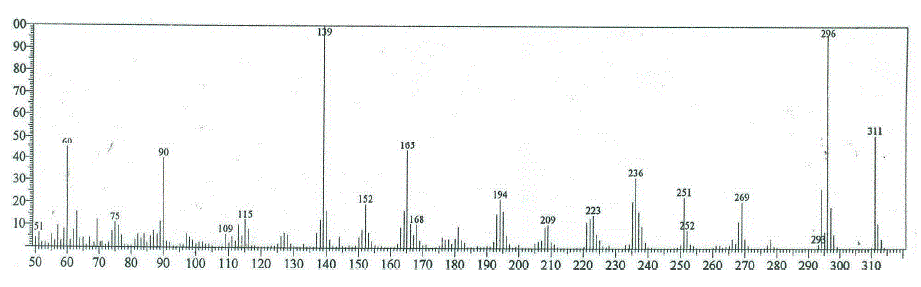 |
| Figure 1: EIMS spectrum of compound 2 |
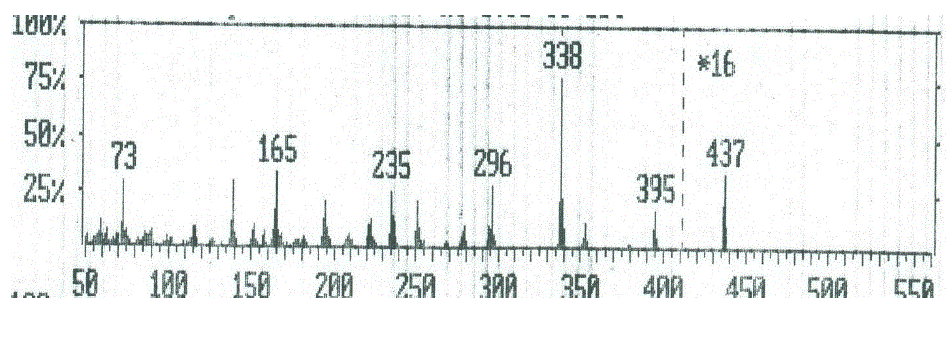 |
| Figure 2: EIMS spectrum of compound 3 |
| Compounds 4, 6 and 8 The mass spectra of the synthesized compounds 4, 6 and 8 showed intense molecular ion peaks at m/z at 351, 393 and 435, consistent with the molecular formula C18H13N3O3S , C20H15N3O4S and C22H17N3O5S, respectively. The molecular ion of compound 4 (m/z=351, Figure 3) underwent fragmentation (Scheme 3) to produce the peak at m/z 336 by losing methyl group. The fragment ion of m/z 336 fragmented via pathway A gave fragment of m/z 221, corresponding to the molecular ion of 3-cyano-benzocoumarin. The ion of m/z 221 broke to give an ion m/z at 193 which lost carbonyl group. Ion of m/z 193 fragmented to give an ion of m/z 265 which lost a carbonyl group(C=O). It further underwent loss of cyano group (CN), eyhylene molecule, C2 and ethylene molecule to give peaks at m/z 139, 113, 89 and 63, respectively. Also, the fragment ion of m/z 336 underwent fragmentation via pathway B to produce a peak at m/z 115, corresponding to the imidazolidin-2-thione radical cation. The loss of nitrogen atom (N) and imino group (NH) from the ion of m/z 115 gave a peaks at m/z 101 and m/z 86, respectively. |
 |
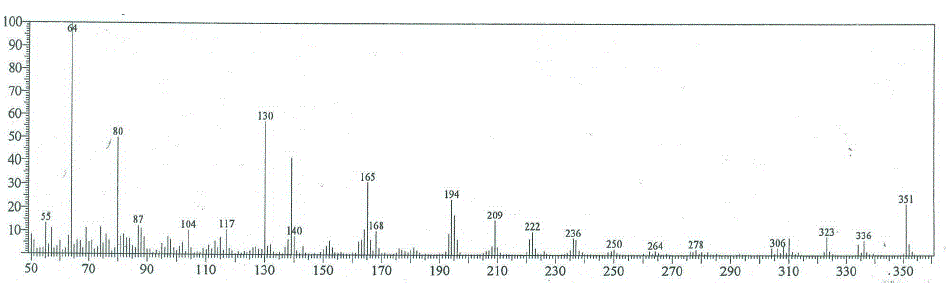 |
| Figure 3: EIMS spectrum of compound 4 |
| From the studies of mass spectra of the compound 8, it was found the molecular ion (m/z 435, Figure 5) for these compound had fragmented to a peak at m/z 393, corresponding to the molecular ion of compound 6 (Figure 4) by losing ketene molecule (CH2CO). The loss of ketene molecule (CH2CO) from the ion with m/z 393 gave an ion at m/z 351, corresponding to the molecular ion of compound 4. The ion of m/z 351 further broke via pathway similar to compound 4 (Scheme 3) |
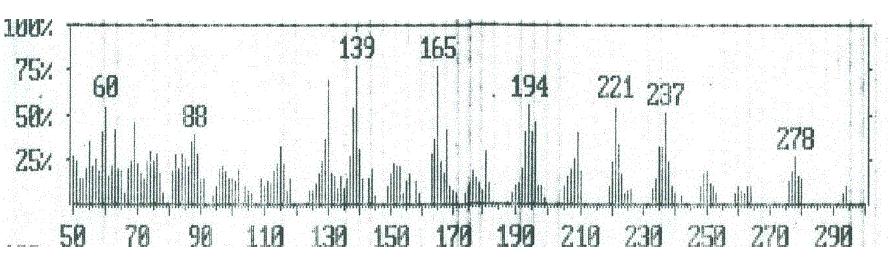 |
| Figure 4: EIMS spectrum of compound 6 |
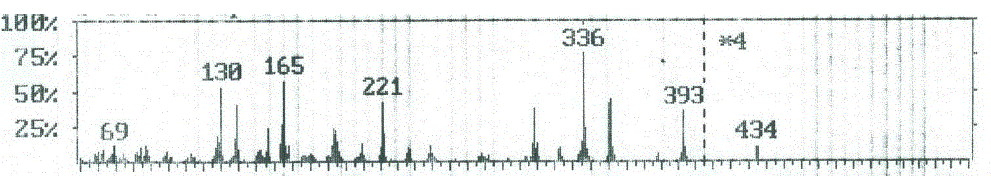 |
| Figure 5: EIMS spectrum of compound 8 |
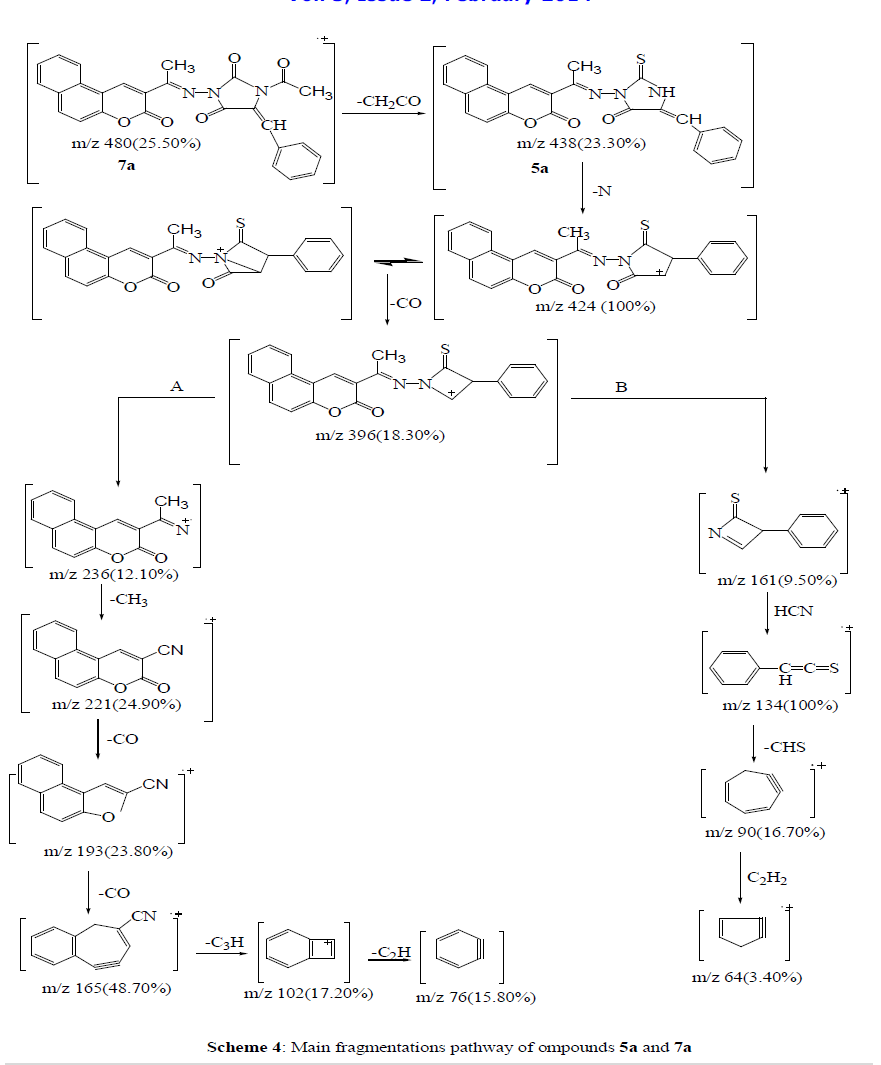
| Compounds 5a and 7a The molecular ion peak of compound 5a (Figure 6) was observed at m/z 438, corresponding to the molecular formula C25H16N3O3S. The loss of nitrogen atom from the molecular ion peak at m/z 438 gave a peak at m/z 424. The ion at m/z 424 underwent loss carbon monoxide (CO) to give peak at m/z 396. The fragmented ion m/z 396 underwent fragmentation via the pathway A to produce a peak at m/z 236, which further broke to give an ion at m/z 221. The loss of two carbonyl group (2CO) from the ion with m/z 221resulted in an ions at m/z 193 and m/z 165, respectively. It further underwent loss of cyano group (CN), C3H and C2H to give peaks at m/z 139, 102 and 76, respectively. Subsequently, the fragment ion m/z 396 fragmented via the pathway B by cleavage of (benzocoumarin- 3ylrthylidene)amino cation to give peak at m/z 161, which lost hydrogen cyanate (HCN) to give peak at 134. The loss of thiocarbonyl (CS) and ethylene molecule from the ion with m/z 134 resulted in an ions at m/z 90 and 64, respectively. The mass spectrum of compound 7a (Figure 7) showed the molecular ion m/z 480, corresponding to the molecular formula C27H18N3O4S. The loss of ketene molecule (CH2CO) from the molecular ion of 480 resulted in an ion at m/z 438, corresponding 5a. The fragment ion of m/z 438 which has further broke via pathway similar to compound 5a (Scheme 4). |
