ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Jagadeesha Gowda G V1, Eraiah B2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
A series of glasses xPr6O11-(35-x) Ag2O-65B2O3 (x=0, 0.1 to 0.5 mol %) were synthesized by melt quenching technique. The absence of any crystalline peaks in the X-ray diffraction patterns of the present glass samples indicates the amorphous nature. Longitudinal and transverse ultrasonic velocities were measured at 5 MHz frequency and at room temperature. Elastic properties, Poisson’s ratio, Debye temperature, micro hardness and glass transition temperature have been calculated from the measured density and ultrasonic velocity. The variations of these parameters have been discussed with respect to Praseodymium oxide concentration.
Keywords |
| Ultrasonic velocities, Elastic moduli, Poisson’s ratio, Silver Borate glass, Praseodymium Oxide. |
INTRODUCTION |
| Among the various non-destructive evaluation (NDE) techniques, ultrasonic technique is a versatile tool for investigating the changes in microstructure, deformation process and mechanical properties of materials. The ultrasonic waves are closely related with the elastic and inelastic properties of the materials [1]. Borate glasses containing silver ions have been studied extensively because of their technological applications in solid state batteries [2]. The ability of boron to exist in three and four oxygen coordinated environments and the high strengths of covalent B-O bonds enable borates to form stable process [3]. In addition to this, rare earth elements are of interest in several high technology and environmental application areas. Glasses have been known for a long time as a convenient host for rare earth and have been widely used for the fabrication of solid state lasers. The physical properties indicate a strengthening of the glass structure as Pr ions are introduced [4]. Combining silver oxide (Ag2O) and rare earth oxide (Praseodymium oxide Pr6O11) with boron Oxide (B2O3) allows one to tune the physical properties in wide range depending on the glass composition. The present work intends to study the role of Pr ion on the elastic properties of Pr6O11-Ag2O-B2O3 glasses by employing ultrasonic technique. |
`MATERIALS AND METHODS |
| The new glass samples of xPr6O11-(35-x) Ag2O-65B2O3 system (where x=0, 0.1, 0.2, 0.3, 0.4 and 0.5 mol %) were prepared by the conventional melt quenching method. The starting materials, Praseodymium oxide (Pr6O11), silver oxide (Ag2O) and boric acid (H3BO3) were weighed in appropriate quantities. The chemicals were first mixed together by grinding the mixture repeatedly to obtain a fine powder. The powder was melted in porcelain crucibles at about 1200 °C in muffle furnace for few minutes to homogenize the melts. A special mould was made to get samples of cylindrical shape of dimensions 10mm x 10mm. The glass melt was poured into the brass mould and covered at the top with another brass block. All these glasses were annealed below their glass transition temperatures. Samples were later cut and polished to obtain end faces parallel and flat in a manner suitable for ultrasonic measurements. The amorphous nature of the samples was confirmed by X-ray diffraction (XRD) technique and X-ray diffraction analysis was carried out at room temperature using a Philips X’pert pro powder X-ray diffractometer by a Cu anode ( Kα, λ=1.54439Ãâ¡Ãº) radiation at 45kV and 30mA. The scanning angle 2θ range was from 100 to 700. The density of glasses were determined by Archimedes’s principle with toluene as buoyant liquid (density = 0.8635 gm/cm3 at RT) using the relation |
