ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Dr. K. Venkatesha Raju1, Dr. R. K. Somashekar2 and Dr. K. L. Prakash3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The study focused on assessment of metal accumulation in certain aquatic macrophytes and benthic organisms to be used as biomonitors. The macrophytes and benthic organisms collected during April to May (2011) from river Cauvery, one of the main fresh water aquatic systems in the south of India. Microwave digested samples of discrete organs of macrophytes and benthic organisms were analyzed for metal concentration using AAS-6300. In macrophytes, among the eight metals investigated, six (Fe, Pb, Ni, Cr, Cu, and Cd) were showed higher degree of accumulation in root whereas, remaining (Mn and Zn) in stem and leaf respectively. Vallesnaria spiralis showed greatest potency to acumulate Fe, Mn and Cu, while Nymphe stellata showed the better ability to accumulate Zn and Cr. Cd was found only in Aquatic neam and Vallisnaria spiralis at downstream stations with very negligible concentration. Based on the accumulation level in macrophytes, the eight metals were arranged in the order of Fe> Mn> Zn> Cu> Pb> Cr> Ni > Cd. Among benthos, mollusca have showed a more accumulation capacity fallowed by crustacean and annelid. Bioaccumulation factor for benthos were observed in the sequence of Cu>Mn>Zn>Ni>Fe>Cr>Pb>Cd.
Keywords |
| Heavy metal, Macrophytes, Benthos, Crustacean, Biomonitoring. |
INTRODUCTION |
| Aquatic macrophytes are unchangeable biological filters and they carry out purification of the water bodies by accumulating dissolved metals and toxins in their tissue (Lilit and Baban, 2006). Many of the macrophytes found to be the potential scavengers of heavy metals from aquatic environment and are being used in wastewater renovation systems (Abida, 2009, Nirmal et al., 2006). The biomonitoring of heavy metals using macrophytes has several advantages and is the most significant in the study of sublethal levels of bioaccumulated metals within the tissues of organisms, which indicates the net amount of pollutants integrated over a period of time. It provides