ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
KuntalShah1, Dr.M.N.Reddy2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Aquatic plants are known to accumulate metals from their environment. The study focuses on assessment of heavy metal accumulation in certain aquatic macrophytesin comparison with water and sediments for phytoremediation. The aim of this study is to measure the concentrations of Cd, Co, Cu, Pb and Zn in four different species of aquatic macrophytes collected from estuarine zone of Tapi river near Surat city of Gujarat. Plants along with surface sediments and water were analyzed for Cd, Co, Cu, Pb and Zn contamination. The plant species selected were : Eichornia cressipes (Mart.)Solms-Loub (root and shoot) Persicaria glabra (Willd.)M.Gomez (root, stem and leaf) Pistia stratiotes L. (root and shoot) and Ceretophyllum demersum L. (whole plant). Samples were analyzed for heavy metals by AAS (Atomic Absorption Spectrophotometer). The greater accumulation of heavy metals was observed in the shoot of Pistia stratiotes. The lowest content was observed in Ceretophyllum demersum. Based on the concentration observed in the river's vegetation, the five heavy metals arranged in the descending order are : Pb > Cu > Zn > Co > Cd. The results of concentration of heavy metals in selected plants were compared with the standard, normal and critical toxicity range in plants. The detected value of Cd falls within normal range, while that of Co, Cu, Pb and Zn were within the critical range. However, Co and Cu showed the highest accumulation with alarming toxicity levels, which areconsidered as one of the most hazardous pollutants in river. Species like Persicaria glabra and Pistia stratiotes are also proposed as bioremediants, which are the most useful plant species in phytoremediation studies due to their ability to accumulate heavy metals in high concentration and their availability throughout the year.
Keywords |
| Aquatic macrophytes, Phytoremediation, Tapi river, Heavy metals, Surat |
INTRODUCTION |
| Rapid urbanization, industrialization, excessive use of fertilizers and pesticides, etc. have resulted in heavy metal pollution of land and water resources. The increasing load of heavy metals has caused imbalance in aquatic ecosystems and the biota growing under such habitats accumulate high amounts of heavy metals (Cu, Zn, Cd, Cr, Ni, etc.) which in turn, are being assimilated and transferred within food chains by the process of magnification (Pergent & Pergent- Martini, 1999). Direct discharge or wet and dry depositions of contaminants increase the concentration of trace elements in aquatic systems, thus resulting in their accumulation in sediments (Dunbabin and Bowmer, 1992; Sinicropeetal., 1992). In contrast with most organic materials, metals cannot be transformed by microorganisms and therefore accumulate in water, soil, bottom sediments and living organisms (Miretzkyetal.,2004). Macrophytes are considered as important component of the aquatic ecosystem not only as food source for aquatic invertebrates, but also act as an efficient accumulator of heavy metals (Janauer, 2001; Pajevićetal., 2001; Samecka-Cymerman & Kempers, 2002; Samecka- Cymermanetal., 2005). Bioavailability and bioaccumulation of heavy metals in aquatic ecosystems is gaining tremendous significance globally. Several of the submerged, emergent and freefloating aquatic macrophytes are known to accumulate and bioconcentrate heavy metals producing an internal concentration several folds greater than their surroundings (Chenetal.,2008). Aquatic plants absorb elements through roots and / or shoots (Pip and Stepaniuk, 1992; Jackson, 1998). Various species show different behavior regarding their ability to accumulate elements in roots, stems and / or leaves. Therefore, it is useful to identify the plant organ that absorbs the greatest amount of trace elements (St-Cyr and Campbell, 1994; Baldantonietal., 2004). In aquatic systems, where pollutant inputs are discontinuous and pollutants are quickly diluted, analysis of plant components provides timeintegrated information about the quality of the system (Baldantoni et al., 2005). |
