ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Manohara B. 1, Belagali S.L.2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
In this paper the composting process was studied for Municipal Solid Wastes in order to characterize the essential plant nutrients and heavy metals during the degradation process. The process was studied in pre-monsoon, monsoon and post-monsoon seasons and samples were collected during 10th to 60th days of composting process. Primary macronutrients like nitrogen, phosphorus, potassium, secondary macronutrients calcium, magnesium and micronutrients/trace minerals like chlorine, manganese, iron, zinc, copper, molybdenum, nickel were analyzed. Heavy metals like lead, cadmium, chromium, were analyzed by atomic absorption spectrophotometer. From the study, the concentrations of essential plant nutrients were found to be under the limits of Ohai- EPA standards and Canadian Council of Ministers of the Environment (CCME) standards. Heavy metals were also found in trace quantities and humification process caused decrease in heavy metal concentration. From the present study, it was observed that composting process was faster during monsoon season and compost produced was better source of plant nutrients.
Keywords |
| Solid waste, Essential nutrients, Heavy metals, Composting, Ohai- EPA and CCME standards. |
INTRODUCTION |
| Management of solid wastes is one of the critical issues for the urban areas of developed and developing countries, since rapid growth of population in urban area is taking place due to migration of rural population in search of better livelihood. Advancement of civilization enhanced the usage of consumer products and manufacturing processes. Usage of vast range of products leads to generation of large quantity of wastes in cities. The lack of proper management of solid wastes leads to air and water pollution which leads to health and environmental impacts. The overall goal of urban solid wastes management is to collect, treat and dispose off solid wastes generated by urban population in an environmentally and socially satisfactory manner using the most economical available means. Municipal solid waste, commonly known as trash or garbage, refuse or rubbish is a waste consisting of everyday items that are discarded by the public. It is classified into Domestic wastes, Commercial wastes, Institutional wastes and Municipal wastes depending upon the sources. Waste Processing/treatment is chief stage of solid waste management. Several traditional and modern methods are adopted for the treatment depending upon the type of wastes collected. Composting is one of the traditional and common methods of processing of Municipal solid wastes in most of the countries. It is the process of converting the active organic portion in the solid waste into a stabilized product, which can be used as a nutrient source for plant growth and/or conditioner to improve soil physical character (Wong et al.2005). But the stability of final product is more important since chemically unstable compost is phytotoxic due