| Where, Wa is the glass sample weight in air, Wb the glass sample weight in buoyant and ρb the density of buoyant. The weight of each sample has been measured using a digital balance. The glass transition temperature (Tg) were determined by differential thermo gravimetric analysis (Setaram Instrumentation Labys DTA/6) at heating rate of 20 K min-1.The molar volumes Vm were calculated as M/ρ, where M is the molecular weight and ρ is the density of the corresponding glass samples. The ultrasonic velocities, Longitudinal (VL) and transverse (VT) at room temperature were obtained using the pulse-echo method (Model: VCT-70A). In this method, X-cut and Y-cut transducers operated at a fundamental frequency 5 MHz and a digital ultrasonic flaw was used. The various elastic properties of prepared glasses have been determined from measured densities and the velocities of longitudinal (VL) and transverse (VT) ultrasonic waves using the standard relations [5]. |
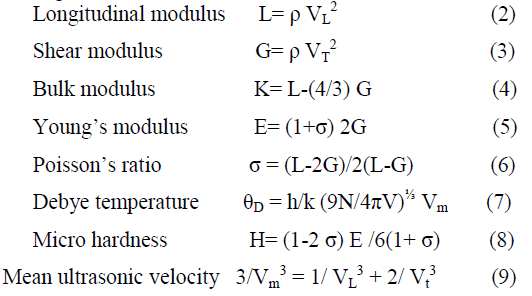 |
| Where L, G, K and E are longitudinal, shear, bulk and Young’s modulus respectively, ρ the density, σ the poison’s ratio, θD the Debye temperature, h the Plank’s constant, K the Boltzmann constant, Vm the mean ultrasonic velocity and (N/V) the number of vibrating atoms per unit volume and equal to (PN) where P the number of atoms in the chemical formula and N the Avogadro number. |
RESULTS AND DISCUSSION |
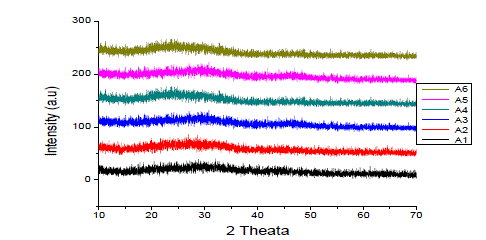
| Figure 1 shows XRD spectrum of all the glass samples. The X-ray diffraction spectra of all the samples showed the diffused bands characteristic of the X-ray diffraction pattern of amorphous materials. The spectra did not show any sharp peaks and this confirms the fact that glass samples are amorphous in nature. |
 |
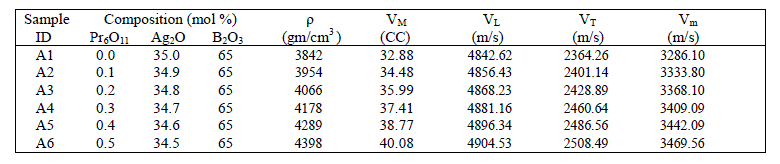
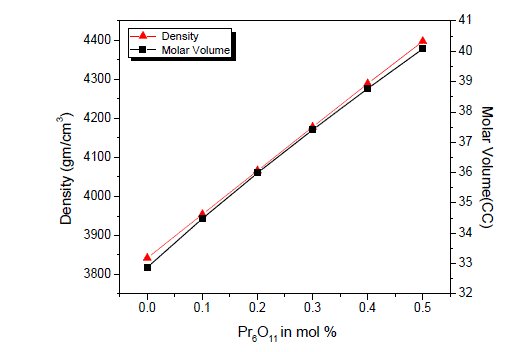
| Density is an effective tool to explore the degree of structural compactness, modification of the geometrical configuration of the glass network, change in coordination and variations of the dimensions of the interstitial holes. The values of density and molar volume are listed in table 1. It can be seen from figure-2, the density and molar volume increases almost linearly with the increase of Pr6O11concentration. This is a predictable result given that Pr ions have a larger mass that Ag and higher coordination number for oxygen [6]. Due to the addition of rare earth oxide Pr6O11, the three coordinated boron [BO3/2]0 units are converted to four coordinated boron tetrahedral [BO4/2]- units and thus the network dimensionality and connectivity increases [7]. The molar volume increases as a result of the creation of non bridging oxygen (NBOs) into bridging oxygen, which strengthens the silver borate glass. This may lead to efficient packing and compactness in the structure. |
 |
 |
| A plot of sound velocities vs composition is indicative of relative structure. The compositional dependence of longitudinal (VL) and transverse (VT) sound velocities are shown in figure 3. The velocities VL and VT increase with the increase of Pr6O11 concentration. The measured values of ultrasonic velocities with variation of Pr6O11 content are listed in Table 1. The longitudinal ultrasonic velocity increases from 4842.62 m/s to 4904.53 m/s and shear velocity increases from 2364.26 m/s to 2508.49 m/s. This increase in the ultrasonic velocities may be due to the strengthening of the glass structure. Addition of Pr6O11 content as a third component into the glass system results the combining of Ag-OAg and O-B-O bonds and this causes the NBOs into the bridging oxygen [4]. |
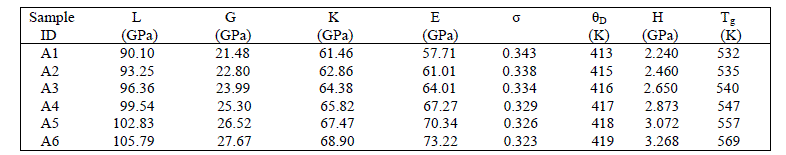 |