time integrated information about the quality of the aquatic system (Ravera et al., 2003; Baldantoni et al., 2005). Several investigators have revealed that aquatic benthic organisms are better accumulator of metal than ambient (water) and are good bioindicator of Heavy metal pollution in the river ecosystem (Kiffney & Clements, 1993). The Crabs and Molluscs are detrivorous, sedentary and do not migrate. Therefore, their weak metabolizing systems can accumulate higher amounts of heavy metals. (Falusi and Olanipekun, 2007) The fleshy tissues of the crab are good accumulator of heavy metals and the nutritional implication of this is that consumers of the animal may be exposed to heavy metal toxicity and bioaccumulation results due to regular consumption (Hsiao-Chien, 2009). Molluscs are capable of achieving tissue concentrations of metals that are 100 to 1000 times higher than the background concentrations (Oyedepo, et al., 2007). Gopinathan and Sobhana, (2003); Ali and Fisher, (2005) revealed that molluscs are useful indicators of the abundance and spatial distribution of metals in aquatic ecosystems. The investigations on the heavy metals accumulation in the Karnataka stretch of Cauvery river system is not frequent and have probably no studies on metals in macrophytes and benthos in were reported. Hence, the present study has greater significance; especially as the water from river is being used for drinking and irrigation at larger extent. |
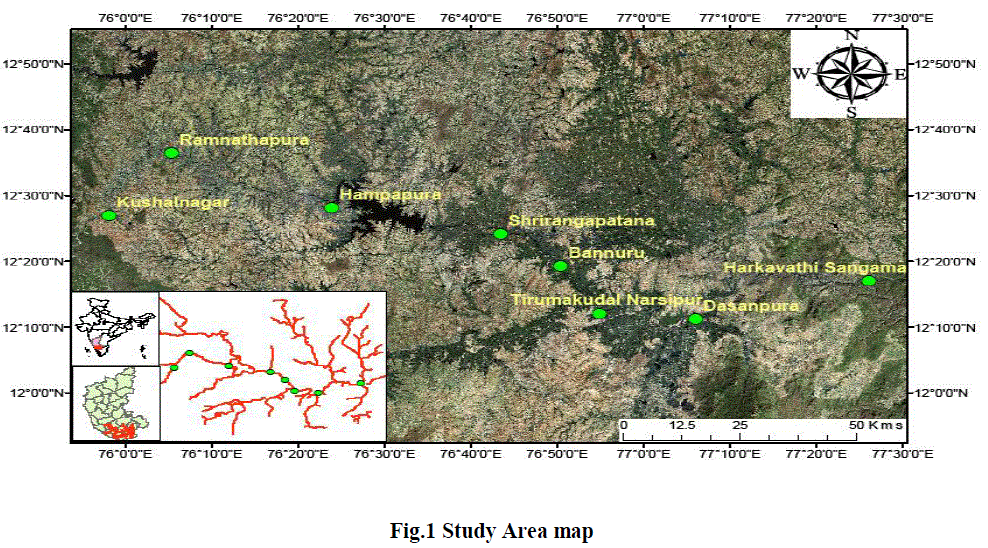
STUDY AREA |
| This investigation was carried out in river Cauvery from Kushalnagara to Arkavathi Sangama of Karnataka stretch (Fig.1) during dry season of April-2011. Three upstream and five downstream to Krishnaraja Sagar reservoir collectively eight stations were chosen as sampling sites for effectual research. |
METHODOLOGY |