to the production of ammonia, ethylene oxide and organic acids (Khan et al. 2008). Compost products contain a considerable variety of macro and micronutrients. Although often seen as a good source of nitrogen, phosphorous and potassium, compost also contains micronutrients essential for plant growth. Since compost contains relatively stable sources of organic matter, these nutrients are supplied through slow-release form. On a pound-by-pound basis, large quantities of nutrients are not typically found in compost in comparison to most commercial fertilizers. However, compost is usually applied at much faster rates; therefore, it can have a significant cumulative effect on nutrients availability. The addition of compost can affect both fertilizer and pH adjustment (lime/sulfur addition). Compost not only provides some nutrition, but often makes current fertilizer programs more effective. Hence the fate of essential nutrient metals and the heavy metals during composting is very important in order to characterize the quality of compost produced. Deficiency of nutrients like nitrogen, potassium, phosphorous or presence of excess toxic metals will affect the quality of the compost. Plants require seventeen essential elements for growth. If any one of these elements is missing, a plant will not grow, even if all the other elements are present in their required concentrations. The elements which plants require in relatively large amounts are called macronutrients. The primary macronutrients are nitrogen, phosphorus, and potassium. Nitrogen promotes are green leaves and foliage growth. Phosphorous stimulates healthy root growth and helps in the formation of flowers, seeds, and fruits. Potassium is required for proper development of flowers and fruit, ensuring good size, colour and quantity. The secondary macronutrients are calcium, magnesium, and sulfur. Calcium is essential part of cell wall structure, must be present for formation of new cells. Magnesium is essential for photosynthesis. Sulfur is responsible for characteristic odours of plants such as garlic and onion (Master Gardener Association of San Diego Country). Essential elements which plants need in relatively small amounts are called micronutrients, like Boron, Chlorine, Copper, Iron, Manganese, Molybdenum, Zinc, and Nickel. Composting organic matter such as grass clippings and tree leaves is an excellent way to provide micronutrients. Bernai et al (1998) were studied maturity and stability parameters of composts prepared with a wide range of organic wastes and concluded that, these parameters for mature compost depended on the wastes from which the composts were made. Number of studies also has been done for the organic matter evalution during the different types of composting processes, through the chemical and spectroscopic methods, i.e., Castaldi et al (2005), Farid Barjea et al (2012), Huang et al, etc. Elango et al (2009) states that the final compost material from the thermophilic reactor provides good humus to build up soil characteristics and some basic plant