ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
V.Parkavi1 , V.S.Benitha2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Mixed metal oxides are synthesized from corresponding oxides and are used as pigments in paints which constitutes the Nanoformulated paints. The behavioural studies of these Nanoformulated paints are investigated in various aspects. The studies show better hiding power and enhanced glossiness. Nanoformulated paint coatings are done on mild steel plate and the coatings were tested in corrosion atmosphere. The results of corrosion tests were compared with commercially available red and white paint. Also the corrosion behaviour was studied for TiO2 and Fe2O3 formulated paint along with TiO2.Fe2O3. The mixed metal oxides TiO2.Fe2O3 shows better corrosion inhibition efficiency. The particle size of the prepared oxide nanoparticles are confirmed with SEM. The structural and functional purity of the oxides were confirmed with XRD and FTIR respectively. Furthermore the studies were extended by evaluating the photocatalytic behaviour of the prepared oxides. It is found that upon UV irradiation on the dye the photocatalytic degradation was significantly increased. This studies confirms that incorporation of these nanoparticles in paints enhances the photocatalytic activity which results in the reduction of volatile organic compounds
Keywords |
| Mixed Metal Oxides, Nanoformulated Paints, Glossiness, hiding power, Corrosion, Photocatalyticbehaviour |
INTRODUCTION |
| Nanotechnology is the science of developing materials by controlling the individual atoms and molecules to create devices that are thousand times smaller than the current technology. They are the structures between 1nm and 100 nm in size. They achieve ethical challenges. It has been applied to many areas of study including electronic engineering, physical sciences, biomedical sciences and many others. Most paints include at least four groups of components: binders, volatile substances, pigments, and additives. In order to increase the efficiency of TiO2 for photocatalytic activity is proposed to use mixed metal oxides to enhance photocatalytic behavior. The oxides such as Fe2O3 was doped with TiO2 to form mixed metal oxides such as Fe2O3.TiO2. These nanoparticles are incorporated into paints to enhance the photocatalytic behavior hence reducing the volatile organic compounds contained in them. Furthermore, glossiness, hiding power and anticorrosion behavior are determined. |
