ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
S. Dhanapal1 and D. Sathish Sekar2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Humic acid is a principal component of Humic substances, which are the major organic constituents of soil (humus), peat, coal, many upland streams, dystrophic lakes, and ocean water. It is produced by biodegradation of dead organic matter. Leonardite act as a best source for humic acid.The antioxidant activity of coal extracted humic acid (CHA) on in vitro propagated Musa accuminata was examined with commercially available Keradix (KHA), Humic Rooting (BHA). The Polyphenols was found to be higher in native plants of about 146.2μg/ml whereas in CHA it is 54μg/ml. Similarly the amount of protein in BHA is found to be 7μg/ml and CHA is 2.65μg/ml. The catalase activity was found to be higher in BHA is 0.09 U/mg compared to CHA which is estimated as 0.048 U/mg. GST was measured and it was found to be significantly increased in CHA of 582.99 U/mg compared with other commercially available products. The Ascorbic acid Peroxidase activity was higher in Keradix of about 725.31 U/mg APX. The SOD activity was inhibited higher in CHA of about 41.34%.
Keywords |
| Antioxidant activity, Humic Acid, Humic Rooting, Leonardite, Keradix, Musa accuminata. |
INTRODUCTION |
| The term “humus” has been familiar for over two hundred years. Humus is a transformation product of the vegetable as well as animal remains, which are resistant to further decomposition than the initial biomass. The process of dead organic matter decomposition in the soil does not come to its completion but stops at a certain stage, when simple decomposition products such as phenols, carbohydrates, and amino-acids transform into much more complex compounds - the humic acids. in order to ensure fertility of the soil, its humus content should be rather high [21]. Humic substances are the naturally occurring organic materials and they result from the decomposition of plant and animal residues [18]. They contain an acidic group carboxy and a phenol OH functional groups [11]. |
| Leonardite is substance known for its soft waxy, black or brown, shiny, vitreous mineraloid texture and that is easily soluble in alkaline solutions. It is an oxidation product of lignite. Leonardite is a rich source of humic acid (up to 90%)used as a soil conditioner, as a stabilizer for ion-exchange resins in water treatment, in the remediation of polluted environments and as a drilling additive [13]. Leonardite is highly rich in humic acids when compared to other organic products. The reason behind it is because the properties of leonardite are highly bioactive through its molecular structure. It acts as a biocatalyst and biostimulant to the plant. They are spread widely over the surface of the earth and occur in almost every terrestrial and aquatic environments [2]. They group into humic and non humic substances. The non humic substances are again grouped by its characteristic which are physical and chemical. They include carbohydrates, peptides proteins, fats, waxes, amino acids and low molecular weight organic acids [26]. |
| The production by excess transfer energy from triplet excited chlorophyll to oxygen (singlet oxygen formation) or photoreduction of oxygen (formation of superoxide, hydrogen peroxide, and hydroxyl radical) [19] we result in the Reactive oxygen species . Excess accumulation of reactive oxygen species results in a series of oxidative injuries to plants [7], [10], [29] and [30].Oxidative injury involves the initial formation of reactive oxygen species and its subsequent reaction with macromolecules. Proteins, lipids, polysaccharides and nucleic acids can be damaged [5], [8], [17] and [18]. As a result, normal cell metabolism can be seriously disturbed. |