| Phytoremediation has several advantages and is the most significant one in study of sub-lethal levels of bioaccumulated contaminants within the tissues / components of organisms, which indicate the net amount of pollutants integrated over a period of time (Lovett-Doustetal., 1994). Biomonitoring of pollutants using some plants as accumulator species, accumulate relatively large amounts of certain pollutants, even from much diluted solutions without obvious noxious effects (Raveraetal.,2003). As the macrophytes concentrate great amount of various substances (eg.Metals) and are consequently useful indicators of local pollution, the aim of present study was to assess the toxicity status induced by five heavy metals (Cd, Co, Cu, Pb and Zn) in four selected aquatic macrophyte species (passive biomonitors) in comparison with water and sediments. |
MATERIALS AND METHODS |
| Study area |
| Tapi River originates from Satpura Mountain in Betal district of Madhya Pradesh at an elevation of 725m above sea level. It has a drainage area of about 6400 km2 and hourly flow of 120,000,000 cubic yards during the season of extended floods. It runs 724 km from its origin through three states (Madhya Pradesh, Maharashtra and Gujarat) before it joins the Arabian Sea (C.P.C.B., 1994). It has a dam over it at Ukai around 98 km away from the mouth of estuary and two Weirs, one at Kakrapar–70 km away from the mouth of estuary and another at Rander–15 km away from the mouth of estuary. There are many estuaries in Gujarat. Out of them Narmada and Tapi are larger ones. Tapi meets the sea near Surat city (21°12 N and 72°50 E). By the construction of Weir Cum Causeway at Rander in Surat in 1994. Tapi river has been divided into two, upstream of the Weir as a fresh water and downstream of the Weir as a marine inlet. Surat is regarded as an industrial capital of Gujarat state. It has large number of chemical, diamond, textile, iron and steel, cement, pharmaceuticals and many other industries. As a result of rapidly expanding industrialization, urbanization and population blast, streams, lakes, ponds and other water bodies are being polluted. The untreated waste water thrown out by the industries in the river Tapi is the major cause of water pollution. The untreated waste water and sewage thrown in the river Tapi have also increased water pollution in this area. Due to lack of sanitation facilities polluted water enters directly into the river. |
 |
| Water and sediment sampling |
| Surface water and composite sediment samples were collected at random from different areas of the river covering all directions. Soon after collection, the water samples were filtered through 0.45μm (poresize) Millipore filter and preserved in plastic bottles by the addition of a few drops of nitric acid. Sediment samples were preserved in air-dry plastic bags. The samples were labeled carefully and brought to the laboratory for further analysis. |
| Plant sampling |
