ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Neha Samber 1, Ajit Varma 2, Nikhat Manzoor3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The composition and antifungal activity of the Mentha piperita essential oil was studied after GC-MS analysis. Mint EO showed a high content of Menthol, Carvone and Menthone, the antifungal activity of which was determined against C. albicans, C. tropicalis and C. glabrata. The average MIC of Mint EO, Carvone and Menthol was 225μg/ml, 248μg/ml and 500μg/ml, respectively. The MFC values of the oil components were twice that of their MICs except the oil. At sub MIC values, Cells showed delayed and suppressed exponential phase of growth. At MIC, cell growth was completely terminated and clear zones of inhibition were observed on agar plates in all cases. Secretion of hydrolytic enzymes and yeast to hyphal transition was studied as virulence factors. These compounds showed very low hemolysis in comparison to fluconazole. The present study indicates that Mint EO and constituents significantly inhibit the virulence of Candida in susceptible and resistant strains.
Keywords |
| Candida, Mentha piperita, Carvone, Menthol, Menthone, Virulence factors |
INTRODUCTION |
| Since the last three decades, fungal infections specifically Candida species have emerged as a major cause of disease mainly in immunocompromised patients and have contributed significantly to microbe related morbidity and mortality [1]. The increase in antimicrobial drug resistance due to overuse of antibiotics along with toxicity and unfavourable side effects of already available drugs has caused serious problems [2], [3]. Polyenes cause serious host toxicity [4] whereas azoles are fungistatic and their prolonged use contributes to the development of drug resistance in C. albicans and other Candida species [5], [6]. This has encouraged search for new and less toxic molecules that can be developed as antifungals. The pathogenicity of Candida species is ascribed to certain virulence factors, such as the ability to evade host defenses, adherence, hyphal formation and the secretion of tissue-damaging hydrolytic enzymes such as proteases and phospholipases [1].The antifungal effect of essential oils of many aromatic plants has been described in several studies [7], [8]. Specific anti-candidal activity for several of these oils and their component molecules is also well established [9], [10]. Mentha piperita L., the peppermint plant is an aromatic perennial herb cultivated in most part of the world, have traditionally been used in folk medicine. Leaves of mint plant are frequently used in herbal tea and for culinary purpose to add flavour and aroma. The distinctive smell and flavour, a characteristic feature of Mentha spp. is due to the naturally