ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Pugazhendhi Sugandhi1, Durairaj Arvind Prasanth2*
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Diabetic foot infection is one of the dreaded complications of diabetes. Hence, proper management of diabetic foot infection requires appropriate selection of antimicrobials based on culture and antimicrobial susceptibility testing. This study was undertaken to evaluate the clinical and the microbial characteristic of diabetic foot infection in patients of Salem district, Tamil Nadu, India. The ulcers were graded and swab samples were collected under aseptic condition. A total of sixty patients with type 2 diabetics presented in this study. Out of sixty patients, fifty patients were culture positive and ten were found to be culture negative. Mono microbial infection was found to be higher than poly microbial infection. The bacteria isolated in this study included Staphylococcus aureus, Pseudomonas aeruginosa, Staphylococcus saprophyticus, Staphylococcus epidermidis, Micrococcus spp., Streptococcus pneumoniae, Pleisomonas spp., Bacillus spp. Escherichia coli, Klebsiella spp., Salmonella spp., Vibrio spp., and Enterococcus spp. MRSA and ESBL strains constituted 50 % in this study. Oxacillin and meropenem were found to be the best drug of the choice against both gram positive and gram negative organisms.
Keywords |
| Antimicrobial susceptibility, Bacterial profile, Diabetic foot ulcer, Staphylococcus aureus, Pseudomonas aeruginosa. |
I. INTRODUCTION |
| The diabetic foot infection is a global concern and develops in regions with improper foot care and increases the lifetime risk for developing foot ulceration [1]. This wound infection begins superficially, but with delay in treatment and impaired body defense mechanisms, can spread to the other subcutaneous tissues and to deeper structures ultimately leading to dreaded complications such as gangrene and amputations [2]. These infections are polymicrobial in nature. Escherichia coli, Proteus spp., Pseudomonas spp., Staphylococcus aureus and Enterococcus spp., are reported as frequent organism isolated from cases of diabetic foot infections [3]. The presence of MRSA and ESBL strains further worsen the prognosis and increase the risk of amputation [4]. Hence there arises the need to evaluate these infecting microorganisms on a routine basis in addition to administering