II.MATERIALS AND METHODS |
2.1 Preparation of Metal Oxide Nanoparticles |
| The metal oxides such as TiO2, ZnO, NiO, Fe2O3are prepared by SOL-GEL process. A colloidal suspension or a sol is formed due to hydrolysis and polymerization reaction of the precursors, which on complete polymerization and loss of solvent leads to the transition from liquid sol into a solid sol phase |
2.2 Preparation of Mixed Metal oxide Nanoparticles |
| The prepared metal oxides are taken individually in a beaker and distilled water is added. Based on their combination such as TiO2.Fe2O3 two metal oxides are taken combined, heated in a hot plate at 60°C.Then it is washed, filtered and dried. |
2.3 Preparation of Nanoformulated based paints |
| The paints are synthesized by mixing the composition of pigment, resin, solvent and additives in a equal proportion |
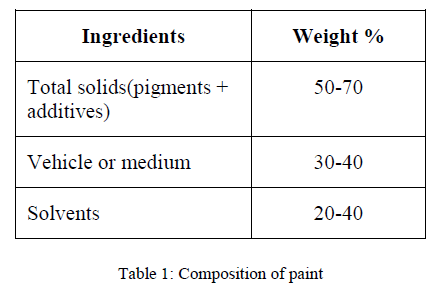 |
2.4 Gloss Test |
| Gloss test is required to ensure uniformity of the surface finish. The gloss value is determined by directing a light, at the test surface and measuring the amount of specular reflection. Gloss is measured at 60°. |
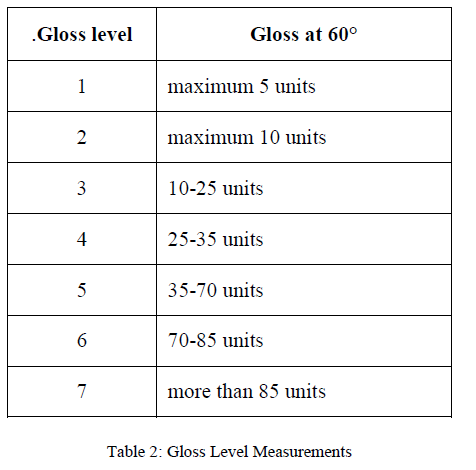 |
2.5 Hiding Power |
| Opacity and hiding power measurement was done using quadruplex film applicator. It act as Multifunctional film applicator with 4 application sides for applying paintfilms. It is placed near one end of a flat panel, like a test chart. A sufficient volume of paint/liquid is placed in front of the applicator. The applicator is then drawn down in the chart manually, which leaves a uniform film behind it. |
2.6 Anticorrosion Studies |
| Corrosion is the destructive attack to metal by chemical or electrochemical reaction with its environment. Mild steel was easily corroded in acidic medium. To reduce this problem the corrosion inhibitor coating was done on it. A corrosion inhibitor is the material which inhibits the corrosion reaction by providing a protective barrier film which in turn stops the corrosive reaction. Paint is a material which prevents the direct contact of corroding media like air and H2O over the metal surface. It forms a uniform thin layer after drying and protects the base metal from the corrosion |