| A. Macrophytes Sampling, Preservation and Microwave digestion: Sixteen macrophytes belong to four species from eight stations were collected by hand once during the study period according to the nature of plant (floating or submerged). Floating macrophytes were collected from 1.0 X 1.0 m quadrant, while the submerged plant species were collected using a known volume of grab sample and washed with the surrounding water to remove the periphyton and sediment accumulated on them. Plastic bags were used to store the samples. These samples were thoroughly washed once again in the lab with distilled water to remove adsorbed elements and cut into small pieces of leaf, stem and root separately, air dried for 2days and dried at 100±10C in an hot air oven for 4hr. Dried samples were grounded to powder and passed through a 1mm sift. |
| A powdered plant samples of 0.5g was introduced into the reference vessels, 4ml HNO3 and 0.2ml H2O2 were added, and the carousel was positioned into the microwave unit. The system was pre-programmed for 1 min of microwave digestion at 250 W power and another 5 min at 500 W power and left for automatic ventilation for 10 min. The digested solution was cooled, filtered using Whatman filter paper No. 40, and made up to 100 mL with Milli-Q distilled water and stored in containers until analysis. |
| B. Benthic/Bottom dwellers Sampling, Preservation and Microwave digestion: Twenty four samples of benthic organisms including crab (Crustacean), mussel (Mollusca) and cheatogaster (Annelida), one each from eight stations sampled during dry season because of less flow rate and low depth. Crabs were collected directly, by hand picking, from their holes on the bank of river with the help of localites. The crabs were kept in a bucket containing river water and transported to the laboratory until dissection. The crabs were washed with tap water, and rinsed thoroughly with distilled water, fleshy tissues was dissected out from the chest and the appendages using surgical knife and placed on clean watch glasses. It was oven dried at 1050C for 1h and cooled in a desiccator (Faulsi and Olanipekun, 2007).The mussel and cheatogaster were sampled using nylon fish net. The samples were sieved through a 0.5 mm sieve and encrusted algae were scraped from shells (Locarnini and Presley, 1996). Samples were rinsed, dried at 50 °C in an oven and homogenized. Mollusca and crustacean species were removed from their shells during sample preparation but Cheatogaster were analyzed without modification. |