| Four aquatic macrophytes from the river were selected as passive biomonitors for estimating the accumulation of five heavy metals (Cd, Co, Cu, Pb and Zn) by them. The plant species selected were : Eichornia cressipes (Mart.)Solms-Loub (root and shoot) Persicaria glabra (Willd.)M.Gomez (root, stem and leaf) Pistia stratiotes L. (root and shoot) Ceretophyllum demersum L. (whole plant). Healthy aquatic plants were collected, washed with river water to remove periphyton and sediment particles. Therefore, the element concentrations in the plant parts refer not only to tissue concentrations but also to adsorbed elements on plant surface. The collected plant species were placed in plastic bags, labeled carefully and brought to the laboratory. Polythene tools were used in sampling and storing the collected matrices to avoid the metal contamination. Plant species were identified according to Shah (1978). |
| Chemical analysis of water, sediment and plant samples |
| Sediment samples were air-dried, sieved through 2mm governorates sieve and kept for analysis. Each fresh aquatic plant species sorted into different parts was dried at 80°C in hot air oven for 48hrs. The duplicate samples of water, sediment and plant-parts were chemically analyzed for detection of heavy metals (Cd, Co, Cu, Pb and Zn). Accurately 0.5g of dry powder of each sample was weighed, and digested with conc. HNO3 and conc. H3PO4 (5:2) as prescribed by Jackson, 1960. Towards the end of the digestion, the flasks were brought to near dryness. The solutions were made to 100ml each in a volumetric flask with double distilled water. The water sample was prepared according to method of APHA 3030H (1975). The blanks were run with set, and the samples were analyzed in Atomic Absorption Spectrophotometer (AAS-4141, Electronic Corporation of India) at Navsari Agriculture University, Navsari, Gujarat. The concentrations of heavy metals such as Cd, Co, Cu, Pb and Zn were analyzed and calculated in mg/l for water and in mg/kg for sediment and plant samples. Mean values of duplicate subsamples of the water, sediment and plant samples were considered. |
| Data analysis |
| The values of the ratios between element concentrations in the sediments and those in the water were calculated. The mean values of heavy metals were calculated for water, soil and plant samples. Ranges of heavy metal contents and toxicity status in the tested plant species were compared with normal and critical ranges in plants. Correlation coefficient was calculated between metal-pairs in plants to check if differences exist between different metal combinations. The products of the correlation coefficient (r) were evaluated as follows: |
| 1. 0–0.3: No correlation; |
| 2. 0.3–0.5: Low correlation; |
| 3. 0.5–0.7: Medium correlation; |
| 4. 0.7–0.9: High correlation; |
| 5. 0.9–1.0: Very high correlation |
| Mobility index was calculated for each level by using the formula: |
 |
| Mobility index showed bio mobility and transport of heavy metals through different levels, for example: waterroot and root-shoot or water-root, root-stem and stem -leaf, which became functional to understand the transport mechanism of heavy metals in plant components such as root and shoot or root, stem and leaves. |
RESULTS |
| Water and sediments |
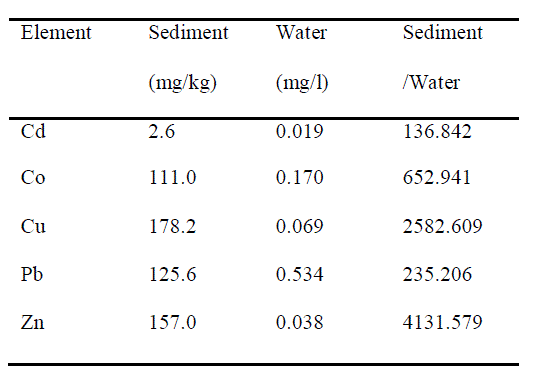
| The concentrations of the elements analyzed were far higher in the sediments than those analyzed in the river water filtered through Millipore filter (0.45μm opening size). Of the elements analyzed metal concentrations arranged in descending order for sediment sample are: Cu (178.2mg/kg) > Zn (157.0mg/kg) > Pb (125.6mg/kg) > Co (111.0mg/kg) > Cd (2.6mg/kg) and metal concentrations arranged in descending order for water sample are: Pb (0.534mg/l) > Co (0.170mg/l) > Cu (0.069mg/l) > Zn (0.038mg/l) > Cd (0.019mg/l). The values of the ratio between element concentrations in the sedimements and those in the water arranged in descending order are: Zn (4131.579) > Cu (2582.609) > Co (652.941) > Pb (235.206) > Cd (136.842) (Table 1). |