| Intrinsic antioxidant defense mechanisms are possessed by plants for coping with reactive oxygen species [24]. Essentially, antioxidant defensive systems fall into three general classes. Firstly, the lipid-soluble, membraneassociated antioxidants (e.g. tocopherol, carotene); followed by the water soluble reductants (e.g. glutathione, ascorbate); and enzymatic antioxidants (e.g. superoxide dismutase, catalase and enzymes of the ascorbate/glutathione cycle [27] and [31]. The toxicity of an externally imposed biotic or abiotic oxidative stress can be partly attributed to the overriding of existing resistance mechanisms. Only when those mechanisms are overwhelmed would injury occur [6]. This suggests that the strengthening of the defense mechanisms, through enhancing the functions of their components (such as ascorbic acid, tocopherol, carotene and superoxide dismutase) may reduce or prevent oxidative injury and improve water stress resistance of plants. |
| There has been a growing interest in polyphenols as potential disease preventing agents and recent trends focus mainly on the characterization and utilization of natural antioxidants. As these compounds are found widely, it would be worth investigating the nature of polyphenols and other antioxidants that are present in Humic acid induced Musa accuminata (clone GW-04), a potential source of antioxidant activities. Hence, the present study has been aimed to evaluate in vitro antioxidant activities using the extracted Humic acid from various sources along with a commercially known Humic Acid in Musa accuminata to assess its protective role against free radical induced cell damage. |
LITERATURE REVIEW |
| Humic substances contain moieties that donate electrons to reactive oxidants under toxic conditions. as such, Humic substances may act as antioxidants in biogeochemical and pollutant redox reactions and in engineered systems, such as water treatment facilities. In this work, we quantified the electron donating capacities of HS as a function of solution pH and redox potential Eh. Phenolic groups were identified as major electron donating moieties [33]. |
| Ascorbic acid is distributed in the cytosol as a water-soluble antioxidant. Dry (and thus sunnier and warmer) conditions have been noted to increase the AA content in turnip greens [34], onions and black currant [35]. Black currant grown in hot, dry years was found to contain more than twice the AA of those grown in wet years. [36] has reported an enhancement in AA content of plants which were subjected to severe water stress. Research results indicate that, under moderately dry conditions, a slight increase in the total AA concentration occurred. There was an extraordinary upsurge in the total AA content along with abnormally high oxidation-reduction ratio and very high level of respiration under extreme drought conditions, suggesting a complete breakdown of the metabolism [37]. [38] noted that AA increased significantly under water stress. In contrast, [39] found a decrease of ascorbate content of droughtstressed treatment in 9 out of 10 grass species. |
