e-ISSN: 2319-9849
e-ISSN: 2319-9849
University of El Oued, VTRS Laboratory, B.P.789, 39000, El Oued, Algeria
Received: 05/03/2013; Accepted date: 13/03/2013; Published date: 25/03/2013
Visit for more related articles at Research & Reviews: Journal of Chemistry
Through this work we adopted Rotating Disk Electrode (RDE) Voltammetry to study the kinetics of oxidation and the effect of hydrazide group on ferrocene in organic medium. Thus, two different electrodes (Pt and Gc) were used in ordre to determine this latter. According to the ferrocene taken as a witness the hydrazide group related to the ferrocene made oxydation more difficult. This ferrocenic derivative showed an electrochemical stability, a reversible electrochemical system and an electronic attractor effect of these substitutional ferrocene groups. Finally, we calculated some electrochemical parameters which were: the diffusion coefficients (D), the layer thickness in addition to the electron transfer rate.
Rotating Disk Electrode (RDE), diffusion coefficient, ferrocene derivative, half-wave potential, Randles-Sevcik equation.
Rotating disk electrode is a hydrodynamic electrode technique which utilizes convection as the mode of mass transport as opposed to CV which is governed by diffusion. Thus a comparison of the kinetic parameters obtained from CV and RDE experiments is informative to elucidate the role of mass transport on electrode reaction kinetics.
In kinetic studies of electrode processes, uniformity of the concentration gradients along the electrode surface and quantitative information concerning these gradients are necessary. In many instances the rotating disk electrode technique provides an effective means for realizing these requirements. Furthermore this technique allows the surface concentration of reactants and products to be varied in a controlled manner through changes in the rotation rate and hence can be used to determine the reaction orders through the dependence of the current on the rotation rate without the necessity of varying the bulk concentrations.
Many studies and analysis by electrochemical methods were effected on the oxido-reducing properties of ferrocene. In general, the cathodic behavior ferrocene in an organic media such as dichorométhane, acetonitrile and DMF can be described by a reversible reduction in an electron, leading to ion Ferrocerium [1,2]. In the present work the oxidation of ferrocene, Fe(C5H5)2, to the ferrocenium cation, Fe(C5H5)2+, was examined in the solvent dichloromethane solution containing tetrabutylammonium tetrafluoroborate, using RDE. The results indicated that redox reactions of ferrocene/ ferricenium couple were a reversible process of diffusion-controlled single electron transfer in the studied solution [3,4].
One of the ferrocene derivatives the compound N'-Ferrocenylmethyl-N'-henylbenzohydrazide is a very important electron-transfer system for molecular electronics owing to its characteristic redox behaviors [5,6], and it could also be expected to play a key role of an electron chemical probe of the electron-transfer process in biological molecules [7,8].
It is well known that N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide easily undergoes one electron oxidation to form ferrocenium cation in a reversible manner [9,10] figure (1). Thus, we investigated the electrochemical N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide behaviors in organic media.
Instrumentation and software
Cyclic voltammetric measurements were performed using PGZ301 potentiostat (radiometer analytical SAS) and a voltammetric cell with a volumetric capacity of 25ml containing a glassy carbon electrode (GCE) working electrode (radiometer analytical SAS), a Pt wire counter electrode, and an Hg/Hg2Cl2reference electrode (3.0M KCl). Solutions were deoxygenated with high purity nitrogen for 3 min prior to each experiment. Data acquisitions were accomplished with a Pentium IV (CPU 3.0 GHz and RAM 1 Gb) microcomputer using VoltaMaster software version 7.08 (radiometer analytical SAS). Graphs plot and calculus were carried out using OriginLab software version 2.0 (Integral Software, France).