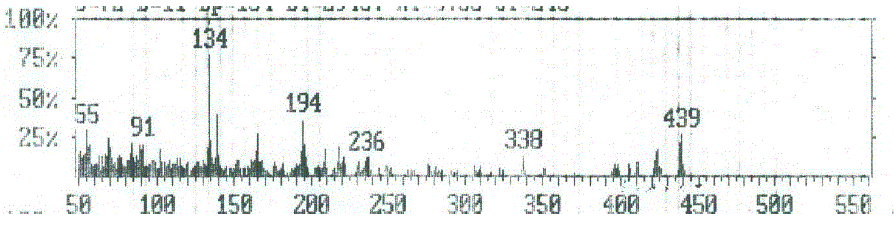 |
| Figure 6: EIMS spectrum of compound 5a |
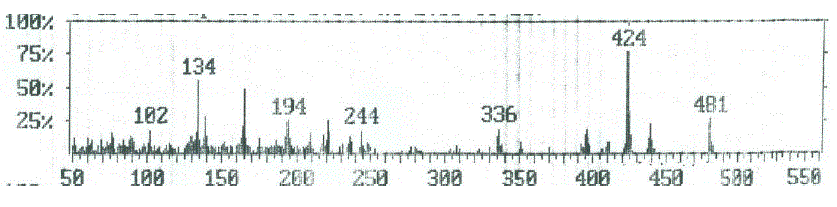 |
| Figure 7: EIMS spectrum of compound 7a |
 |
III) - Biologicalactivity |
III-1) - Antioxidant activity |
| The antioxidant of some prepared heterocyclic benzocoumarin derivatives (2, 4, 5, 6, 7a and 8a) were determined by the scavenging of synthetic radicals 2, 2-diphenyl-1-picrylhydrazyl (DPPH) in polar organic solvent11. A method solution of the test compounds was prepared. Absorbance measurements were recorded immediately with a Milton Roy spectronic 201 UV-visible spectrophotometer. The decrease in absorbance at 515 nm was determined continuously, with data being recorded at 1 min intervals until the absorbance stabilized (16 min). Tocotherol was used as a reference standard at the same concentration of used tested compounds. The absorbance of the DPPH radicals without antioxidant was also measured as control and 95% methanol was used as blank. All the determinations were performed in three replicates and averaged. % scavenging of the DPPH free radical was measured using the following equation:- DPPH radicals scavenging= [(Absorbance of control-Absorbance of tested samples)/(absorbance of control)]x100 Tested samples had been submitted for qualitative evaluation of the antioxidant activity. The provided samples had different antioxidant activity using DPPH radicals scavenging method as shown by the following table 1. |
 |
III-2) – Anticancer Activity |
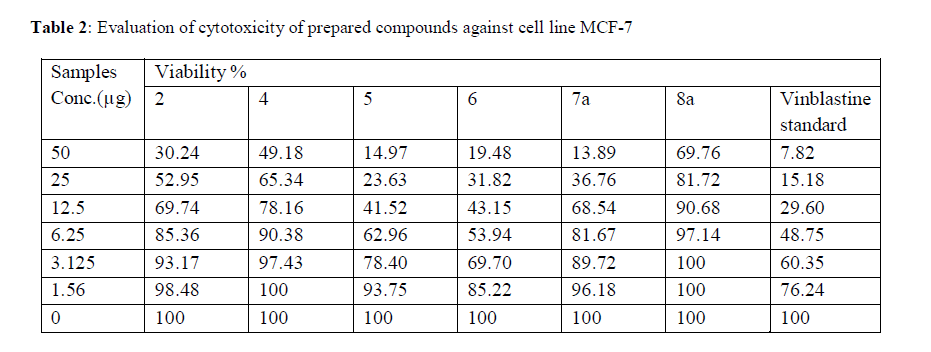
| Cytotoxic and antitumor activities of synthesized compounds (2-8a) were evaluated against cell lines MCF-7 and HepG-2 according to the method of mosamann12 and Vijayan et al13. The drug vinblastine was used as standard. Inhibitory activity against breast carcinoma cells (MCF-7 cell line) hepatocarcinoma cell line (HepG-2 cell line) was tested using different concentrations of the samples (50, 25, 12.5, 6.25, 3.125, and 1.56 μg), and cell viability (%) was determined by colorimetric method. The 50 % inhibitory concentration (IC50) of the MCF-7 cell line was calculated from Table 2 and Fig 8, 9. |
 |
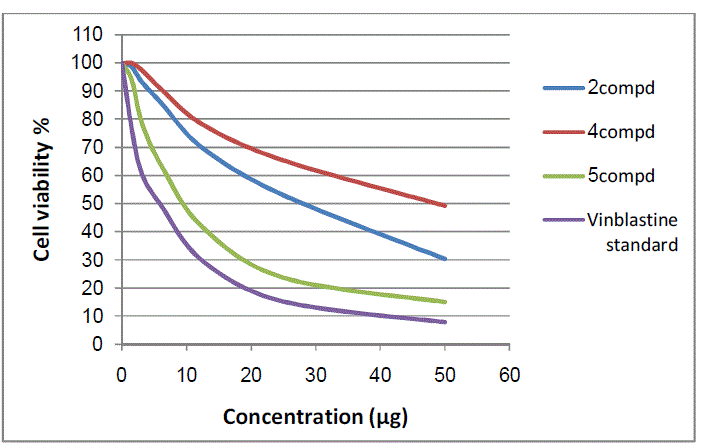 |
| Figure 8: The inhibitory activities against MCF-7 cell lines |
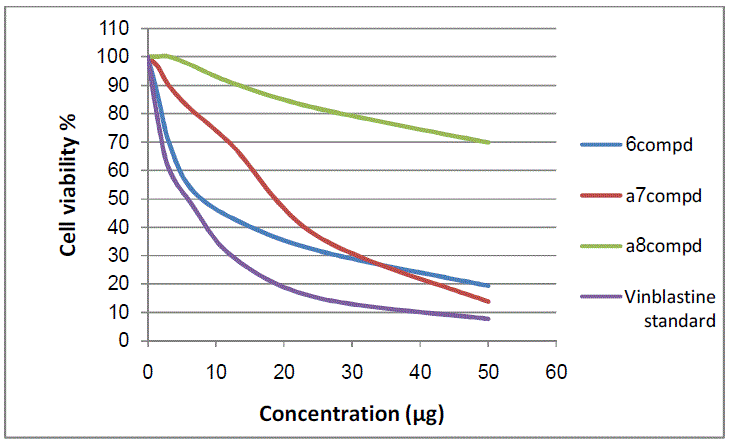 |
| Figure 9: The inhibitory activities against MCF-7 cell lines |
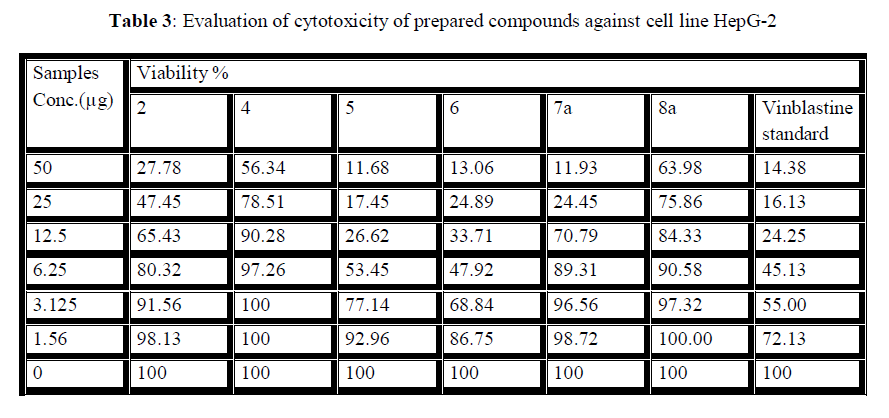
| The 50 % inhibitory concentration (IC50) of the HepG-2 cell line was calculated from Table 3 and Fig 10, 11. |
 |
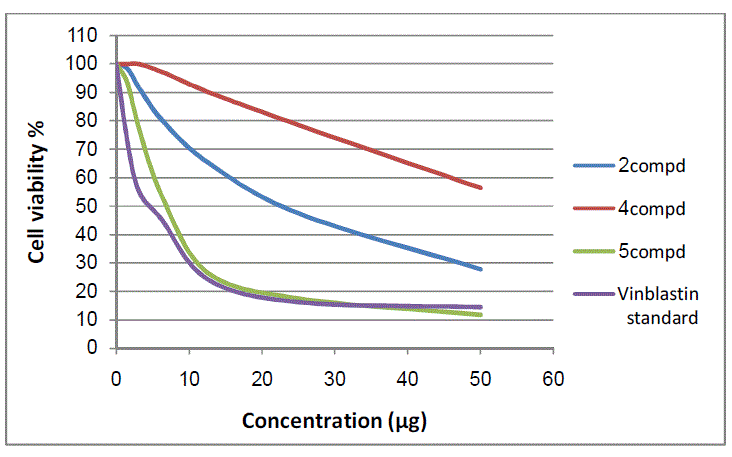 |
| Figure 10: The inhibitory activities against HepG-2 cell lines |
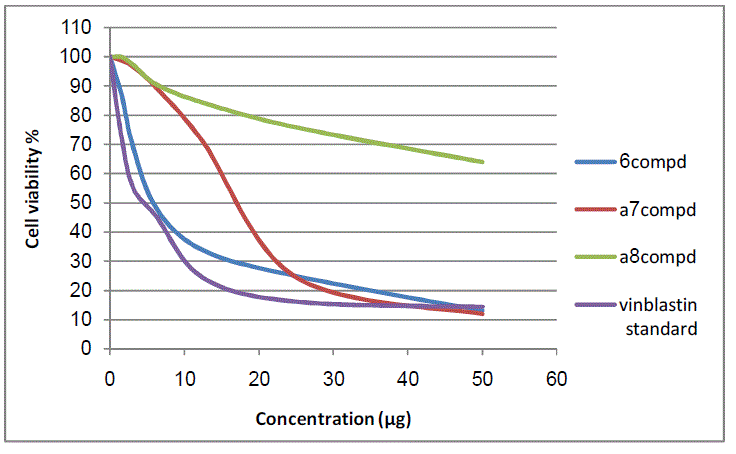 |
| Figure11: The inhibitory activities against HepG-2 cell lines |
| The results of 50 % inhibitory concentration (IC50) data are summarized in table 4. |
 |
| The IC50 value is the concentration that induces 50 % growth inhibition compared with untreated control cells. MCF-7: Human breast carcinoma cell line. HepG-2: Human hepatocellular carcinoma cell line Compounds 5 and 6 were found to be near active from standard antitumor drug vinblastine against MCF-7 and HepG-2 cell lines. In comparison with standard antitumor drug vinblastine, compounds 2 and 7a were found to be active against MCF-7 and HepG-2 cell lines, while compounds 4 and 8a were observed to be more weakly active against MCF-7 and HepG-2 cell lines |