occurring cyclic terpene alcohol called menthol. The essential oil of this herb is rich in menthone (14 - 32%) and menthol (30 - 50%). A large volume of literature is available on the medicinal properties of essential oils present in Mentha spp. A recent study was conducted to assess the phytochemical contents, antioxidant and antimicrobial properties of the locally grown mint plant leaves. However, to our knowledge no study has been done to explore the exact mode of action of this medicinally important herb. Previously we had reported the antifungal effects of Tulsi (Ocimum sanctum) essential oil [11]. In the present study we have explored the effect of Peppermint and its three lead components (carvone, menthol, menthone) against 5 standard Candida laboratory strains (C. albicans ATCC 10261, C. albicans ATCC 44829, C. albicans ATCC 90028, C. tropicalis ATCC 750 and C. glabrata ATCC 90030) on various virulence factors. |
II. MATERIALS AND METHOD |
| A. Strains, media and growth conditions Extra pure grade of Mint essential oil (Mint EO, ISO 9001:2008) was purchased from Mohan Perfumery Co., Tilak Bazar, Delhi, India and stored at 4ºC. Carvone, Menthol and Menthone were purchased from SAFC (USA), Aldrich (Germany) and MP Biomedicals (USA), respectively. All media, media components and fluconazole (FLC) were purchased from Hi Media (India) whereas all inorganic chemicals were of analytical grade and procured from E. Merck (India). Stock cultures of C. albicans (ATCC 10261, ATCC 44829, and ATCC 90028), C. tropicalis (ATCC 750) and C. glabrata (ATCC 90030) were maintained on slants of nutrient agar (1% yeast extract, 1% peptone and 2% dextrose and 2.5% agar) at 4ºC. B. Growth Studies (Turbidometric measurement) Candida strains were grown at 37oC in YEPD liquid media for 24 hrs and 1x106cells (A595 = 0.1) were inoculated into fresh media. 100 μl of test molecules containing required concentration was added to the culture. Growth was recorded turbidometrically at 595nm using Labomed Inc. Spectrophotometer (USA). The experiment was performed in triplicate for each concentration and average of each was taken into consideration. C. Disc Diffusion Assay Antifungal activity of Mint EO and its lead molecules in solid media was determined by the Disc Diffusion Method [12]. Candida cells (105cells/ ml) were inoculated in molten YEPD agar (~ 40ºC) and poured into a petri plates. Filter discs were placed on solid agar and different concentrations of test compounds (dissolved in 10% DMSO) were applied on the disc in 10μl volume. The average diameter of the zone of inhibition was recorded in millimeters after 3 days. The experiment was performed in triplicate. D. Proteinase Assay Fungal strains were transferred to flasks containing 5 ml YEPD and incubated at 37°C