| 1g dry weight of the sample was introduced in to teflon reaction vessel and 5mL of 65% Nitric acid was added. The vessel was inserted into microwave chamber and subjected for digestion for 5 minute at 240W; 1 minute at 360W; 5 minutes at 300W, cooled and the digester was reopened. Again 1mL of 30% Hydrogen peroxide was added and power set to 300W for 3 minutes, 600W for 3minutes, cooled and the digester was reopened. Further 1mL of 20%Perchloric acid was added and power set to 600W for 2 minutes, cooled and the digester was opened. The resulted sample was cooled and appropriately diluted using deionised water. |
| C. Analysis: Atomic absorption spectrophotometer (AAS-6300, Shimatzu made) with Wizard software was used to estimate the heavy metals viz., Fe, Pb, Zn, Mn, Ni, Cu, Cr, Co and Cd. Two types of atomizers viz., flame and graphite furnaces were functioned to evaluate the metal concentrations at ppm and ppb levels respectively. The sample whose metal concentration was below the detection limit in flame furnace was subjected to graphite furnace analysis. |
 |
RESULT AND DISCUSSION |
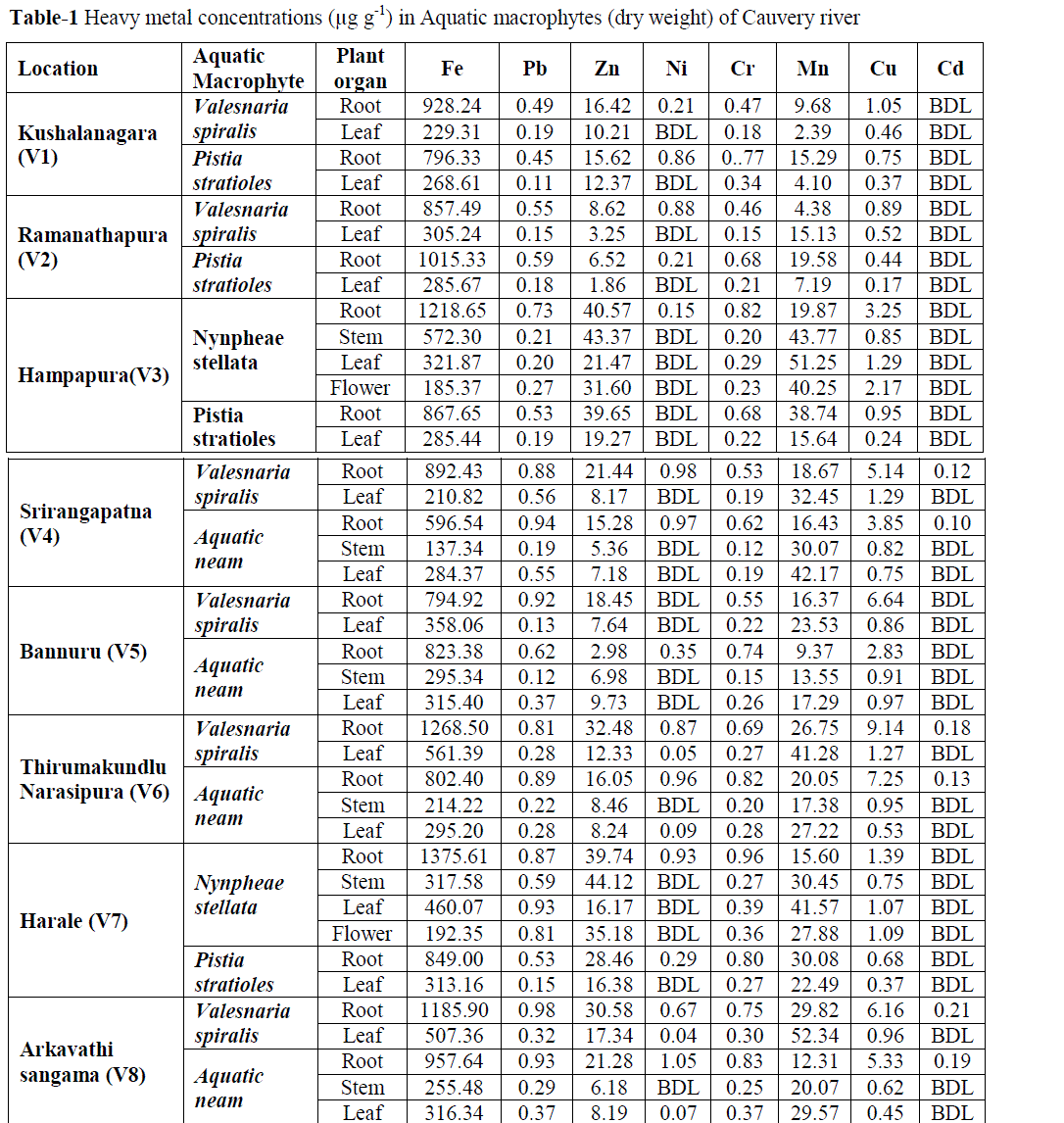
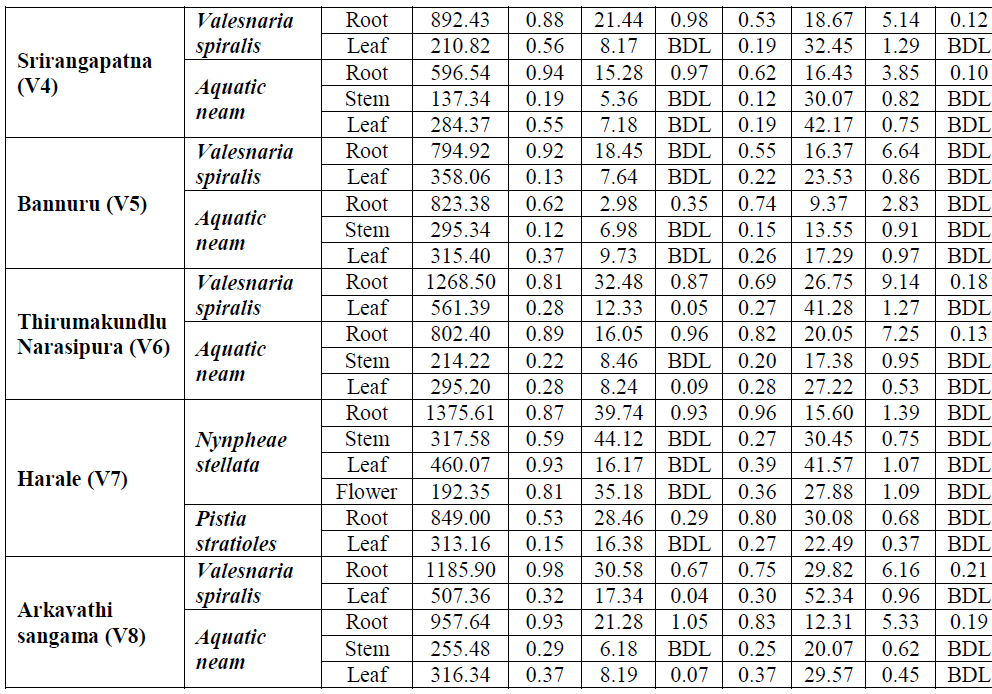
| A. Metals in Macrophytes: The concentration of heavy metals in macrophytes was presented in Table-4.9. In general concentration of Fe was observed more (1375.6 μg g-1) in roots of Nymphae stellata from station V7 whereas, Aqatic neam stem from station V4 showed lesser accumulation of 137.34 μg g-1. Highest concentration (0.98 μg g-1) of Pb was found to be accumulated in Vallisnaria spiralis roots from station V8. While, Pistia stratioles leaf from station V1 recorded lowest concentration (0.11 μg g-1). Zn was observed highest accumulation (44.12 μg g-1) in stem of Nymphae stellata, from station V7 and lower concentration (1.86 μg g-1) in Pistia stratioles from V2 station. Among the macrophytic organs, only root of all the species showed considerable Ni accumulation with highest concentration (1.05 μg g-1) in Aqatic neam of station V8. Nymphae stellata root from V7 