IV)- EXPERMENTAL |
| Melting points were determined MEL-TEMP 11melting points apparatus and uncorrected infrared spectra were recorded on a perkin-Elmer 1420 spectrometer and a Biorad FTS7 (KBr). NMR spectra were recorded on a General Electric QE300 instrument and chemical shifts were given with respect TMS. Mass spectra were recorded on GC/MS with CI (chemical ionization) and a Hewlett Packard MS Engine Thermoscopy and ionization by electron impact at 70 eV the accelerating voltage was 6 KV, the temperature of the source was 200 ÃÂæC and the emission current 100 mA. Microanalyses were conducted using an elemental analyzer 1106. |
IV-1)-3- Acetylbenzocoumarin (1) |
| A mixture of 2-hydroxy-1-naphthaldehyde (0.01mol), ethyl acetoacetate (0.01 mol) and piperidine (1ml) was fused on a hot-plate for 2-3 min., then added ethanol (30 ml). The reaction mixture was heated under reflux for 2-3hr, then cooled and poured into ice-diluted hydrochloric acid. The resulting solid was filtered off, washed with water, dried and purified by recrystallization with ethanol to give 1 as yellow crystals, yield 78 %, m.p. 185 ÃÂæC. IR(KBr): 1735 (C=O of pyran), 1695 (C=O of ketene), 1605, 1585 (C=C), 1215, 1095 (C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.45 (s, 3H, CH3), 6.98-7.83 (m, 6H, Ar-H), 8.56 (s, 1H, H-pyran) ppm. MS: m/z (%) = 239 (M++1, 12.55), 238 (M+, 70.61), 224(16.08), 223(100), 196(4.93), 195(24.57), 169(1.99), 168(11.55), 167(2.13), 163(1.23), 153(1.68), 152(4.74), 151(5.87), 150(3.05), 149(2.53), 140(15.08), 139(80.86), 138(6.69), 137(5.22), 129(1.75), 127(3.15), 126(3.05), 125(2.39), 121(1.05), 119(1.49), 116(1.25), 115(5.07), 114(3.83), 113(11.10), 112(3.28), 102(1.12), 101(2.04), 100(1.34), 99(4.46), 98(5.83), 97(7.25), 96(2.88), 95(3.89), 91(1.72), 90(1.34), 89(10.35), 88(3.96), 87(8.07), 86(5.67), 85(6.89), 84(3.49), 83(6.60), 82(3.58), 81(5.90), 79(5.29), 78(1.44), 77(3.09), 76(4.24), 75(5.63), 74(4.88), 73(4.10), 71(8.43), 70(4.78), 69(11.96), 68(2.38), 67(3.86), 65(3.31), 64(4.97), 63(13.94), 62(5.11), 60(4.44). Anal. Calcd. For C15H10O3: C, 75.63; H, 4.20. Found: C, 75.56; H, 4.09. IV-2)-1-[(benzocoumarin-3-ylethyliden)amino]-thiourea (2) A mixture of 1(0.01 mol) and thiosemicarbazide (0.01 mol) in acetic acid (30 ml) was heated under reflux for 2hr, then cooled and poured into water. The solid obtained was filtered off. Washed with water, dried and purified by acetic acid to give 2 as yellow crystals, yield 87%.m.p. 251 ÃÂæC. IR(KBr): 3395, 3185(NH2), 3225(NH), 1732(CO of pyran), 1336(C=S), 1225, 1093(C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.24(s, 3H, CH3), 7.31-8.17(m, 6H, Ar-H), 8.61(s, 1H, H-pyran), 9.01(S, 2H, NH2), 10.40(S, 1H, NH)ppm. MS: m/z (%) = 312(M++1, 11.15), 311(M+, 51.29), 298(6.32), 297(18.37), 296(96.56), 295(7.46), 294(26.71), 278(4.13), 277(1.67), 270(4.20), 269(20.63), 268(11.89), 267(2.32), 266(4.04), 253(2.02), 252(8.03), 251(22.73), 239(2.79), 238(9.95), 237(16.22), 236(31.42), 235(20.79), 224(6.23), 223(14.60), 