nutrients. Kanmani et al (2013) investigated the heavy metal contamination in soil due to leachate migration from an open dumping site. Study area: The study area chosen is Mysore city which is one of the major cities in Karnataka State (India). Mysore is spread over an area of about 128 sq. km with the growing population (0.76 millions in 2001 to 0.92 millions in 2011) at faster rate due to influx of many industrial activities. The city is situated in south of Karnataka state at a latitude of 12° 18´ N and longitude 76° 39´ E. The average annual rainfall of the district is 782 mm. The temperature ranges from 11°C to 38°C.The town is having a municipal area of 128.42 sq. km with 1, 74,956 households. In Mysore City, for all the 65 wards, door to door garbage collection is in progress. 60 wards are handled by outsourced labors and 5 wards are by federation of Mysore City wards parliament. 72 autotippers, 396 pushcarts are deployed for the waste collection. Chicken and mutton market wastes are collected separately. In addition to this, 168 numbers of 2 compartments auto tippers are procured under JnNURM to collect the dry and wet wastes from households. |
II. MATERIALS AND METHODS |
| Composting of municipal solid wastes: One of the main solid waste treatment plants for the wastes collected from the Mysore city is located at Vidyaranyapuram. The plant at Vidyaranyapuram is processing 200 tonnes of solid wastes per day. The bio-degradable wastes are collected daily, while the dry wastes like plastics are picked up once in a week. It includes wastes from households, shops and establishments, schools, temples, hotels, choultries, market, vegetable shops & meat shops, hospitals & clinics, industries and street wastes. It contains huge quantities of degradable and non-degradable waste materials. These wastes are stored in different piles and then treated with water and Bio-inoculum in order to maintain the moisture content and to enhance the degradation process. Turnover of piles has been carried out with the help of machines for the supply of oxygen. When the oxygen supply is too low, there will be anaerobic degradation resulting with unpleasant odours due to emission of volatile compounds like carbon sulphide, hydrogen sulphide, etc. After the degradation process, non-degradable substances like polyethene bags, glass materials, clothes etc., are separated mechanically. The degraded organic fraction of the solid wastes is called as compost and the final product was dried and powdered. After checking quality of the final compost, it applied to the agricultural lands as a fertilizer. Present study was carried out during pre-monsoon (Feb-May), monsoon (June-Sept) and post-monsoon (Oct-Jan) seasons. |