| Table1. Heavy metal concentration in sediments and water and ratios between the concentration in the sediments and that in the water |
 |
| Macrophytes |
| Table 2 shows the values of concentration of five heavy metals in analyzed plant parts of selected aquatic macrophytes along with mean values of heavy meal concentration in each selected plant part and mean concentration of each heavy metal in all selected plant parts. |
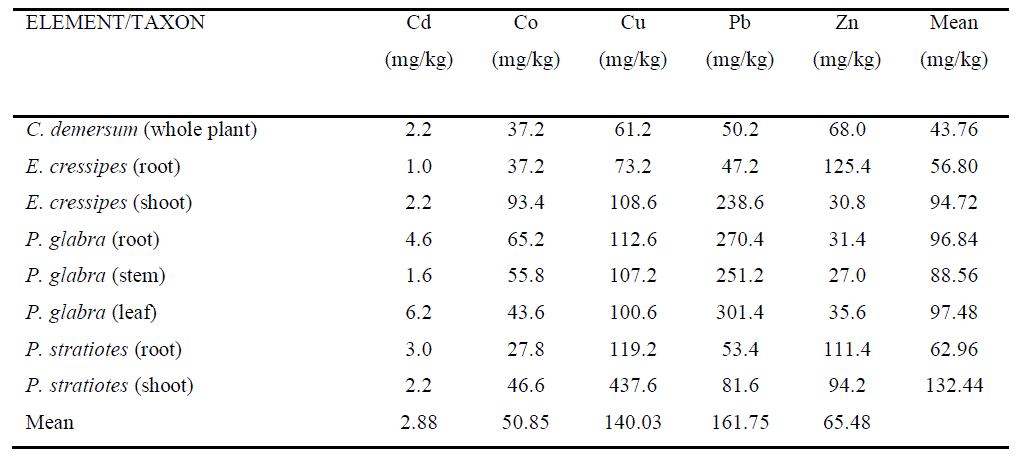 |
| The mean concentration values of heavy metals in analyzed plant arranged in descending order are: Pb > Cu > Zn > Co > Cd. P. stratiotes (shoot) showed the highest capacity of accumulation with highest concentration of Cu (437.6mg/kg) and lowest concentration of Cd (2.2mg/kg) followed by P. glabra (leaf), P. glabra (root), E. cressipes (shoot), P. glabra (stem), P. stratiotes (root), E. cressipes (root) and C. demersum showed the lowest capacity of accumulation with highest concentration of Zn (68.0mg/kg) and lowest concentration of Cd (2.2mg/kg). In all the plants analyzed, more accumulation of heavy metals was observed in shoot system in comparison with that observed in root system. |
| The concentration of individual metal also varies from species to species. The content of Cd ranged from 2.2 mg/kg in C. demersum (whole plant), E. cressipes (shoot) and P. stratiotes (shoot) to 6.2 mg/kg in P.glabra (leaf). The Co content was found lowest in P. stratiotes (root) (27.8 mg/kg) and highest in E. cressipes (shoot) (93.4mg/kg). On the other hand, C. demersum (whole plant) showed lowest amount of Cu (61.2mg/kg), while highest amount of the same was recorded in P. stratiotes (shoot) (437.6mg/kg). Besides, lowest concentration of Pb was recorded in C. demersum (whole plant) (50.2 mg/kg), while highest concentration of the same was observed in P. glabra (leaf) (301.4 mg/kg). The minimum concentration of Zn was recorded in E. cressipes (shoot) (30.8 mg/kg), while maximum content of the same was recorded in E. cressipes (root) (125.4 mg/kg). |
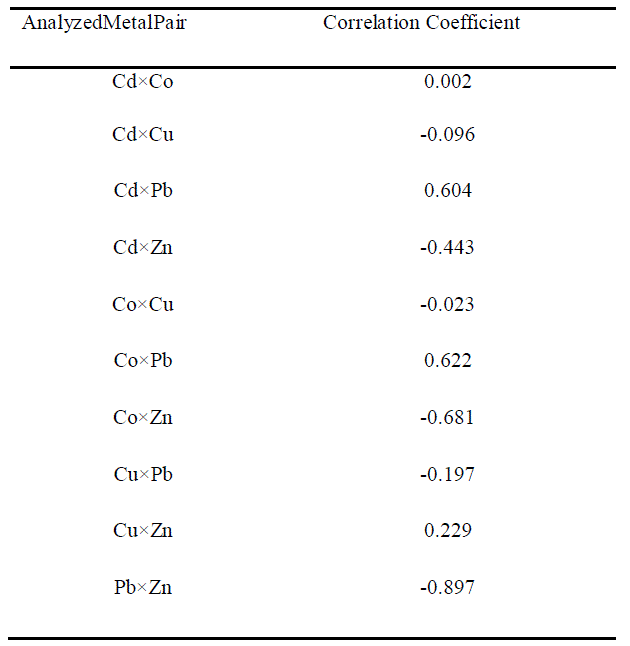 |
| Results of correlation coefficients calculated for different metal pair showed that medium positive correlation (0.5-0.7) was observed in Cd and Pb, and Co and Pb whereas medium negative correlation was observed only in a single metal pair i.e., Co and Zn and high negative correlation was observed in a single metal pair i.e., ,Pb and Zn. |
| State of heavy metal pollution in plants |
| Comparing with standard normal and critical range in plants, the mean concentration of Cd (1.0-6.2 mg/kg) falls within the normal range whereas the mean concentrations of Co (27.8-93.4 mg/kg), Cu (61.2-437.6 mg/kg), Pb (47.2-301.4 mg/kg) and Zn (27.0-125.4 mg/kg) were recorded within critical range. Concentration of Co was encountered much higher than its critical range (1-8 mg/kg) in all plants analyzed with the highest concentration in the shoot of E. cressipes (93.4 mg/kg). Concentration of Cu was encountered higher than its critical range (25-90 mg/kg) in all plant parts analyzed except C. demersum (whole plant) and E. cressipes (root), but its concentration was far higher than its critical range in shoot of P. stratiotes (437.6 mg/kg). Thus Co and Cu seem to be hazardous in the study area,as their concentrations are extremely high. |