2.7 Photocatalytic behaviour studies |
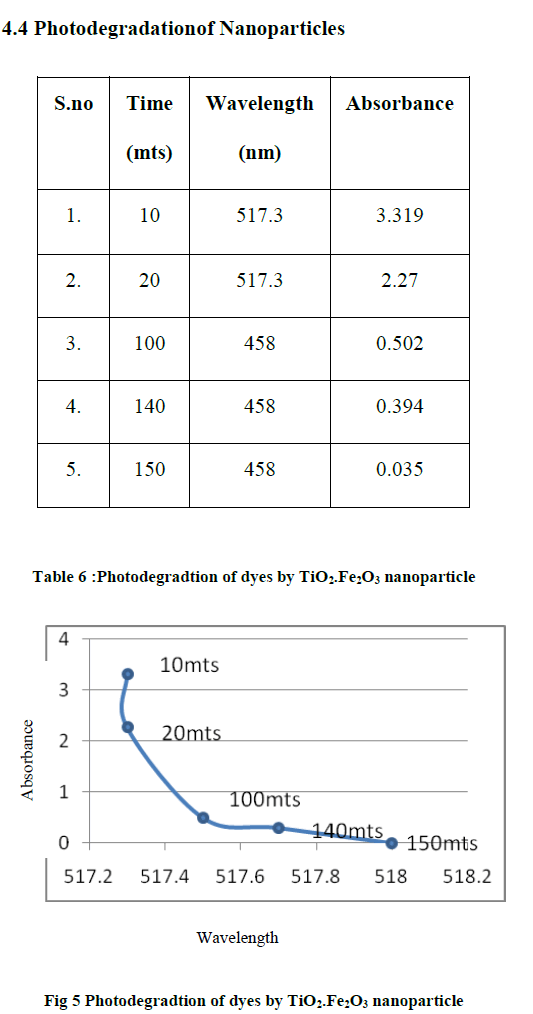
| The photocatalytic behaviour in paints is studied under UV light.The photocatalytic degradation is due to absorbance of the peak. TiO2 is a photocatalyst when it is illuminated by light of energy higher than its band gap, electrons in TiO2 will jump from valence band to conduction band and electrons and holes will form on the surface of the photocatalyst. The negative electrons and oxygen will combine to form radical ions whereas positive electric holes and water will generate hydroxyl radicals OH-Since both products use unstable chemical entities when the organic compound falls on surface of photocatalyst it will combine with O2 - and OH and turns to form CO2 and H2O. |
 |
III. CHARACTERISATION STUDIES |
3.1 XRD analysis |
| The structural features of TiO2.Fe2O3 is explored from XRD data. In Fig 3.1 the XRD pattern of final powders revealed well developed reflection of TiO2.Fe2O3 nanoparticles (JCPDS.No 89-8104, 89-6975, 89-4920.). |
 |
 |
| This figure reveals the presence and uniformity of the distributed particles. It was clear that the particles obtained were in nano size ranging in the diameter from 150- 200nm. |
 |
| It represents the 3D AFM images of the TiO2.Fe2O3 coatings. The shape and thickness of the coated particles is around 80nm and it is uniformly distributed. |
 |
| FT-IR spectrum TiO2nano particles (fig 5.10) showed significant absorption peaks at 990cm-1&1648cm-1.The absorption band 990cm-1 was assigned to Ti-O stretching vibration. The weak band near 1648cm-1is assigned to H-0- H bending vibration |
IV. EXPERIMENTAL RESULTS |
4.1 Glossiness |
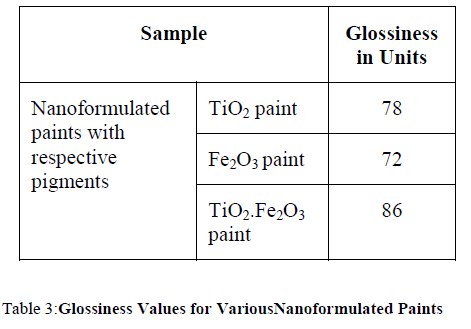 |
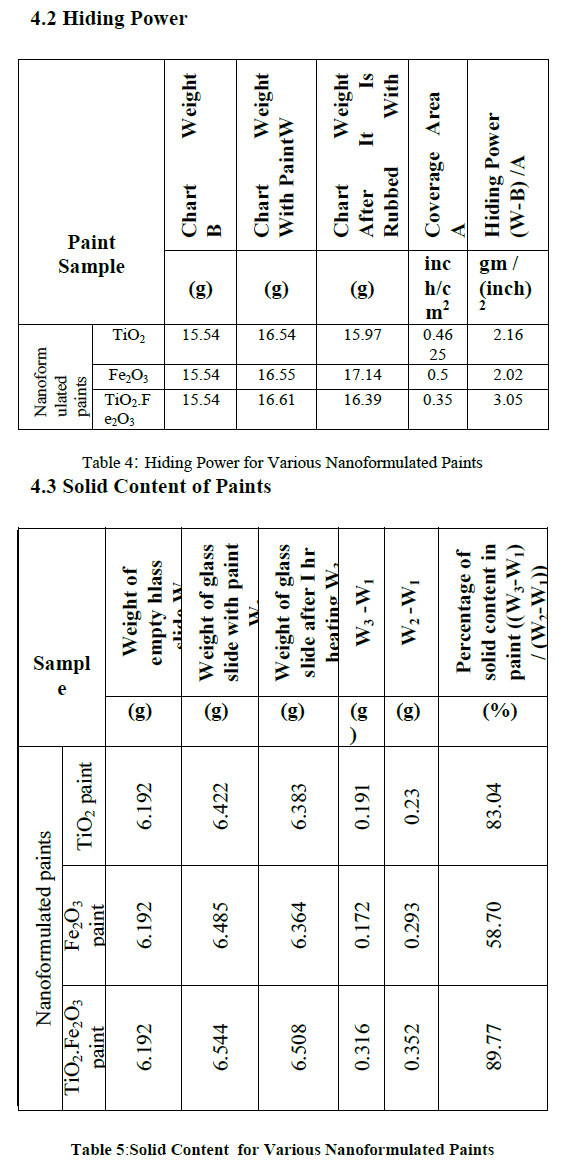 |
 |
 |
V. CONCLUSION |
| Nanoformulated paints such as titanium oxide, iron oxide and mixed oxides were prepared. The properties for both nanoformulated paints and commercially available paints are analysed. Glossiness, SolidContent and Hiding power of Nanoformulated paints are higher when compared to commercial paints. Corrosion behaviour is low in Nanoformulated paints and therefore it has high inhibition efficiency. |
ACKNOWLEDGEMENT |
| This work was supported by Centre for nanoscience and Technology, MepcoSchlenk Engineering college, Sivakasi and Vigneshwara paint company ,Sivakasi. |
References |
|