| Physico-chemical characteristics & heavy metals: The quality of the compost will be characterized by characteristics like temperature, pH, electrical conductivity, alkalinity, etc., along with elemental analysis. Samples were collected at 10 days intervals, randomly in 5 different places of the pile, at 1ft depth. Collected samples were mixed together and dried by exposing to sunlight followed by hot air oven at 105º C. Dried samples were crushed well and sieved through 0.2 mm mesh. The powdered samples were then used for further physico-chemical and spectral characterization Temperature (°C) was noted during the collection of compost samples at each 10 days interval. Moisture content (%) was estimated by gravimetric method. pH and Electric conductivity were measured for the 1:5 compost:water suspensions. Organic carbon (%) and Organic matter (%) were estimated using Walkey and Black method. Water holding capacity (%) was measured using filtration method. Phosphorus (ppm) was determined through Vanadomolybdate method. Carbonates, bicarbonates and alkalinity (mg CaCO3/L) were estimated by titration of compost: water (1:5) extract with standard H2SO4 solution using Methyl orange and Phenolphthalein as indicators. Calcium and Magnesium were estimated by titration of compost: ammonium acetate (1:5) extract with standard EDTA solution using Eriochrome Black-T and Patton-Reeder’s indicators. Brucine method was used for the estimation of nitrate-nitrogen, sodium and potassium concentrations were determined using Flame photometer. Humification Index was estimated through UV-Visible spectrophotometry. For estimation of heavy metals like lead, manganese, zinc, copper and iron etc, triacid (nitric acid: sulphuric acid: perchloric acid in 9:2:1 ratio) extract of the compost sample were used for Atomic Absorption Spectrophotometry. |
III. RESULTS AND DISCUSSIONS |
| Physico-chemical characteristics: Physico-chemical characteristics and essential nutrients estimation for samples collected from different stages of composting during the pre-monsoon, monsoon and post-monsoon seasons are listed in Tables 1, 2 and 3 respectively. |
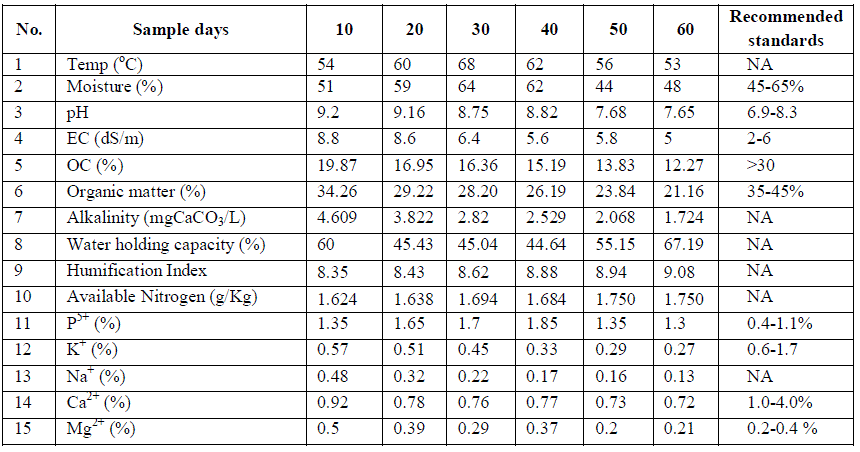 |
| The temperature of the composting pile was increased upto 68°C on 30th day of composting process in pre-monsoon season and it was 60°C and 66°C during monsoon and post-monsoon seasons respectively. The high temperature (50- 60°C) is important for distruction of weed seeds and for killing pathogens present in solid waste (Khan et al. 2008). The existence of microorganisms differs with the temperature of the composting pile i.e., at 13-21°c psychrophiles, at 21-32°c mesophiles and at 32-93°c thermophiles are responsible for the degradation process. Decrease in temperature at later stages of composting indicates the stablization of decomposed organic fraction into compost. After reaching certain temperature it remains constant even after additon of water, which indicates that compost had become stabilized. The pH of the compost was found to be basic in both pre-monsoon and post-monsoon seasons but acidic pH was observed in monsoon season. It is also observed that, pH was tending to neutral value towards the end of composting process indicating that the matured compost will have neutral pH and hence it will not affect the soil pH during its use. Electrical conductivity was higher for the samples collected during monsoon