| Superoxide dismutases are closely related to the capability of plant tolerance to various stresses, such as water stress, chilling stress, herbicides, and pathogens [40]-[42]. It was reported that SOD enhances water stress tolerance of plants. In tomato, cytosolic Cu/Zn-SOD was induced strongly by drought, while chloroplastic Cu/Zn-SOD remained largely unaffected [42]. In two mosses, the drought-tolerant Tortula ruralis and drought-sensitive Cratoneuron filicinum were compared [43]. The drought-tolerant moss showed lower levels of lipid peroxidation, together with increased levels of SOD; the opposite occurred in the sensitive moss. Drought tolerant and intolerant maize in breds were analyzed by [44], and resistance was found to correlate with Cu/Zn-SOD. Drought stressed tomato showed increases both in transcript levels of cytosolic Cu/Zn-SOD gene (eight-fold) and enzyme activity (five-fold), whereas an increase in enzyme activity was observed in chloroplastic Cu/Zn-SOD (2.4-fold) without a corresponding increase in transcript levels [45]. |
MATERIALS AND METHODS |
EXTRACTION OF HUMIC ACID FROM LEONARDITE: |
| SAMPLE COLLECTION |
| The coal sample (leonardite) was collected from Mines II of Neyveli lignite corporation, Neyveli and the multistage culture of Grand naine (Musa accuminata) was collected from Genewin Biotech, Hosur. |
EXTRACTION OF HUMIC ACID WITH VARIOUS SOLVENTS: |
| Humic acids were extracted from the resulting leonardite, using extraction methods that are capable of extracting humic acids. The extraction of humic acid from leonardite 5 g was extracted with 50 ml of (0.1 M NaOH, 0.1 M KOH, 0.1 M Na4P2O7, 0.25 M NaOH, 0.25 M KOH, 0.25 M Na4P2O7) and stirred for 1 min. The pH of the suspension was maintained at 13 by addition of NaOH (20%, w/v) and allowed to stand for 3 h. The mixture was centrifuged at 3500 rpm for 15 min in order to eliminate the precipitation. The supernatant was then acidified with 50 ml of 0.1 M Hcl and stirred for 1 min. The pH of the suspension was adjusted to 1 by the addition of Hcl (10%, w/v), and it was allowed to stand for overnight. Both Fulvic acids (supernatant) and Humic acids (precipitation) fraction were obtained by centrifuged at 3500 rpm for 15 min in order to eliminate the supernatant and precipitation washed once with distilled water. The Humic samples were dried at 60oC, the highest yield from each solvent extract is weighed and concluded [22]. |
ESTIMATION OF % HUMIC ACID: |
| The estimation of the percentage of humic acid was elaborated by Stevenson in 1994. 0.1g of Humic acid was weighed and ground in to a fine powder. It was then dissolved in 10 ml of extraction buffer containing 0.2M NaOH, 0.0032 M DTPA (Diethylene triamine pentaacetic acid, ROLEX-Mumbai), 2% ethanol. The sample is mixed well, centrifuge the aliquot of the sample to remove any particulates. The supernatant is saved as the sample; 1 ml of the sample was taken and mixed with 5 ml of water. The OD was taken at 450 nm using Titan Biotech Humic acid as standard (50-300mg). |
ENHANCEMENT OF ANTIOXIDANT POTENTIAL IN MUSA ACCUMINATA: |
| ESTIMATION OF POLYPHENOL |
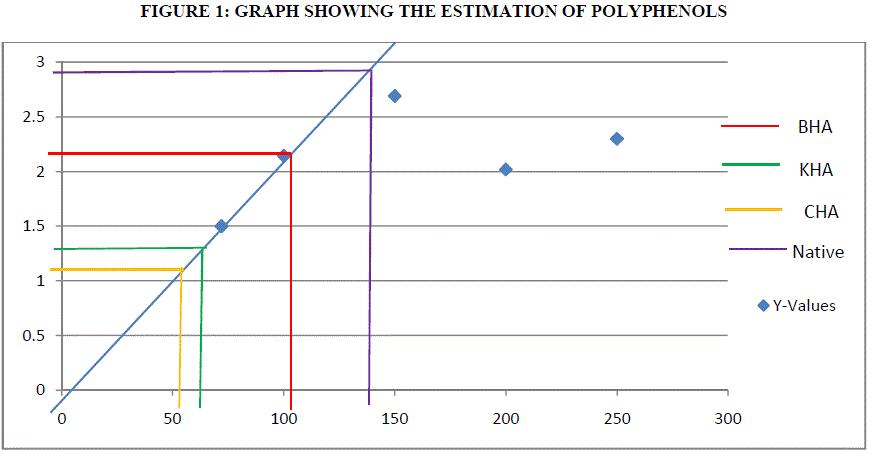
| The amount of polyphenols in the leaf sample (growth regulator supplemented and humic acid propagated) was determined by Folin-Ciocalteu’s method as described by [15]. Leaves were homogenized in 80% ethanol and centrifuged separately. The supernatant was saved. The residue was again extracted with 80% ethanol. The supernatant was collected and evaporated to dryness. The residue was dissolved in water and 2 ml was taken for the experiment. The volume was made up to 2.5 ml with water and 0.5 ml of diluted folin ciocalteu reagent wad added. After 3 minutes, 2 ml of 20% Na2CO3 was added. Incubated in boiling water bath for 1 minute. Measure OD at 650 nm. A standard graph was plotted using catechol as standard with the concentration ranging from 50-250μg. The results were expressed as milligrams of Gallic acid equivalent per gram of dry weight (GAE g/dw). |
PREPARATION OF ENZYME EXTRACT |