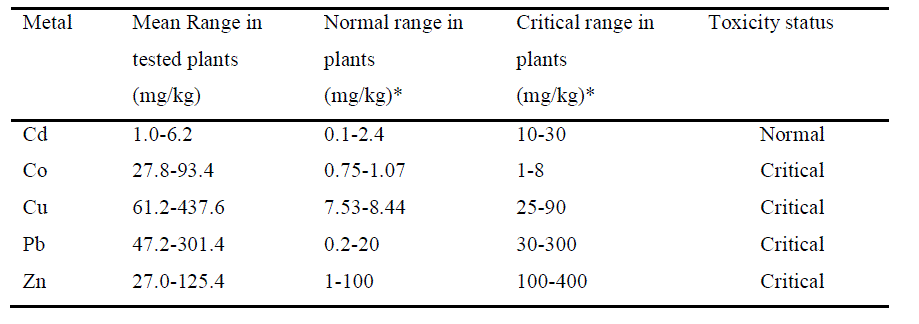 |
| Mobility index (MI) showed bio mobility and transport of heavy metals through different levels. Present findings revealed that MI of Pb (94.01) was lowest and that of Zn (1789.47) was highest in C .demersum. Comparing other three plants, mean MI for water to root was found highest in E. cressipes followed by P. stratiotes and P. glabra. In E. cressipes, W-R MI of Cu (5033.59) was highest and that of Cd (52.63) was lowest, whereas R-Sh MI of Pb (5.06) was highest and that of Zn (0.25) was lowest. In P. stratiotes, W-R MI of Zn (2931.58) was highest and that of Cd (157.89) was lowest, whereas R-Sh MI of Cu (3.67) was highest and that of Cd (0.73) was lowest. In P. glabra, W-R MI of Cu (1631.88) was highest and that of Cd (242.11) was lowest, whereas R-St MI of Cu (0.95) was highest and that of Cd (0.35) was lowest, whereas St-L MI of Cd (3.88) was highest and that of Co (0.78) was lowest. |
DISCUSSION |
| The present study revealed that some of the aquatic plants possess greater accumulation ability for selected heavy metals. These results agree with the reports of Peverly (1985); Sawidisetal (1995) and Abouel-kheiretal (2007,a&b). In present study very high accumulation of Cu was found in shoot system of P. stratiotes (437.6 mg/kg), which indicates that P. stratiotes can be effectively used as phytoremediant for Cu contaminated waterbodies. Same way Cd, which is considered as one of the most toxic elements, was found in very higher amount in root (4.6 mg/kg) and leaf (6.2 mg/kg) of P. glabra, which suggests that P. glabra can remediate Cd contaminated sites more effectively. Concentration of Pb was found very high in all parts of P. glabra (root-270.4 mg/kg, stem-251.2 mg/kg and leaf-301.4 mg/kg), which indicates high potential of P. glabra to be used as phyto remediant at the sites contaminated by Pb, one of the most hazardous pollutants. |
| Generally the submerged plants show less accumulation of heavy metals. Same results were recorded in present study. The submerged species C. demersum showed least accumulation of heavy metals. Lovett-Doustetal. (1994) reported that the accumulation levels of pollutants in aquatic ecosystems may be higher in sediments than in plants. The present study agrees with this finding. Mean concentration of all elements in all selected plant parts except that of Pb and Cd, were less in studied plants compared to the concentrations of the same elements in sediment. Comparing with permissible limits of heavy metals in water, the concentrations of Cd, Pb and Zn in river water were beyond the permissible limits of BIS (Bureau of Indian Standards), 2003. The concentration of Pb (0.534 mg/l) was at most alarming level which was ten times more than the permissible limit (0.05 mg/l), which makes water toxic. Though the concentration of Zn in river water was within the permissible limit as prescribed by BIS, 2003, due to its higher accumulation by plants, its concentration within the studied plants is in critical range. By the process of biomagnifications this critical concentration can enter the food chain and organisms can become victim of toxic effects of Zn. So that it is not wrong to conclude that the permissible limits in water are not safe always. |
CONCLUSION |
| It can be concluded from the present study that P. stratiotes and P. glabra accumulated heavy metals in much higher concentration. Perhaps it might be the reason that these two species are more efficient in uptakeof heavy metals. Therefore these plants can be used more effectively as ‘Phytoremediants’ than other aquatic macrophytes. |
References |
|