Chemicals
Electrochemical characterization was carried out on a potentiostat type voltalab 40 of radiometer, with a three-stand electrode cell. Cyclic voltammetric experiments were performed in deoxygenated CH2Cl2 solution of N'-Ferrocenylmethyl-N'- Phenylbenzohydrazide with respectively 10-1 M of Bu4NBF4 as supporting electrolyte and N'-Ferrocenylmethyl-N'- Phenylbenzohydrazide concentration of 10-3 M. The three electrodes used were glassy carbon and Platinium disk as the working electrodes, saturated calomel electrode as a reference electrode, and Pt wire as an auxiliary electrode. The working electrode was polished with 0.05 μm alumina slurry for 1–2 minutes, and then rinsed with double-distilled and deionized water. This cleaning process is done before each cyclic voltammetry experiment.
The N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide was synthesized according to literature procedures [7] . Fig. 2 (A,B) shows RDE Polarogrammes for ferrocene and N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide at a series of rotation rates. It is evident from the data that the current generated by the RDE method is much larger than that generated under diffusion control. The much larger current that was obtained using RDE, reflects the efficiency of this method.
The diffusion current limit, the current half-wave and half-wave potential are calculated at different rotation speed of the two electrodes, Table 2.
Calculation of diffusion coefficient
The Levich equation predicts the current observed at a rotating disk electrode and shows that the current is proportional to the square root of rotation speed. The equation is:
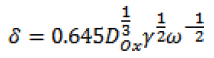
Where Dox: diffusion coefficient of the oxidant is expressed in cm2.s-1
Where Dox: diffusion coefficient of the oxidant is expressed in cm2.s-1
ω: rotational speed of the electrode (rad s-1)
γ: kinematic viscosity in cm2. s-1
Kinematic viscosity: is the ratio of the viscosity on the density, we have for dichloromethane:
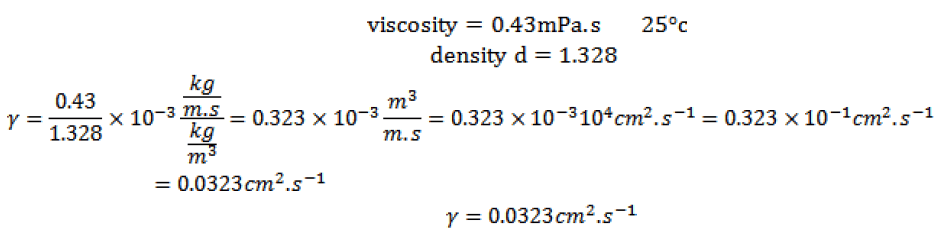
The kinematic viscosity (≈ 10-6 m2 s-1, for an aqueous solution at 25 ° c)
The relationship between i and the square root of rotation speed 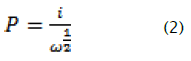
On another hand the limited current is given by, 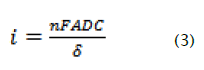
Whereas: n, number of electrons
F: is the Faraday (9.65.104 C/mol)
A: is the area of the working electrode (cm2).
D: is the coefficient diffusion (cm2.s-1)
C: is the concentration (mol/cm3), in our case is equal to 10-3 mol/l
Replacing equations 2 and 3 in 4 gives,
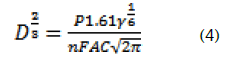
Applications
For a rotating rate of the working electrode equal to 400 t/min., the coefficient diffusion of N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide in dichlormethane is.
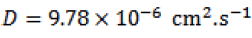
The coefficient diffusion of ferrocene in aqueous ethanol is calculated as above. Table 2 summarize the obtained values.
Voltammetry analysis on a RDE of N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide in an organic solution indicates that the electrochemical reaction of N'-Ferrocenylmethyl-N'-Phenylbenzohydrazide in the studied solution is a diffusion controlled process using two different electrodes. The layer thickness δ at ethe (GC) electrode was thicker than δ at the (Pt). The same was observed in the coefficient diffusion.
This ferrocenic derivative showed an electrochemical stability, a reversible electrochemical system and an electronic attractor effect of these substitutional ferrocene groups.
The authors gratefully acknowledge Mr A. Khelef Maitre assistant at The University of El Oued for his help and advice, and the Technical staff in the laboratory of VPRS for its support.