ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Rama Rao.K1, Rambabu.R2, Raghu Babu.K3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
direct and simple oxidimetric titrimetric method for the estimation of ascorbic acid, hydrazine, sodium sulphite, thiosulphate and vanadium(II) with chloramine-T in EDTA medium is reported both potentiometric and visual end point detection. Indicators used are cacotheline, p-anisidine, methylene blue, erioglucine-A, thionine and starch successfully.
Keywords |
| ascorbic acid, hydrazine, sodium sulphite, thiosulphate, vanadium(II), chloramines-T, titrimetric method and potentiometric method. |
I. INTRODUCTION |
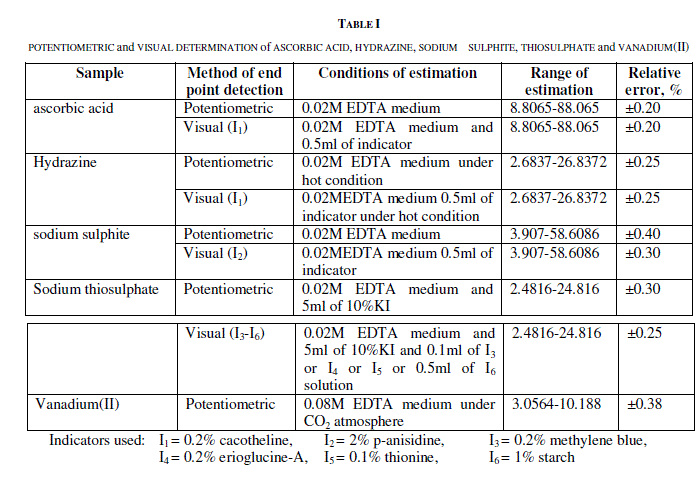
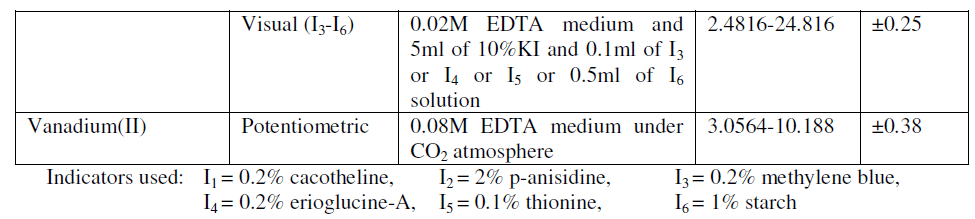
| Although wide variety of methods are reported for the estimation of ascorbic acid[1], hydrazine[2], sodium sulphite[3], thiosulphate and vanadium(II) a simple potentiometric and volumetric method of the estimation of the above ions are reported in simple aqueous medium of 0.02M EDTA. The medium of EDTA proposed is new in its attempt and have the advantage of eliminating mineral acid media. The scheme of different estimations are given in table-I. |
 |
 |
II. EXPERIMENTAL |
| A. Apparatus |
| The potentiometric measurements are made with a digital potentiometer(pacific-India) which is standardized with Weston Cadmium Electrode, a bright platinum rod is used as an indicator electrode and a saturated calomel electrode as the reference electrode. Electrical contact between the sample solution and the reference electrode is ensured with saturated potassium chloride salt bridge containing sintered glass ends. |
| B. Reagents |
| Solution (0.1N) of ascorbic acid, hydrazine, sodium sulphite, thiosulphate, vanadium(II) and chloramine-T were prepared and standardized([4]-[9]). EDTA solution of 0.1M is prepared and used in this investigation. 0.2% solutions of cacotheline, methylene blue, erioglucine-A, 0.1% thionine, 2% p-anisidine and 1% starch are prepared in distilled water and used. |
| C. Procedure |
| 1) Potentiometric estimations using chloramine-T as oxidant: An aliquot volume of ascorbic acid or hydrazine or sodium sulphite or thiosulphate or vanadium(II) solution is treated with enough volume of 0.1M EDTA solution to give an overall concentration of 0.02M (0.08M for vanadium(II)) in a total volume of 50ml. to this 5ml of 10% potassium iodide is added for the estimation of sodium thiosulphate only. The mixture is made up to the volume with distilled water. Carbon dioxide is passed through the contents for 3-5min when vanadium(II) is being titrated. The contents are titrated with chloramine-T solution potentiometrically. |
| 2) Visual estimations using chloramine-T as oxidant: Under the similar conditions adopted in potentiometric method, the visual method of estimation of ascorbic acid or hydrazine is carried out by adding 0.5ml of cacotheline; for sodium sulphite, 0.5ml of p-anisidine and for sodium thiosulphate 0.1ml of methylene blue or erioglucine-A or thionine or 0.5ml of starch as indicator. Then the solution is titrated with standard chloramine-T solution visually. |
III. RESULTS AND DISCUSSION |
| In the potentiometric estimation of all the ions carried, immediate stable potentials are observed through out the titration and sharp potential break is observed at the condition prescribed, but at lower concentrations of EDTA medium lesser values of estimation are observed in the instance of vanadium(II). Potential break observed per 0.04ml of chloramine-T solution is 260mv for ascorbic acid, 370mv for hydrazine(after a wait for 30sec-1min), 200mv for sodium sulphite, 200mv for sodium thiosulphate and 663mv for vanadium(II). The value of potential break has no significant change with the concentration of EDTA. |
| In the visual estimations sharp equivalence point is observed with all the indicators used. The colour transission of the indicator at the equivalence point is from pink – yellow with cacotheline, colourless – pink with panisidine, blue to green with methylene blue or erioglucine-A or thionine, and from colourless to blue with starch. The effect of concentration of the indicator is studied and it is generally observed except in very low concentrations of the indicator, the colour change is found to be sharp at all concentrations. In all instances of different indicators used the indicator correction is found to be negligible where the chloramine-T acts with the indicator to give colour. In the procedures adopted in certain instances potassium iodide is found to accelerate the reaction and the effect of potassium iodide concentration is also studied and the optimum concentration needed for the respective estimation is already mentioned. The procedures adopted are simple volumetric procedures and is accessible at all level of laboratories. |
ACKNOWLEDGEMENTS |
| The authors gratefully acknowledge the keen interest and advice of Prof.N.Krishna Murty, Emeritus Scientist-CSIR Honourary professor, and thanks to GVP College of Engineering (A) authorities for providing research facilities. |
References |
|