station recorded maximum concentration of Cr (0.96 μg g-1) whereas, lowest concentration (0.12 μg g-1) for Aqatic neam stem from V4 station. Maximum Mn concentration (52.34 μg g-1) was observed for Vallisnaria spiralis leaf sample of station V8 and minimum concentration (2.39 μg g-1) for leaf of the same plant from station V1. Root of Valisnaria spiralis from stationV6 showed highest Cu concentration (9.14 μg g-1). In contrast lowest (0.17 μg g-1) was observed for Pistia sratioles leaf sample from station V2. Trifling concentrations of Cr was observed in all the macrophytes except Pistia stratioles. |
| Among the eight metals investigated in macrophytes, six (Fe, Pb, Ni, Cr, Cu, and Cd) were accumulated at highest rate in root, whereas remaining Mn and Zn in stem and leaf respectively (Table-4.9). A plant with numerous thin root systems would accumulate more metals than one with few thick roots. Factors such as light intensity, oxygen stress and temperature are known to affect the uptake of minerals (Devlin, 1967). Moreover, the energy derived from photosynthesis and the oxygen released can improve conditions for the active absorption of elements. However, interactions between metals are often complex, and they are dependent on the metal concentration and pH of the water body (Balsberg-Pahlsson, 1989).Further no such spatial variation was found in metal accumulation by macrophytes. Even though, downstream stations (V4-V8) managed to accumulate moderately elevated metal concentrations than upstream stations (V1-V3). |
| From the present annotations it is documented that there is a uniform pattern of heavy metal variation in the macrophytes of the river Cauvery (Table- 4.9). However the accumulation of a particular metal depends to a large, on the presence of the metal in the water column and sediment (Lilit and Baban, 2006). Among macrophytes, Vallesnaria spiralis shows the greatest potential to accumulate Fe, Mn and Cu up to 914.94 μg g-1, 41.08 μg g-1 and 5.20 μg g-1 respectively. Nymphe stellata showed the better ability to accumulate Zn (34.25 μg g-1) and Cr (0.49 μg g-1). The role of Zn and Mn in respiration as