| Frozen leaves are homogenized at 4°C in an prechilled mortar and pestle with liquid nitrogen in 100 mm potassium phosphate buffer (pH 7.8), 1mm EDTA, 1% triton x 100, 15% glycerol, with 50 mg of PVP per gram of leaf. Crude homogenates was centrifuged at 15,000 rpm 15 minutes at 4°C. Supernatant was frozen at -20° C. These enzyme extract is used to determine the protein content by Bradford assay using BSA as standard. |
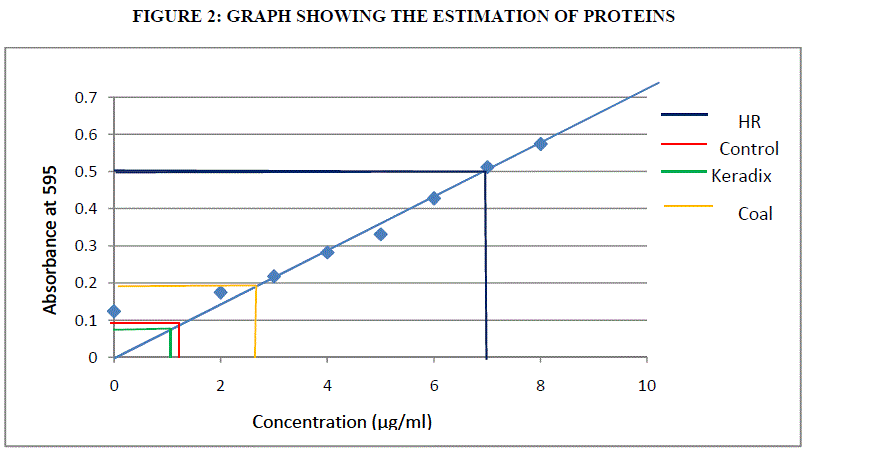
ESTIMATION OF PROTEIN BY BRADFORD ASSAY |
| Pipette a standard BSA 50- 250 μl with the concentration of 50- 250 μg and 50 μl of sample in test tubes. The volume was adjusted to 0.250 ml with distilled water. 3ml of Bradford reagent was added and mixed thoroughly; it was then incubated at room temperature for 10 minutes. The optical density was read on spectrophotometer at 595 nm and the readings were recorded. The calibration curve or standard curve was drawn by plotting average optical density reading on Y axis against standard protein concentration on x axis. |
| The value X from the graph was recorded corresponding to optical density of the test sample. The concentration of protein in the test sample was calculated using the formula |
| Protein concentration in test sample = X/V (mg/ml) (1) |
| Where X – Value from graph in μg |
| Y – Volume of sample in μl |
SUPEROXIDE DISMUTASE |
| Superoxide dismutase was generated by xanthine oxidase and detected by nitro blue tetrazolium (NBT) reduction method [14]. Reagents in this study are prepared with 50mM potassium phosphate potassium hydroxide buffer (pH 7.8). The reaction buffer should contain 50 μl of 0.6 mM NBT, 20 μl of 15 mM Na2EDTA(pH 7.4), 30 μl of xanthine oxidase solution, 150 μl of enzyme sample and 1.5 μl of potassium phosphate potassium hydroxide buffer is used as control. Reaction was initiated by the addition of xanthine oxidase at 25º C. The absorbance at 405 nm was recorded every 20 seconds for 5 minutes in UV visible spectrophotometer. Results were expressed as % of inhibition relative to control, given by |
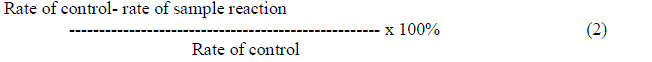 |
GLUTATHIONE S TRANSFERASE (GST) |
| Glutathione S Transferase activity was determined by using IZZ0 AND IZZ0 method. [12]. 0.5 gram of plant material was homogenized in 5 ml of 0.1 M of potassium phosphate buffer (pH 7.4) that contains 1Mm of EDTA and 0.2 mM PVP. The extract was centrifuged at 8,000 rpm at 4º C for 20 minutes. 1ml of sample was added to 3 ml of 0.1 M phosphoric acid buffer (pH 7.0) that containing 2mM of CDNB and 2mM of reduced glutathione. The increase in absorbance was recorded at 340 nm for7 minutes. The molar extinction coefficient of 9.6 mM/cm for CDNB GSH was used and expressed in units of enzyme activity per 1 mg of protein. |
| Volume activity =[(Total volume) / (Extinction Coefficient x dilution factorx sample)] x [(A / incubation time)] (5) Specific activity =Protein concentration / Volume activity (6) |
ASCORBIC ACID PEROXIDASE (APX) |
| APX activity was determined according to [28]. APX extraction was performed in 1.5 ml of suspension solution including 50 mM Tris-HCl (pH 7.2), 2 % PVP, 1 mM Na2EDTA, and 2 mM ascorbate. Assay solution contained 50 mM potassium phosphate buffer (pH 6.6), 2.5 mM ascorbate, 10 mM, H2O2 and enzyme containing 100 μg protein in a final volume of 1 ml. The enzyme activity was calculated from initial rate of the reaction using the extinction coefficient of ascorbate (ε = 2.8 mM cm-1 at 290 nM). Volume activity =[(Total volume) / (Extinction Coefficient x dilution factorx sample)] x [(A / incubation time)] (5) Specific activity =Protein concentration / Volume activity (7) |
RESULTS AND DISCUSSION |