222(13.07), 221(11.64), 210(2.99), 209(10.35), 208(9.65), 207(3.46), 206(3.05), 196(5.50), 195(16.48), 194(21.51), 193(15.15), 192(2.95), 182(3.42), 181(9.48), 180(4.12), 178(3.89), 177(3.64), 176(4.50), 169(3.22), 168(10.34), 167(5.78), 166(10.73), 165(43.77), 164(16.66), 163(9.05), 153(6.59), 152(19.39), 151(7.96), 150(4.35), 140(16.61), 139(100), 138(12.27), 137(6.50), 128(5.76), 127(6.61), 126(5.08), 116(8.34), 115(12.31), 114(5.10), 113(10.16), 111(4.92), 109(5.88), 103(1.56), 102(2.48), 101(2.26), 99(3.10), 98(4.41), 97(6.06), 91(2.55), 90(40.00), 89(5.88), 87(7.54), 86(4.81), 82(5.27), 81(3.37), 77(5.26), 76(9.55), 75(11.02), 74(7.18), 71(2.28), 69(12.28), 68(1.85), 67(4.09), 65(3.94), 64(3.68), 63(15.76), 62(7.61), 60(44.74), 59(8.08). Anal. Calcd. For C16H13N3O2S: C, 61.74; H, 4.18; N, 13.50; S, 10.29. Found: C, 61.63; H, 4.04; N, 13.39; S, 10.18. IV-3)-1-Acetyl, 1-[(benzocoumarin-3-ylethylidene)amino]-3, 3-diacetyl thiourea (3) A solution of 2 (0.01 mole) in acetic anhydride (20 ml) was heated under reflux for 1-1.5hr, then cooled and poured into ice-water. The resulting product was filtered off, washed with water, dried and purified by recrystallization from benzene to give 3 as pale yellow crystals, yield 53%, m.p.136 ÃÂæC. IR(KBr): 1729(CO of pyrane, 1701-1687)(CO of ketene), 1631(C=N), 1605, 1589(C=C), 1321(C=S), 1115,1087(C-O)cm-1. 1H-NMR (DMSO-d6): δ 2.24(s, 3H, CH3), 2.35-2.41(s, 9H, 3XCOCH3), 7.31-8.11(m. 6H.Ar-H), 8.71(s, 1H, H-pyrane)ppm. MS: m/z (%) = 438(M++1, 16.20), 437(M+, 41.20), 436(M+-1, 22.10), 396(4.10), 395(16.90), 394(8.50), 354(2.60), 353(11.40), 352(4.20), 351(1.80), 340(8.70), 339(21.80), 338(100), 337(21.30), 311(0.70), 298(2.30), 297(6.30), 296(27.20), 295(7.80), 294(9.90), 293(3.50), 280(2.30), 279(9.40), 278(7.00), 277(3.80), 269(2.10), 268(2.90), 267(1.90), 253(2.60), 252(8.90), 251(20.40), 250(7.10), 238(3.70), 23710.60), 236(14.40), 235(25.20), 234(6.30), 224(4.30), 223(12.60). 222(9.90), 221(10.20), 220(4.70), 208(4.10), 207(3.10), 197(2.40), 196(4.30), 195(8.40), 194(20.10), 193(12.80), 192(3.80), 182(2.90), 181(4.90), 180(3.60), 178(2.70), 177(3.30), 176(3.40), 176(3.40), 168(6.10), 166(7.70), 165(33.30), 164(16.10), 163(7.40), 162(3.40), 153(3.10), 152(10.40), 151(7.20), 150(2.40), 141(2.50), 140(5.70), 139(28.80), 138(10.80), 137(2.50), 117(2.70), 116(8.30) 115(8.30), 114(2.80), 113(3.60), 102(3.00), 101(3.40), 100(1.80), 99(4.80), 98(2.20), 90(6.70), 89(6.00), 88(4.10), 87(4.10), 86(5.20), 77(2.10), 76(3.10), 75(7.30), 74(5.20), 73(28.40), 72(9.30), 69(4.60), 67(2.50), 65(1.40), 63(6.70), 62(4.90), 60(4.90), 59(11.00), 57(4.90), 55(3.30), 51(4.50), 50(3.30), Anal.Calcd. forC22H19N3O5S: C, 60.41; H, 4.35; N, 9.61; S, 7.32 Found: C, 60.27; H, 4.25; N, 9.5; S, 7.18. IV-4)-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-imidazolidin-2-thione (4) A mixture of 2 (0.01 mol) and ethyl chloroacetate (0.01 mol) in acetic acid (50 ml) in the presence of fused sodium acetate (0.03 mol) was heated under reflux for 2-3hr, then cooled and poured into water. The resulting solid was filtered off, washed with water, dried and purified by recrystallization from acetic acid to give 4 as yellow crystals, yield 67%, m.p.285 ÃÂæC. IR(KBr): 3328(NH), 1732(CO of pyrane, 1695(CO of imidazolidine), 1631(C=N), 1605, 1595(C=C), 1332(C=S), 1135,1087(C-O)cm-1. 