regular glycemic control, wound care, surgical debridement, pressure-offloading and maintaining adequate blood supply [5]. So proper management of diabetic foot ulcer requires selection of appropriate antibiotics based on the culture and the antimicrobial testing. The microbial pattern of these diabetic foot infections have been studied widely from various regions indicating areaspecific studies for assessing the problem of these infections [6]. This study is one such to evaluate the clinical and the microbial characteristic of diabetic foot infection in the patients of Salem district, Tamil Nadu, India. |
II. RELATED WORK |
| In Southern India, with the available literatures the studies on diabetic foot infection are very meager. However, the results obtained in these studies have shown varying pattern of microorganism distribution and sensitivity pattern based on the severity of the infection and are usually contradictory. Though previous studies have showed predominantly gram-positive aerobe such as Staphylococcus aureus and gram negative organism such as Pseudomonas aeruginosa as the frequent isolates in these diabetic foot infections [7-12]. The choice of therapy is very much based on the nature of the infecting organism, its sensitivity pattern to the combination of antibiotics and will help the clinician in deciding the therapy for cases that are not responding for therapy [13]. |
III. MATERIALS AND METHODS |
| Collection and Processing of Sample |
| This prospective study was conducted involving diabetic foot infection patients of both sexes, clinically suspected of having microbial infections in their foot presenting at outpatient unit of tertiary care hospitals, Salem, Tamil Nadu from December 2013 to May 2014 for a period of six months. The institutional ethical clearance was obtained. The clinical history of the patients such as age, sex, types of diabetes, duration of diabetes, size of ulcer and duration of ulcer were recorded on a proforma. The ulcers were graded according to the Wagner’s grade classification [14]. A total ofsixty swabs were collected and processed for bacteriological investigations. The samples were processed by direct inoculation on to culture media like Sheep Blood agar (SBA), Brain Heart infusion Agar (BHIA) and Nutrient Agar (NA) incubated at 37°C for 24 hrs. The bacterial isolates were identified and confirmed according to the Bergey’s manual of Determinative Bacteriology [15]. |