HA ENHANCING THE ANTIOXIDANT POTENTIAL OF MUSA ACCUMINATA |
| In the previous study the antioxidant activity in Musa accuminata was evaluated by using coal extracted Humic acid sample only. Now in the present study it was extended with comparison of commercially available Humic acid products like Humic rooting and Keradix [32]. |
ESTIMATION OF POLYPHENOLS: |
| Polyphenols are the secondary metabolites of plants and are involved in defense against the pathogens. Catechol is used a standard curve. Antioxidant action of phenolic compounds is due to their high tendency to chelate metals. Phenolics possess hydroxyl and carboxyl groups, able to bind particularly iron and copper [3]. Humic acid exposed plants were found to exhibit 104 μg of GAE g/dw for BHA, 54 μg of GAE g/dw for CHA (Coal) and 70 μg of GAE g/dw for KHA (Keradix) while native plant exhibited 146.2 μg of GAE g/dw. The increase of polyphenols in the Humic acid exposed plants may be due to the exposure of roots to the heavy metals that are present in activated charcoal used in rooting medium or due to metal ions in water used. But HA exposed plants found to inactivate iron ions by chelating and additionally suppressing the superoxide-driven fenton reaction, which is believed to be the most important source of ROS [1]. Direct chelation action of polyphenols is observed in rhizome of Nympheae for Cr, Pb and Hg. |
 |
ESTIMATION OF PROTEIN BY BRADFORD ASSAY: |
| Proteins are the amino acids which are important for its growth and development. The proteins are estimated by Bradford assay method using BSA (Bovine Serum Albumin) as standard. Using formula (1) the estimation of protein was done. |
 |
CATALASE TEST: |
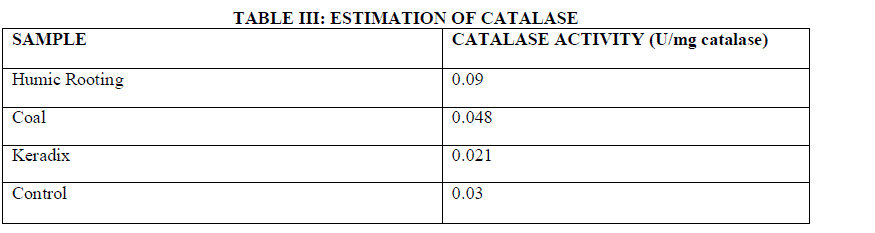
| Catalases are found in plants that help in degradation of Hydrogen per oxide. This activity was measured by reading the absorbance at 240 nm. Catalase is an enzyme that converts hydrogen peroxide to water. This enzyme rapidly destroys a vast majority of H2O2 produced in peroxisomes during photorespiration and formed as a result of mitochondrial electron transport, but it allows low steady state levels to persist presumably to maintain redox signaling pathways [20]. In this work, a slight increase in the CAT activity is observed in humic acid exposed plants than native plants. GW04-BHA exhibited least activity at 0.09 U/mg catalase, GW04-CHA exhibited 0.048 U/mg catalase, GW04- KHA exhibited a specific activity of 0.021 U/mg catalase and GW04-NA exhibited only 0.03 U/mg catalase . Increase of CAT activity is observed in NaCl treated barley roots. CAT has been observed as the major enzyme detoxifying hydrogen peroxide in barley under salt stress [25]. |
 |
| Using formula (3) and (4) the estimation of CAT was evaluated. Table III indicates the Catalase activity was highest for Humic Rooting of 0.09 U/mg catalase and least for commercially available Keradix of 0.021 U/mg catalase. The Coal sample exhibited an enzyme activity of 0.048 U/mg catalase. |
GLUTATHIONE S TRANSFERASE ACTIVITY: |
| Glutathione S Transferase (GST) is an abundant protein in plants that help in cellular metabolism and they were estimated by reading the absorbance at 340 nm for 7 min. The raised GST activity is suggested to be a marker for the enhanced antioxidant activity in HA exposed plants. It is possible that chlorophenols such as CDNB may induce GST directly or indirectly by toxic organic radicals, ROS or through lipid peroxidation. least GST activity is found in GW04-BHA of about 79.28 U/mg GST, GW04-CHA activity was maximum and found to be 582.99 U/mg GST, 564.6 U/mg GST was found in GW04-KHA while GW04-native exhibit a specific activity of 557.21 U/mg GST. [16] noted increased GST activity L. esculentum roots under salinity stress. Using formula (5) and (6) the estimation for GST was carried out. |