1H-NMR (DMSO-d6): δ 2.21( s, 3H, CH3), 3.63(s, 2H, NCH2CO), 7.31- 8.10(m, 6H, Ar-H), 8.51(s, 1H, H-pyrane), 10.60(s, 1H, NH)ppm. MS: m/z (%) = 352(M++1, 5.03), 351(M+, 21.90), 336(6.20), 335(1.37), 334(4.89), 324(1.82), 323(7.85), 322(1.57), 313(1.22), 311(1.69), 310(7.28), 308(4.30), 306(3.02), 282(1.48), 280(1.51), 278(2.31), 277(1.45), 276(1.60), 265(1.09), 264(1.73), 262(1.68), 250(2.51), 249(1.78), 248(1.37), 239(1.24), 238(2.13), 237(6.60), 236(7.18), 235(2.18), 2232.89), 222(10.08), 221(6.76), 220(1.37), 210(3.45), 209(14.89), 208(3.85), 207(2.00), 197(1.19), 196(6.50), 195(17.18), 194(23.76), 193(9.22), 182(1.93), 181(3.31), 180(1.94), 179(1.21), 178(1.72), 177(2.24), 176(2.84), 169(2.82), 168(10.20), 167(1.89), 166(6.33), 165(31.03), 164(11.01), 163(6.46), 162(5.66), 153(3.14), 152(5.91), 151(3.86), 150(2.40), 140(7.97), 139(41.17), 138(6.46), 137(3.45), 132(4.48), 131(3.85), 130(57.19), 129(2.37), 128(2.48), 127(3.59), 126(3.09), 119(1.52), 118(2.77), 117(10.81), 115(7.04), 114(3.14), 113(5.74), 112(2.41), 111(4.21), 110(2.06), 109(2.66), 105(2.99), 104(10.47), 102(5.16), 101(3.59), 100(1.59), 99(2.86), 98(6.35), 97(7.71), 96(3.01), 95(4.76), 91(2.14), 90(2.20), 89(7.57), 88(11.18), 87(12.30), 86(2.96), 85(3.77), 84(6.55), 83(6.90), 82(8.68), 81(7.70), 80(50.05), 79(2.78), 77(5.73), 76(6.63), 75(4.75), 74(11.78), 73(3.13), 71(5.78), 70(5.16), 69(11.33), 68(2.71), 65(3.93), 64(100), 63(7.91), 60(5.50). Anal.Calcd: for C18H13N3O3S: C, 61.54; H, 3.70; N, 11.96; S, 9.12. Found: C, 61.32; H, 3.68; N, 11.79; S, 9.02. IV-5)-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-5-arylidene-imidazolidin-2-thiones (5a, b) A mixture of 4 (0.01 mol), aromatic aldehydes (such as benzaldehyde and 4-hydroxy benzaldehyde) (0.01 mol) and piperidine (1 ml) was fused on a hot plate at 120-125 ÃÂæC for 1 hr. The reaction mixture was cooled and acidified with diluted hydrockloric acid (2N). The crude product was filtered off, washed with water, dried and purified by recrystallization from ethanol to give 5. 3-[(benzocoumarin-3-ylethylidene) amino]-4-oxo-5-benzylidene-imidazolidin-2-thione (5a) as yellow crystals, yield 63%, m.p. 245 ÃÂæC. IR(KBr): 3237(NH), 1732, 1689(C=O), 1625(C=N), 1605, 1589(C=C), 1321(C=S), 1215, 1093(C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.23(S, 3H, CH3), 6.95-8.01(M, 12H, Ar-H and H-olefine), 8.46(S, 1H, H-pyrane), 10.77(S, 1H, NH)ppm. MS: m/z (%) = 439(M++1, 23.20), 438(M+, 22.30), 423(6.20), 422(5.20), 421(3.10), 412(6.20), 411(5.30), 338(6.40), 325(2.40), 323(3.70), 311(2.00), 310(5.40), 309(2.04), 307(3.40), 306(6.10), 283(3.10), 282(2.00), 281(5.80), 277(5.10), 276(6.50), 266(2.70), 265(2.70), 263(3.40), 252(3.70), 251(2.70), 250(5.10), 249(5.10), 239(2.70), 237(10.90), 236(10.90), 235(8.20), 231(8.50), 230(5.40), 222(6.50), 221(11.60), 220(7.80), 219(6.80), 218(17.70), 209(16.70), 208(7.80), 205(4.10), 204(5.80), 197(3.70), 196(11.20), 195(14.40), 194(35.00), 193(15.60), 191(5.40), 190(6.50), 182(3.90), 181(6.80), 180(4.80), 178(3.40), 177(5.40), 176(8.20), 168(9.90), 167(8.20), 166(8.80), 165(25.20), 164(15.30), 163(9.50), 154(4.10), 153(10.20), 152(9.90), 151(6.80), 141(6.10), 140(11.90), 139(39.10), 138(11.20), 136(10.50), 135(22.40), 