season, showing decreasing trend in all the three seasons which indicate that, the concentration of ions is reduced during the composting process. Percentage of organic carbon was due to liberation of carbon dioxide and methane as organic gases during the degradation of organic matter. Lowering of carbonates and bicarbonates and hence alkalinity is also observed during composting process. Water holding capacity was in the range of 40-60%, decreased trend at the beginning and then increased trend towards the later stages of composting process. |
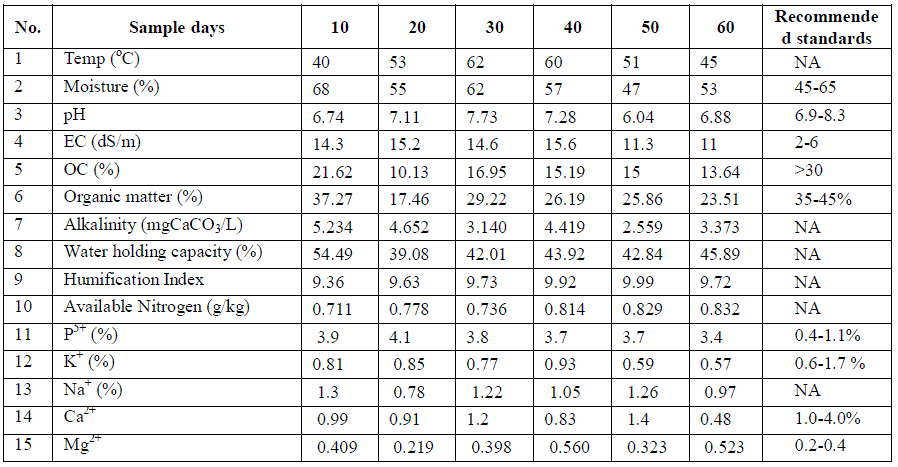 |
| TABLE 2 PHYSICO-CHEMICAL CHARACTERISTICS DURING DIFFERENT MATURITY STAGES OF COMPOSTING (MONSOON SEASON) |
| Essential nutrients: The primary macronutrients available are nitrogen, phosphorus; potassium and micro-nutrients like calcium, magnesium, sodium present in the composting samples at pre-monsoon, monsoon and post-monsoon seasons are also listed in Tables 1, 2 and 3 respectively. In macronutrients, available nitrogen was found in lesser concentrations (0.2-0.7g/kg) during post-monsoon season and it was higher during pre-monsoon season. Organic carbon concentration was high (10-21 %) during monsoon season. Hence, the carbon to nitrogen ratio was higher during the monsoon season. Percentages of organic carbon and available nitrogen decreased dramatically during the composting process. According to US Environmental Protection Agency, composting may result in i) CH4 emissions from anaerobic decomposition; (ii) long-term carbon storage in the form of undecomposed carbon compounds and (iii) nonbiogenic CO2 emissions from collection and transportation of the organic materials to the central composting site and from mechanical turning of the compost pile. Under anaerobic conditions there is large amount of ammonia that is produced from ammonification of organic compounds which cannot be oxidized by nitrification processes to form nitrates. Therefore, to conserve nitrogen in composts, it is crucial to maintain aerobic conditions. Apart from ammonia, nitrogen in compost is also lost as nitrogen gas (N2) and Nitrous oxides (NO and N2O). Percentage of phosphorus was higher (3.4-3.9%) during monsoon season and lesser during post-monsoon season (0.9- 1.05%). There was no significant change in the phosphorous concentration during composting but it helps to enhance the transformation of organic phosphorus into mineral forms and also to keep the magnitude of fixation of released phosphorus into insoluble inorganic forms at low concentration, thus increasing the availability of phosphorus in these compounds (Chattopadhyay et al. 1988). The sodium and potassium concentrations were fluctuating for the samples analyzed for composting process, whereas the concentrations on 60th day was lower than that of 10th day. This is due to run-off and leaching of the metal ions with the water content present in the solid waste during the process. |
| TABLE 3 PHYSICO-CHEMICAL CHARACTERISTICS DURING DIFFERENT MATURITY STAGES OF COMPOSTING (POST-MONSOON SEASON) |