| Antimicrobial susceptibility pattern |
| The antimicrobial susceptibility testing was performed as per the CLSI guidelines [16]. The antimicrobial discs included; Aztreonam (30μg), Amoxyclav (30μg), Cefpodoxime (10μg), Cefepime (30μg), Cefoperazone (75μg), Cefoperazone / sulbactam (75/10μg), Cefixime (5μg), Piperacillin (100μg), Ceftazidime/clavulanic acid (30/10μg), Ceftriaxone (30μg), Amikacin (30μg), Rifampincin (5μg), Meropenem (10μg), Cefoxitin (30μg), Ticarcillin/Clavulanic acid(75/10μg), and Piperacillin/Tazobactam (100/10μg) for the Gram negative organisms. Erythromycin (15μg), Methicillin (5μg), Chloramphenicol (30μg), Clindamycin (10μg), Vancomycin (30μg), Tetracycline (30μg), Oxacillin (1mcg) and Ciprofloxacin (5μg) were used for Gram positive organisms. All discs were obtained from Hi-Media labs, Mumbai, India. |
IV. RESULTS AND DISCUSSION |
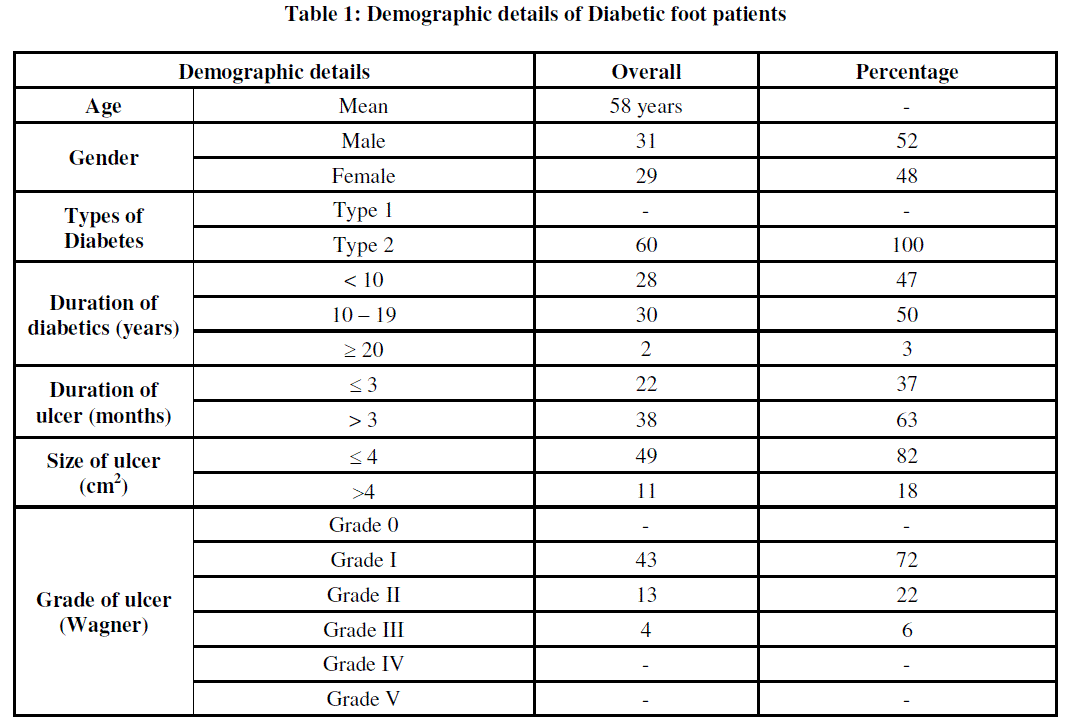
| Diabetic wound are injuries to the body tissues caused by physical trauma and proceed to deeper layers if untreated due to the high sugar level in the blood [3]. Although there are numerous studies with regard to the clinical profile and microbiology of these infections, but the magnitude of this problem often remains the same [17]. A total ofsixty patients with Type 2 diabetes presented in this study, which included male 31 (52%) and 29 female (48%). The mean age group was found to be 58 years. The duration of the ulcer ranged from 10 to 19 years and the enrolled cases were of Wagner’s grade I to III. In our study, 43 (72%) patients were Grade I, 13 (22%) patients were Grade II and 4 (6%) patients were Grade III (Table 1) |
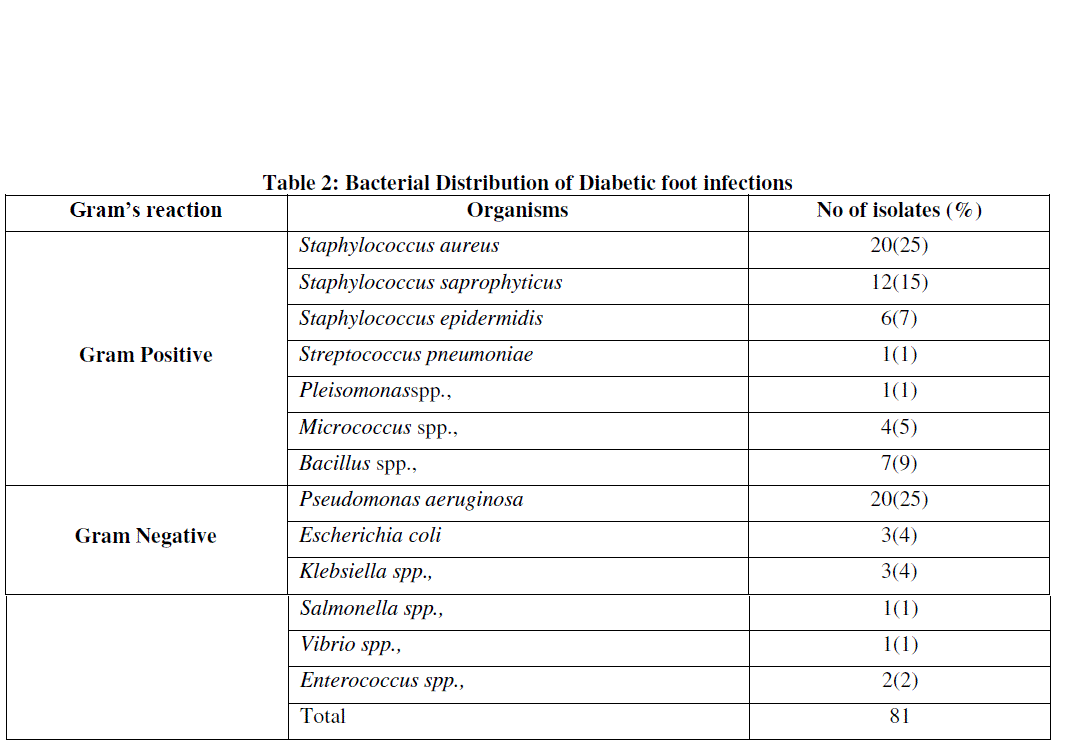
| Out of the sixty patients, 50 (83%) patients were positive for culture while 10 (17%) patients did not grow any organism. In that, 28 (56%) had mono microbial infection and 22 (44%) had polymicrobial infections. Mono microbial infections were in greater percentage than the poly microbial infection. The findings of the study is similar to the one reported by Dhanasekaran et al., [18]. A possible reason for such low incidence of poly microbial infection might be due to the role of severity of these infections [19]. Among the 50 culture positive cases, gram positive organism constituted 51 (63%) cases and the gram negative constituted 30 (37%) cases. Although