134(100), 133(16.30), 130(10.20), 129(6.10), 127(7.80), 126(8.20), 125(5.80), 119(6.80), 117(3.70), 116(5.10), 115(11.60), 113(10.90), 112(6.80), 111(6.80), 110(7.10), 108(9.20), 106(5.10), 105(8.20), 103(8.50), 102(16.00), 99(7.80), 98(10.20), 97(10.20), 96(7.50), 91(20.10), 90(16.70), 89(19.40), 88(10.20), 87(12.20), 78(7.10), 77(10.90), 76(11.90), 75(11.60), 73(9.50), 71(10.50), 70(18.40), 69(23.10), 68(12.20), 67(13.30), 65(12.20), 63(12.60), 60(6.80), 59(5.80), 58(7.50), 57(20.10), 56(18.00), 55(28.80), 54(14.30), 53(12.60), 51(14.60), 50(13.50). Anal.Calcd. for C25H16N3O3S: C, 68.49; H, 3.65; N, 9.59; S, 7.30. Found: C, 68.29; H, 3.46; N, 9.41; S, 7.23. 3-[(benzocoumarin-3-ylethylidene) amino]-4-oxo-5-(P-hydroxy)benzylidene-imidazolidin-2-thione (5b) as yellow crystals, yield 64%, m.p. 233 ÃÂæC. IR(KBr): 3420-2985(br-OH), 3287(NH), 1730, 1689(C=O), 1629(C=N), 1610, 1589(C=C), 1321(C=S), 1171, 1089(C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.31(S, 3H, CH3), 6.93-8.12(M, 11H, Ar-H and H-olefine), 8.46(S, 1H, H-pyrane), 10.62(S, 1H, NH), 11.30(S, 1H, OH)ppm. MS: m/z (%) = 455(M++1, 11.80), 454(M+, 21.35), 418(1.00), 417(4.30), 392(3.10), 391(5.70), 390(13.80), 389(11.80), 352(9.40), 351(44.30), 350(21.90), 337(4.10), 336(10.60), 335(6.50), 334(11.60), 324(5.10), 323(15.00), 322(7.10), 311(3.50), 310(3.80), 309(5.90), 308(6.90), 306(6.70), 304(6.50), 278(4.30), 277(3.50), 276(4.10), 275(4.10), 266(2.00), 265(1.20), 262(2.80), 252(2.40), 251(3.10), 250(6.50), 249(4.70), 248(4.10), 237(14.60), 236(15.00), 235(8.10), 231(6.50), 230(5.90), 229(8.50), 223(5.50), 222(23.00), 221(17.90), 220(6.10), 210(8.50), 209(31.90), 208(11.90), 207(7.50), 196(16.10), 195(36.60), 194(46.10), 193(20.30), 192(10.00), 188(4.90), 187(4.70), 181(7.10), 178(6.10), 177(6.90), 176(6.30), 168(24.40), 167(8.50), 166(11.80), 165(46.50), 164(25.40), 163(15.00), 162(10.00), 153(6.50), 152(14.90), 151(8.90), 150(9.60), 140(16.90), 139(65.50), 138(32.90), 132(16.70), 131(12.00), 130(100), 129(18.70), 117(5.70), 116(8.50), 115(18.50), 114(9.80), 113(10.20), 111(6.70), 105(7.30), 104(8.90), 102(12.00), 101(7.10), 98(7.10), 97(9.30), 91(7.90), 90(8.90), 89(12.40), 88(22.60), 87(25.40), 86(12.00), 84(15.00), 82(12.80), 78(5.90), 77(13.80), 76(15.00), 75(11.80), 74(11.40), 70(20.10), 69(20.50), 65(8.70), 64(7.30), 63(21.10), 62(11.80), 60(6.90), 59(10.20), 57(10.00), 56(9.80), 55(15.00), 53(7.50), 51(13.00), 50(11.80). Anal.Calcd for C25H16N3O4S: C, 66.08; H, 3.52; N, 9.25; S, 7.05. Found: C, 65.89; H, 3.43; N, 9.07; S, 6.91. IV-6)-1-Acetyl-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-imidazolidin-2-thione (6) 1-Acetyl-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-5-arylidene-imidazolidin-2-thiones (7a, b) A solution of 4 and 5 (0.01 mol) in acetic anhydride (25 ml) was heated under reflux for 2 hr, then cooled and poured onto ice-water. The resulting product was filtered off, washed with water, dried and purified by recrystallization from benzene to give 6 and 7. 