 |
| Table IV showed the GST activity was highest in coal sample of 582.99 U/mg GST than other three samples and least for Humic rooting of 79.28 U/mg GST. |
ASCORBIC ACID PEROXIDASE (APX) ACTIVITY: |
| APX has a higher affinity for H2O2 (mM range) than CAT and POD (mM range) and it may have a more crucial role in the management of ROS during stress. APX activity was found to be highest in the commercially available Keradix GW04-KHA of 725.31 U/mg APX followed by coal GW04-CHA of 408.75 U/mg APX. The native plant activity was higher in BHA. It was found to be 620.85 U/mg APX and for native, it was found as 379.38 U/mg APX which is least than all the samples. The findings of [23] suggest that cytosolic APX1 plays a key role in protection of plants to a combination of drought and heat stress. Using Formula 7, estimation of APX was done. |
 |
| Table V revealed that APX specific activity was highest in Keradix GW04-KHA of 725.31 U/mg APX and least in Native plant of 379.38 U/mg APX. |
SUPEROXIDE DISMUTASE (SOD): |
| Superoxide anion is one of the most representative free radicals. In cellular oxidation reactions, superoxide radicals have their initial effects magnified as they produce oxidizing agents eg., radi hydroxyl radicals [9].The dosage of the extract is expressed as in μg of dry weight of the extract (compound) per ml of the assay mixture. IC50 value represents the concentration of test extract or compound where the inhibition of test activity reached 50%.The ethanolic extracts of GW04-CHA showed maximum decolourization at 41.34%, GW04-BHA showed maximum inhibition at 35.50%, GW04-KHA gave an inhibition of 35.03% respectively. The values were determined using formula (2). |
| The SOD activity was found to be inhibited highest in the coal sample of 41.34% than other samples. It was least in Keradix of 35.03%. |
CONCLUSION |
| The estimation of polyphenols in Humic rooting of native plants, was found to have higest activity of 146.2 μg/ml, the CHA had least activity of 54 μg/ml followed by Commercially available Keradix having 70 μg/ml. BHA of 104μg/ml of polyphenolic activity was estimated. The protein activity was highest in Humic Rooting sample (BHA) of 7 μg/ml followed by Coal and Keradix of 2.65 μg/ml and the least protein activity was found in the KHA of 1.2 μg/ml. The Catalase activity was highest for BHA sample of 0.09 U/mg catalase and least for Keradix (KHA) of 0.021 U/mg catalase. The GST activity was highest in coal sample of 582.99 U/mg GST than other three samples and least for Humic rooting of 79.28 U/mg GST. APX specific activity was highest in Keradix GW04-KHA of 725.31 U/mg APX and least in Native plant of 37938 U/mg APX. The SOD activity was found to be inhibited highest in the coal sample of 41.34% than other sample and was least in Keradix of 35.03%. |
ACKNOWLEDGMENT |
| The authors record their sincere thanks to the Management and Dr. M. Subbiah, Pricipal of Aringar Anna College (Arts and Science), Krishnagiri, Tamil Nadu, India for their unstinting support rendered towards us. We also extend our gratitude to Dr.V.Ravikumar, Assistant professor, Department of Biochemistry, Bharathidhasan University, Trichy for his valuable suggestions throughout this research work. Last but not least we thank Dr. V. Palani, Managing Director and Mrs. Manasa Satheesh, Director of Genewin Biotech, Hosur for providing necessary laboratory facilities to carry out this work in their DBT certified laboratory. |
References |
|