in gram positive organism Staphylococcus aureus (25%) was the predominant isolate followed by Staphylococcus saprophyticus (15%), while Pseudomonas aeruginosa (25%) was the predominant isolate followed by Escherichia coli (4%) in gram negative organism. Gram positive organisms accounted to higher numbers than gram negative organisms, similar to the report of Abdulrazak et al., [20]. (Table 2) |
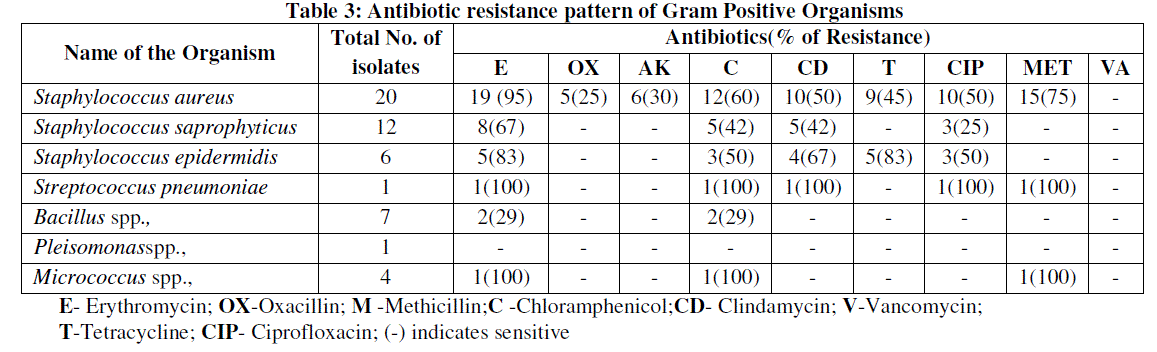
| Diabetic foot ulcers had a high frequency of colonization with antimicrobial-resistant organisms, including methicillin-resistant Staphylococcus aureus (MRSA) and extended spectrum beta-lactamase producing gram negative organisms (ESBL) [21]. This statement was similar to our study in which MRSA and ESBL isolates constituted 50%. The accuracy of the culture methods is important in the proper selection of antibiotics in the therapy of foot ulcers. The antimicrobial susceptibility pattern showed oxacillin and vancomycin as effective antibiotics against gram positive organism while meropenem, piperacillin, piperacillin/ tazobactam, ticarcillin/clavulanic acid and amikacin as effective antibiotic against gram negative organism. Among the combinations antibiotics, piperacillin/tazobactam was found to be the effective drug of choice that can be used for treating foot ulcers. This results was similar to the one reported by Tiwari et al., [6]. (Table 3 and 4) |
 |
 |
 |
 |
| * AT - Aztreonam; AMC - Amoxyclav; CPD - Cefpodoxime; CPM - Cefepime; CPZ - Cefoperazone; CFS - Cefoperazone / Sulbactam; CFM - Cefixime; PI - Piperacillin; CAC - Ceftazidime/clavulanic acid; CTR - Ceftriaxone; AK - Amikacin; RIF - Rifampincin; MRP - Meropenem; CX - Cefoxitin; TCC - Ticarcillin/clavulanic acid; PIT - Piperacillin/Tazobactam; ( - ) indicates sensitive |
V. CONCLUSION |
| In conclusion, the prevalence of gram negative infection was higher in diabetic foot patients from our region. Oxacillin and meropenem were found to be the best drug of the choice against gram positive and gram negative organisms. Among the combinations antibiotics, piperacillin/tazobactam was the best drug of choice that can be used for treating diabetic foot infections. Appropriate usage of antibiotics based on local antibiogram pattern can certainly help the clinician in reducing the burden of diabetic foot infections, which ultimately reduces the rate of amputations. |
References |
|