1-Acetyl-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-imidazolidin-2-thione (6) as pale yellow crystals, yield 57%, m.p. 178 ÃÂæC. IR(KBr): 1733(CO of pyrane), 1705-1689(CO), 1632(C=N), 1609, 1592(C=C), 1321(C=S), 1225, 1085(C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.21(S, 3H, CH3), 2.31(S, 3H, COCH3), 3.56(S, 2H, NCH2CO), 7.21-8.01(M, 6H, Ar-H), 8.43(S, 1H, H-pyrane)ppm. MS: m/z (%) = 394(M++1, 22.90), 393(M+,22.90), 378, 337(12.50), 336(35.90), 335(18.80), 334(14.60), 323(18.80), 322(7.30), 310(18.80), 308(22.90), 307(11.50), 304(10.40), 280(14.60), 279(15.60), 278(26.00), 277(17.70), 276(12.50), 268(9.40), 260(9.40), 252(9.40), 251(11.50), 250(17.70), 249(16.70), 298(10.40), 239(10.40), 238(24.00), 237(51.00), 236(32.30), 235(31.30), 234(13.50), 223(16.70), 222(33.30), 221(59.20), 220(24.40), 219(10.40), 210(17.70), 209(39.50), 208(25.00), 207(19.80), 197(10.40), 196(44.80), 195(40.50), 194(55.20), 193(40.60), 192(19.80), 191(11.50), 182(11.50), 181(29.20), 179(11.50), 178(14.60), 177(17.70), 176(11.50), 169(10.40), 168(41.70), 167(16.70), 166(22.90), 165(85.40), 164(36.50), 163(27.10), 155(12.50), 153(20.80), 152(20.80), 151(21.90), 150(16.70), 143(14.60), 141(13.50), 140(30.20), 139(100), 138(53.10), 137(26.00), 132(15.60), 131(16.70), 130(68.80), 129(35.40), 128(22.90), 118(13.50), 116(21.90), 115(31.20), 114(21.90), 113(17.70), 112(11.50), 111(12.50), 105(7.30), 104(10.40), 102(18.80), 101(12.50), 100(14.60), 98(17.70), 97(20.80), 96(19.80), 91(13.50), 90(13.50), 89(28.10), 88(38.50), 87(34.40), 86(20.80), 77(16.70), 76(28.10), 75(24.00), 74(29.20), 73(24.00), 70(21.90), 69(45.80), 68(24.00), 67(19.80), 65(17.70), 64(19.80), 63(41.70), 62(20.80), 60(54.20), 59(39.60), 57(25.00), 56(20.80), 55(34.90), 51(22.90), 50(26.00). Anal. Calcd for C20H15N3O4S: C, 61.07; H, 3.82; N, 10.69; S, 8.14. Found: C, 60.93; H, 3.67; N, 10.73; S, 8.02 1-Acetyl-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-5-benzylidene-imidazolidin-2-thione (7a) as pale yellow crystals, yield 56%, m.p. 163 ÃÂæC. IR(KBr): 1732 (CO of pyrane), 1705-1693(CO of ketene and imino), 1629(C=N), 1605, 1588(C=C), 1321(C=S), 1215, 1093(C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.21(s, 3H, CH3), 2.35(s, 3H, CO CH3), 6.95-8.13(m, 12H, Ar-H and olefinic proton), 8.51(s, 1H, H-pyrane) ppm. MS: m/z (%) = 481(M++1, 23.20), 480(M+, 22.30), 439(15.20), 438(20.10), 437(10.20), 425(31.10), 424(100), 423(87.90), 422(7.00), 411(8.10), 410(8.10), 396(18.30), 395(14.30), 338(7.30), 337(5.10), 336(18.70), 310(2.80), 308(5.50), 251(2.8), 280(3.70), 277(4.40), 253(2.60), 250(4.80), 249(7.00), 248(8.80), 245(5.10), 244(17.20), 237(8.40), 236(12.10), 235(7.70), 222(16.10), 221(24.90), 220(9.90), 218(13.60), 217(6.60), 210(6.20), 209(15.80), 208(8.40), 196(6.20), 195(11.70), 194(24.90), 193(23.80), 192(12.50), 188(5.50), 187(5.90), 186(9.90), 185(6.20), 176(4.40), 175(10.60), 174(4.80), 168(6.20), 167(6.20), 166(11.00), 165(48.70), 164(20.90), 163(10.60), 152(8.80), 151(7.30), 150(5.90), 140(12.10), 139(28.20), 138(10.60), 135(10.50), 134(55.30), 133(15.40), 132(9.50), 130(12.50), 129(12.80), 117(3.30), 116(6.20), 115(7.30), 105(5.50), 103(4.40), 102(17.20), 101(7.00), 98(7.70), 97(4.40), 91(8.40), 90(11.40), 89(13.20), 77(11.40), 76(15.80), 75(7.00), 74(6.20), 73(8.10), 64(1.80), 63(10.30), 62(7.70), 61(5.90), 60(11.40), 51(10.60), 50(5.10), Anal. Calcd. For C27H18N3O4S: C, 67.50; H, 3.75; N, 8.75; S, 6.66. Found: C, 67.33; H, 3.67; N, 8.58; S, 6.46. 