activators of enzymes involved in glycolysis and Krebs’ cycle has been documented. High Cu content is known to reduce photosynthesis and respiration rates in aquatic macrophytes (Vazguez et al., 2000). Cd was almost absent in all macrophytic samples. However, very negligeble Cd concentration was noticed in Aquatic neam and Vallisnaria spiralis, of downstream stations. The presence of Cd in the macrophytes suggests the possible accumulation of cadmium in river bed and suspended sediment (Zakova and Kockova, 1998). When arranged according to their accumulation, the following order was observed for the eight elements - Fe> Mn> Zn> Cu> Pb> Cr> Ni > Cd. The effects of trace elements in an aquatic ecosystem can be assessed by changes in the community structure, physiological activity and ultra structural components of macrophytes (Patricia et al., 2004). The concentration of highly toxic metals like – Cr, Cd and Pb were lower compared to the essential metals like Fe, Mn, Ni and Zn.. The low availability of heavy metals in the river system could be due to the flow and its rate compare to stagnant lake system (Arnulf, 1999). |
 |
 |
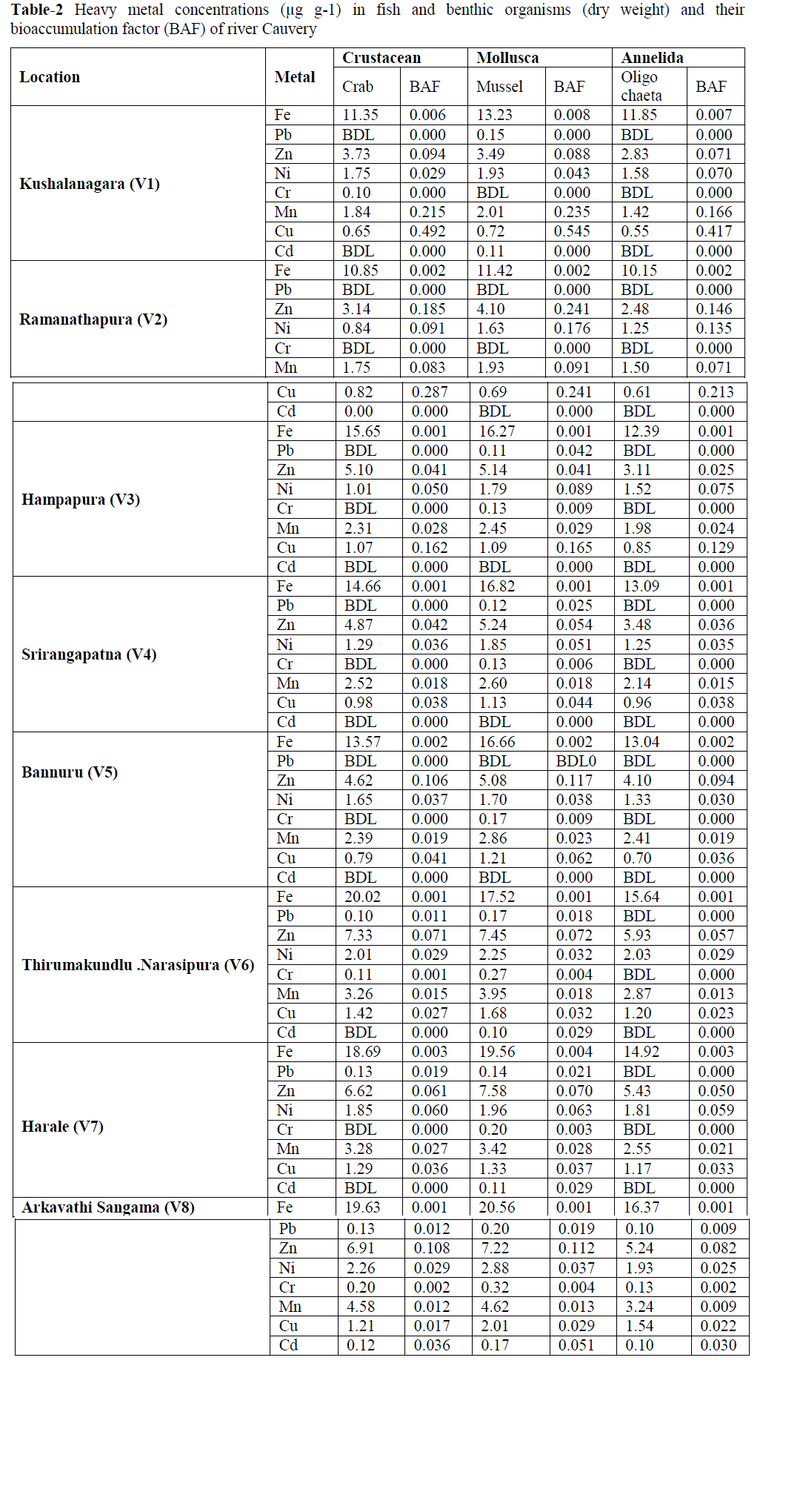
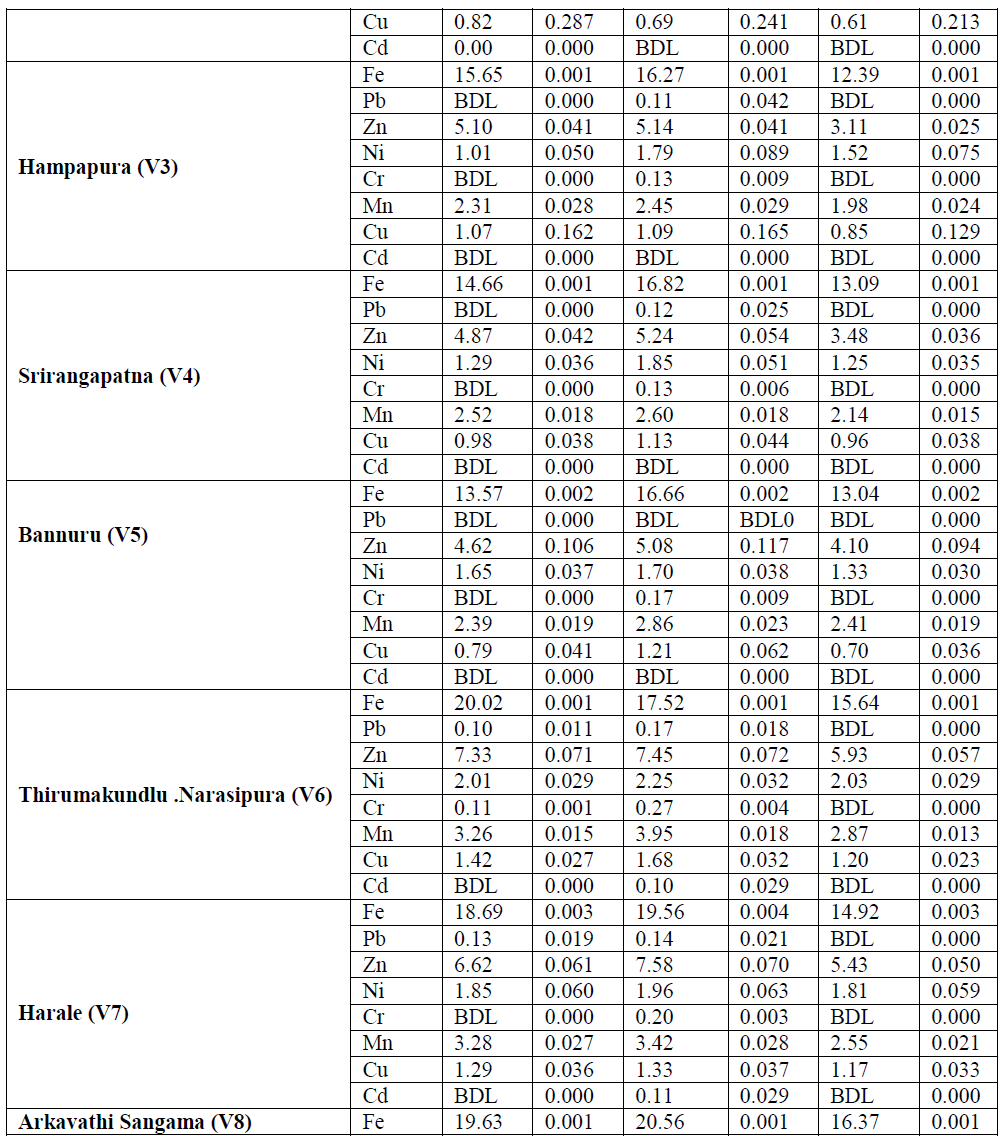
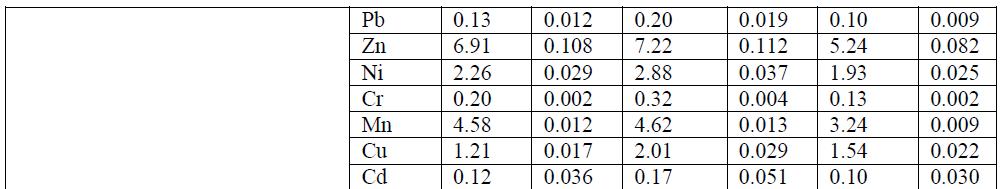
| B. Metals in Benthos: The concentration of heavy metals in benthos was presented in Table-2. Fe and Mn showed a similar distribution profiles which suggests that these metals were derived from the same source. Fe content in the benthos showed their highest values (20.56 μg g-1) for mollusca and lowest value was recorded (10.15 μg g-1) for annelida. Mn concentration showed to more extent a similar distribution and spatial trend as iron for all benthos with highest value for mollusca (4.62 μg g-1) and lowest for annelida (1.42 μg g-1). Zn stands next to the iron in terms of benthos accumulation. As essential element, Zn content among the benthos has not expressed much variation. However, spatial distribution was appeared to be at substantial extent. Comparatively Zn concentration was high in mollusca (7.58 μg g-1) followed by crustacean (7.43 μg g-1) and annelida (5.93 μg