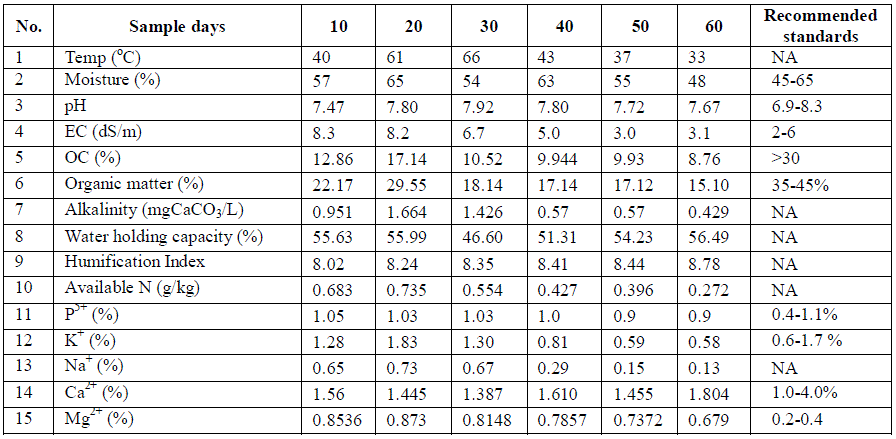 |
| On 60th day of composting process, calcium and magnesium concentrations were higher during post monsoon season (1.804 & 0.679 %) but calcium concentration was lower (0.48 %) during monsoon season, whereas magnesium was lower (0.21 %) during pre-monsoon season. Calcium is one of the most important nutrients required by the organisms and can also aid in maintaining the structure of plant cells and soil condition. Magnesium has an important role in photosynthesis, because, it forms the central atom of chlorophyll. From the results, it is observed that percentage of calcium and magnesium concentrations were below the permissible limits of Ohai EPA standards. |
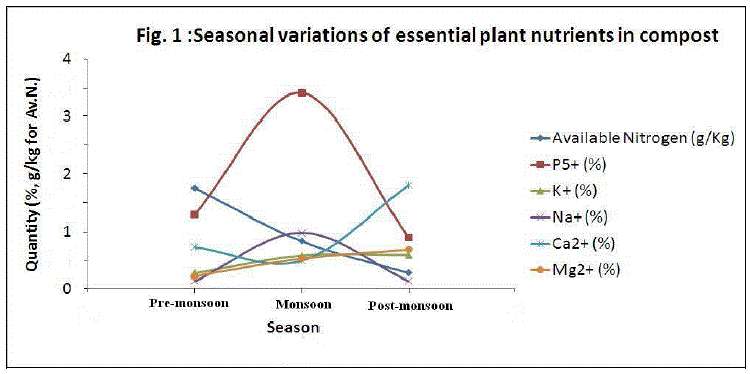 |
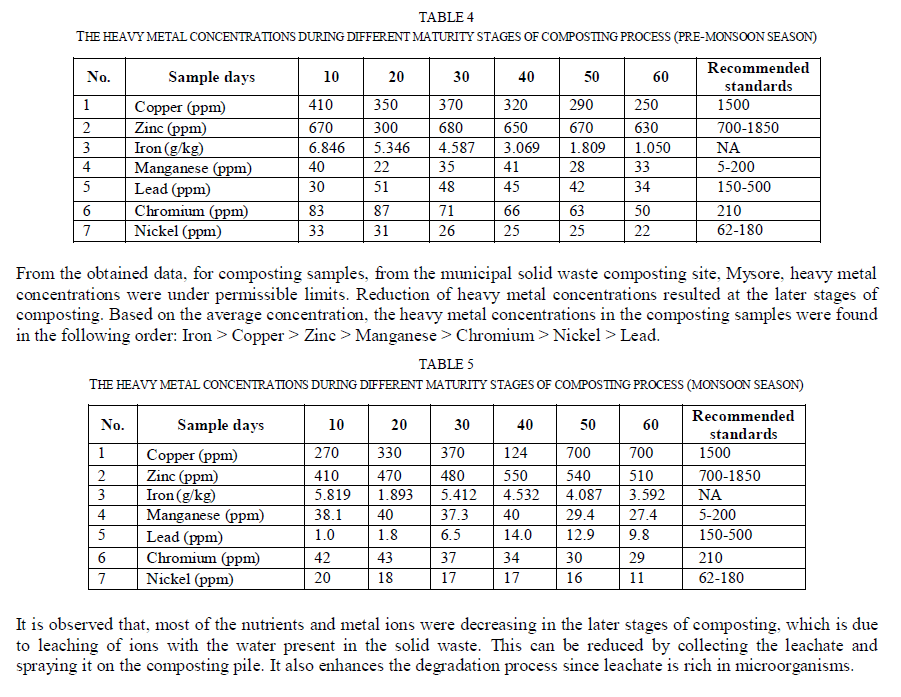
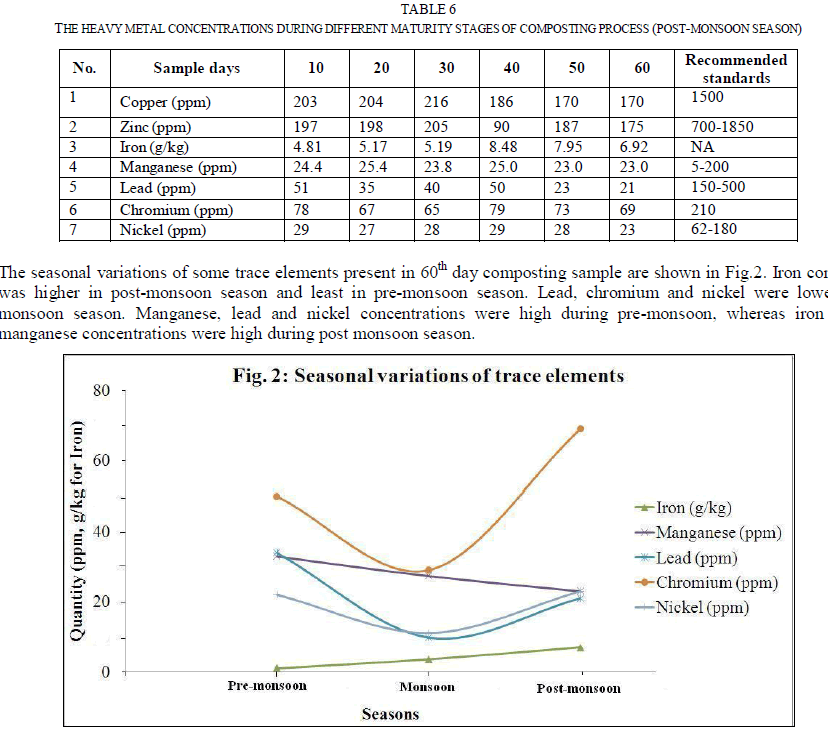
| Fig. 1 represents the seasonal variations in essential plant nutrients during 60 days old composting samples. The seasonal variation pattern for different plant nutrients was different in composting samples. Except calcium and available nitrogen, all other nutrient concentrations were found to be maximum during monsoon season. This may be due to presence of higher amounts of organic wastes and faster decomposition of organic matter. Since the moisture content was higher during the monsoon season, the decomposition of organic matter by microbial activity got increased. Heavy metals analysis: Heavy metals uptake by plants from the soil and successive accumulation in human tissues and biomagnifications through the food chain causes both human health and environment concerns. The heavy metal concentrations at different maturity stages of composting process during pre-monsoon, monsoon and post-monsoon seasons are listed in Table 4, 5 and 6 respectively. |
 |
 |
IV. CONCLUSION |
| Compost quality is difficult to assess from a single character, thus more characteristics should be taken into account for the said purpose. The compost prepared from the municipal solid wastes is a good source of nutrients for plants. From the present study, it is observed that, essential plant nutrients and heavy metal concentrations were found under the permissible limits of Ohai EPA standards and Canadian Council of Ministers of the Environment (CCME) standards. The total concentrations of trace elements like chromium, zinc, copper, lead, manganese and nickel were very low rendering final compost acceptable for agricultural use. The composting samples of monsoon season were rich in essential plant nutrients, contain lesser amount of heavy metals and hence it can be considered as good fertilizer. Excess of heavy metals in compost enhances the toxicity of soil. Since, nutrient and heavy metal concentrations in Municipal solid wastes were below the permissible limits, it is advisable to limit severe loss of nutrients during processing of municipal solid waste into compost. |
ACKNOWLEDGMENT |
| One of the authors, Mr. Manohara B. is grateful to University Grants Commission, New Delhi, for providing the fellowship in the form of JRF. |
References |
|