1-Acetyl-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxo-5-(P-acetoxybenzylidene)-imidazolidin-2-thione (7b) as pale yellow crystals, yield 61%, m.p. 175 ÃÂæC. IR(KBr): 1745-1732 (CO of ester and pyrane), 1703-1689(CO of imino and ketene), 1631(C=N), 1608, 1595(C=C), 1321(C=S), 1185, 1073(C-O) cm-1. 1H-NMR (DMSO-d6): δ 2.25(s, 3H, CH3), 2.34-2.36(s, br, 6H, 2x CO CH3), 6.98-8.10(m, 11H, Ar-H and olefinic proton), 8.53(s, 1H, H-pyrane) ppm. MS: m/z (%) = 539(26; 10), 538(M+, 21.70), 508(26.10), 412(21.70), 411(13.00), 356(21.70), 355(21.70), 354(26.10), 323(26.10), 322(13.00), 318(21.70), 317(21.70), 247(13.00), 246(26.10), 245(26.10), 244(21.70), 238(13.00), 237(30.40), 236(13.00), 235(30.40), 228(21.70), 222(39.10), 221(17.40), 213(34.80), 212(26.10), 211(26.10), 210(43.50), 209(26.10), 208(17.90), 206(21.70), 205(17.40), 197(30.40), 193(39.10), 189(30.40), 183(21.70), 178(4.30), 176(30.40), 174(26.10), 156(8.7), 164(43.50), 160(30.40), 158(26.10), 152(39.10), 151(17.40), 145(26.10), 141(26.10), 140(39.10), 139(30.40), 130(26.10), 129(34.80), 128(30.40), 120(21.70), 118(26.10), 117(39.10), 116(8.70), 115(17.40), 114(17.40), 108(34.80), 106(17.40), 102(43.50), 101(43.50), 100(13.00), 98(100), 97(39.10), 96(26.10), 94(42.50), 91(52.20), 90(56.50), 89(13.00), 87(25.10), 86(52.20), 85(13.00), 80(43.50), 79(21.70), 78(52.20), 77(13.00), 74(60.90), 73(69.50), 68(13.70), 67(30.40), 64(78.30), 63(30.40), 61(17.40), 60(78.30), 59(30.40), 52(30.40), 51(69.60). Anal. Calcd for C29H20N3O6S: C, 64.68; H, 3.72; N, 7.81; S, 5.95. Found: c, 64.46; h, 3.53; n, 7.66; s, 5.77 |
IV-7)-1,5-DIacetyl-3-[(benzocoumarin-3-ylethylidene)amino]-4-oxoimidazolidin- 2-thione (8) |
| A mixture of 4(0.01 mol) and fused sodium acetate (0.05mol) in acetic anhydride (25 ml) was heated under reflux for 2-3 hr, then cooled and poured into water. The resulting solid was filtered off, washed with hot water, dried and purified with benzene to give 8 as colorless crystals, yield 51%, m.p. 156 ÃÂæC.1H-NMR (DMSO-d6): δ 2.13(s, 3H, CH3), 2.30-2.36(br. S., , 6H, 2x CO CH3), 7.5-8.24(m, 6H, Ar-H ), 8.76(s, 1H, H-pyrane), 9.0(s, 1H, NCH(CO)2) ppm. IR(KBr): 1731(pyrane), 1707-1689(br. CO of imino and ketene), 1631(C=N), 1611, 1585(C=C), 1321(C=S), 1213, 1121, 1087(C-O) cm-1. MS: m/z (%) = 436(M++1, 2.10), 435(M+, 2.30), 434(M+-1, 2.60), 395(3.00), 394(10.5), 393(35.20), 378(5.70), 377(2.90), 353(4.20), 352(9.30), 351(43.80), 350(40.70), 338(8.60), 337(23.40), 336(100), 335(14.00), 327(9.7), 322(8.00), 310(13.00), 308(37.00), 307(12.80), 282(4.70), 281(1.90), 279(2.60), 278(5.30), 277(3.00), 263(1.60), 251(2.90), 250(6.30), 249(10.60), 248(6.20), 222(14.50), 221(41.80), 220(9.50), 210(4.70), 209(12.50), 208(6.30), 196(7.00), 195(13.80), 194(22.20), 193(23.90), 192(11.50), 181(4.70), 180(5.20), 168(11.70), 167(5.00), 166(11.20), 165(57.00), 164(25.80), 163(13.20), 152(8.30), 151(6.60), 140(6.90), 139(39.50), 138(16.60), 131(3.70), 130(53.00), 129(14.20), 128(18.30), 127(9.50), 118(2.10), 117(1.30), 111(3.60), 102(2.40), 101(2.90), 99(6.40), 98(3.90), 89(4.90), 88(8.30), 87(10.70), 76(2.30), 70(4.60), 69(11.50), 68(5.00), 67(4.60), 65(2.00), 63(8.20), 62(5.40), 60(6.90), 59(9.80), 58(5.90), 51(2.10), 50(2.60). Anal. Calcd for C22H17N3O5S: C, 60.69; H, 3.90; N, 9.65; S, 7.35. Found: C, 60.79; H, 3.76; N, 9.41; S, 7.17. |
References |
|