| The elastic moduli are proportional to the square of velocity variation of elastic moduli as a function of Pr6O11 concentration is shown in figure 4. Elastic moduli increases with the increase of Pr6O11concentration over the entire composition studied. The variations of elastic moduli are listed in Table 2 and can be explained on the basis of the structural consideration of borate network. As discussed in the earlier section, the vitreous B2O3 consists of planar [BO3/2]0 units [7, 8]. The addition of Pr6O11to B2O3 network creates [BO4/2]- units. This leads to increase in dimensionality and connectivity. Hence, both velocities and elastic moduli increase with the increase of Pr6O11 concentration [9]. |
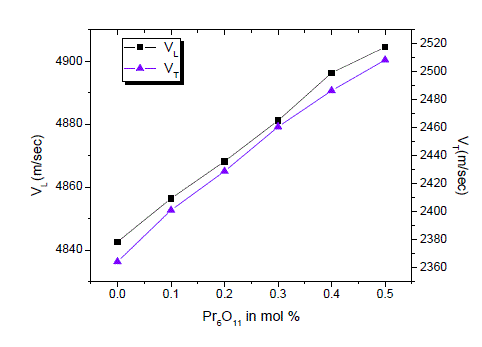 |
| The compositional dependence of Poisson’s ratio and Debye temperature as a function of Pr6O11 concentration is shown in figure 5. Poisson’s ratio is the ratio of transverse and linear strains for a linear stress. It can be seen that Poisson’s ratio decreases from 0.343 to 0.323 with increase of Pr6O11 concentration [10, 11]. Generally, as the glass structure weakens, the value of Poisson’s ratio increases but for this glass system the Poisson’s ratio decreases. This behaviour may be due to the change in the cross link density of the glass network [12]. Debye temperature θD plays an important role in solid materials in the determination of elastic moduli and atomic vibrations. θD represents the temperature at which all modes of vibrations in a solid are excited and its increase implies an increase in the rigidity of glass. In the present glass system the value of θD increases from 413K to 419K, as Pr6O11concentration increases. The observed increase in the Debye temperature supports the claim that the addition of Pr ion will strengthen the structure of silver borate glass which reduces the vibration of the lattice of the formed glasses [13]. As it can be seen from figure 5, the gradual increase of Debye temperature also suggests increase in the compactness in the structure leading to increase in mean sound velocity [14]. |
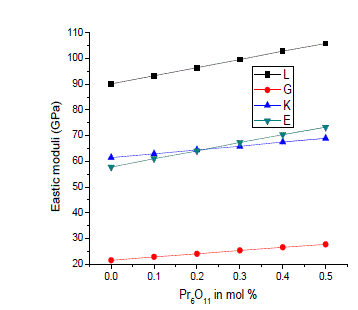 |
 |
| Micro hardness (H) of the glasses is shown in table 2, which was calculated according to the equation (8). The Variation of Micro hardness and Transition temperature (Tg) is shown in figure 6. The increasing trend of Micro hardness presumably, because of the connectivity of glass structure increases with the increase of Pr6O11 concentration. Glass transition temperature (Tg) is one of the fundamental properties related to viscosity of the glass and dependant on the composition of the glass. The values of glass transition temperatures are listed in table 2. It is clear from table 2, the values of Tg increases from 532 K to 569 K as the increasing concentration of Pr6O11. The increasing concentration of Pr6O11 makes the glass network progressively strong, which results in a increasing value of Tg. The above observation based on the elastic properties, indicates that the Pr ion prefers network forming position. |
CONCLUSION |
| The Pr6O11-Ag2O-B2O3 glass samples were successfully prepared. Based on the result obtained, it demonstrated that the density and molar volume increases with increases of Pr6O11 concentration. The ultrasonic velocities (VL and VT) were measured at room temperature using the pulse-echo method. The elastic constants, Poisson’s ratio and Debye temperature have been evaluated. The increase in the values of ultrasonic velocity and elastic constants has been attributed to an increase in the packing density and rigidity of glass samples. The Micro hardness (H) and Transition temperature (Tg) of the glass samples increases with increasing concentration of Pr6O11, which is due to strengthening of glass network. |
References |
|