g-1). Cu concentration was elevated towards the downstream stations. Highest concentration was recorded for mollusca (2.01 μg g-1) and lowest for annelida (0.55 μg g-1). The concentrations of Ni in mollusca are usually higher than that of crustacean and Annelida in all sampling localities (Table-4.10). The values varied from 1.63 to 2.88 μg g-1 (mollusca), 0.84 to 2.26 μg g-1 (crustacean) and 1.25 to 2.03 μg g-1 (annelida). Negligible extent of Cd concentrations were observed for all the benthos with highest value (0.32 μg g-1) for mollusca of station V8. In case of annelida only station V8 has showed Cr accumulation at very lesser extent. Station V2 of the study area has not recorded chromium content in any benthos samples. |
| It is evident that, as the nonessential elements Pb and Cd accumulation in benthos was very insignificant. Mollusca collected from downstream station (V8) showed highest concentration for Pb (0.20 μg g-1) and Cd (0.17 μg g-1). In annelida except station V8 all other exhibited below detection limit (BDL). Downstream stretch of the study area usually has high concentrations of most metals. Such differences were apparent for almost all metals. These differences may well be attributable to changes in anthropogenic input of metals or to changes in physicochemical factors which affect the uptake of many trace metals (Rainbow, 1986). From the present investigation, it is obvious that mollusca have showed comparatively higher accumulation capacity among the benthic organisms for almost all the metals followed by crustacean and annelida. Similar results have reported by Mohamed (2005) in their studies on benthic invertebrates. Molluscs have the ability to filter large volume of water, uptake and accumulate metals in their bodies without noxious effects (Metcalfe- Smith et al., 1994; Byrne and Vesk, 2000). The results of current study indicate that mollusca have the highest concentrations of most trace metals measured. This accumulation of several metals is due to the low capacity of the mollusca for discriminating among metals which are similar in some characteristics such as ionic radius (Metcalfe-Smith, 1994). Mollusca also possess a variety of effective detoxification mechanisms to reduce the toxicity of the metal uptakes (Byrne and Vesk 2000). |
| C. Bioaccumulation factor (BAF): The bioaccumulation of all eight heavy metals in benthic organisms in the river Cauvery was quantified with bioaccumulation factor (Table-2). The BAF elucidated that increase trend from upstream to downstream for all the metals i.e., the downstream of the river has high values of most metals which can be explained on the basis of anthropogenic pollutants income to the Cauvery in the downstream stretch. Also, the BAF values showed elevated trend in case of essential trace metals (Fe, Mn, Zn, Ni and Cu), but an obvious decrease in nonessential trace metals (Pb and Cd). BAF for benthos in the study area is in the sequence of Cu>Mn>Zn>Ni>Fe >Cr>Pb>Cd. The values of BAF for different heavy metals from water to benthos or sediment to benthos are a key component of human exposure to the metal through food chain. The lowest BAF value in this study is for Cd; one of the likely reasons is being that it is more mobile in the natural environment than the other metals. |
 |
 |
 |
CONCLUSION |
| This study concludes that Vallesnaria spiralis showed significant biomonitor for the trace metals in the fresh water river system and accumulation is in the order of Fe>Mn>Zn>Cu>Pb>Cr>Ni>Cd. Among benthos mollusca have showed more accumulation capacity fallowed by crustacean and annelida. Bioaccumulation factor for benthos observed in the sequence of Cu>Mn>Zn>Ni>Fe>Cr>Pb>Cd. Elevated trend was noticed among the essential metals and decreased trend among the non essential metals in both macrophytes and benthos. Samples from downstream stations with high degree of metal accumulation compare to upstream stations revealed odds of pollutants from nonpoint sources too. |
ACKNOWLEDGEMENT |
| The authors would like to thank Natiional River Conservative Directorate (NRCD), The funding agency, Ministry of Environment and Forest (MoEF), New Delhi for providing invaluable financial support to conduct research on water quality of river Cauvery. |
References |
|