for 18 hrs. Following incubation, cells were centrifuged at 3000 rpm for 5 min. The pellets obtained were washed twice by resuspension in saline and centrifuged under the same conditions to remove the residual culture medium. After standardizing the scale of cell suspensions to a MacFarland 5 index (in saline), cell suspensions were added to desired concentrations of test compounds [13]. These suspensions (1 μl) were overlaid at equidistant points on proteinase agar plates (agar 2%; BSA 0.2 g; yeast nitrogen base w/o amino acids; ammonium sulfate 1.45 g; glucose 20 g; distilled water to 1000 ml). The plates were incubated at 37°C for 3-4 days [14]. Degradation of BSA was measured by dividing the diameter of the colony by the diameter of the colony plus zone of clearance (Pz) [15]. E. Phospholipase assay The phospholipase assay was performed according to the method described by [15]. After standardizing the scale of cell suspensions to MacFarland 5 index (in saline), cell suspensions were added to the desired concentrations of test compounds. These suspensions (2 μl) were overlaid at equidistant points on agar peptone media (agar 2%, peptone 10 g, glucose 30 g, NaCl 57.3 g, CaCl2 0.55 g, distilled water added to 900 ml) enriched with 10% egg yolk emulsion (HiMedia). The plates were incubated at 37°C for 2-4 days. Pz value was measured by dividing the diameter of the colony by the diameter of the colony plus zone of clearance [15]. F. Yeast to Hyphal Transition To initiate growth, Candida cells from agar slants were grown in Lee’s simplified medium [16] in Erlenmeyer flasks at 25 ºC up to the late log phase of growth (5×108 cells /ml). Cells were then inoculated into a fresh flask, grown up to stationary phase at 25ºC and maintained for 48 hr, resulting in a synchronous population at a final density of 2.5×108 cells/ml. For yeast to hyphal transformation, stationary phase G1 singlets (1.5×108 cells), maintained as described above, were transferred to 300 ml of fresh Lee’s simplified medium in Erlenmeyer flasks at 37ºC and pH adjusted to 6.5 to induce hyphae. The desired concentrations of test compound were also added to the Lee’s simplified medium. External pH was adjusted after every hour to the original setting (pH 6.5). Cell divergence was observed microscopically using Motic AE31 (Germany) by taking aliquots at different time intervals. At least 60 cells were counted on each cover slip, and the percentage of hyphae was obtained by taking the ratio of the number of hyphae to total number of cells. The maximum hyphal length observed was arbitrarily taken as 10 units, with the rest of the lengths graded accordingly on a relative basis. |
III. RESULTS AND DISCUSSION |
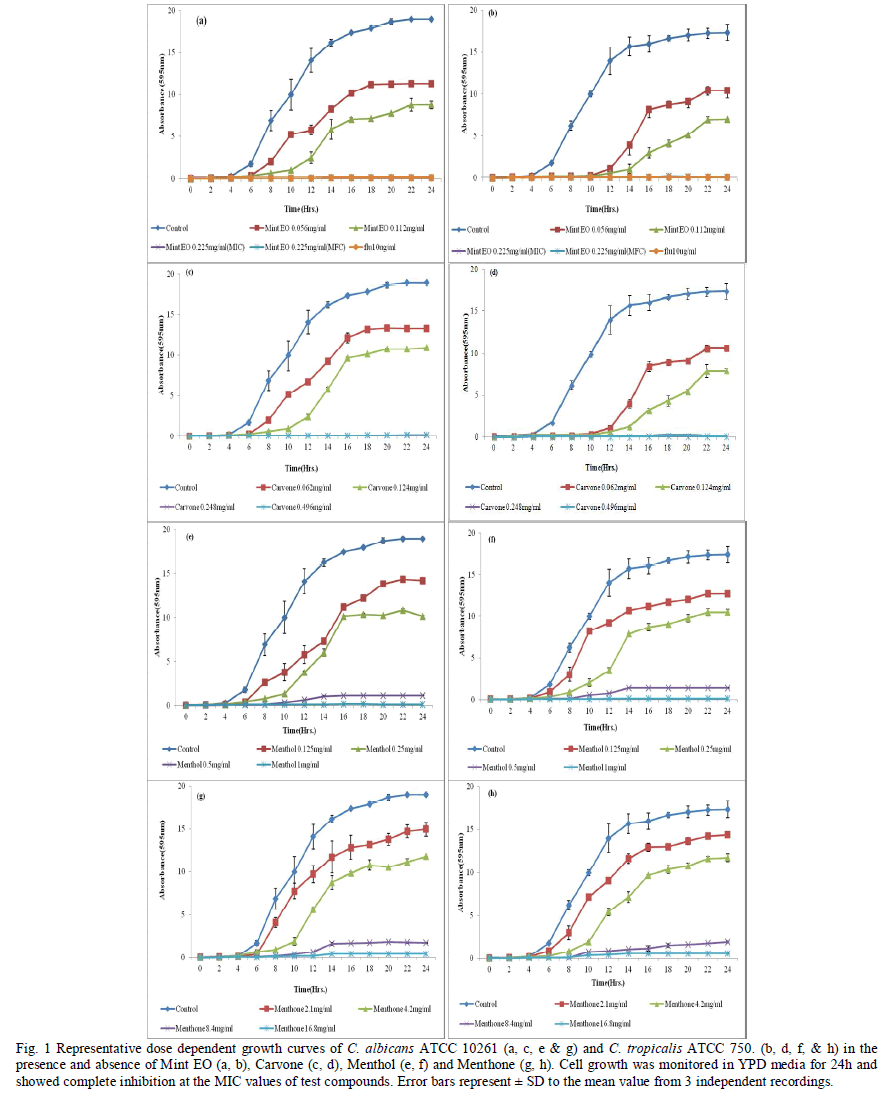
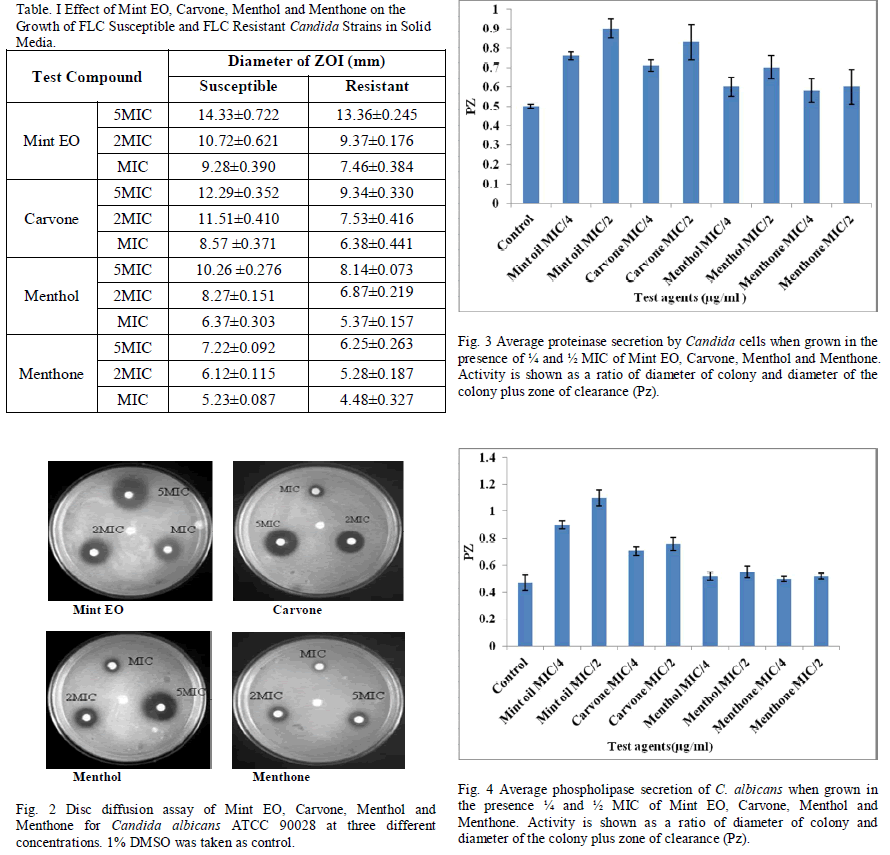
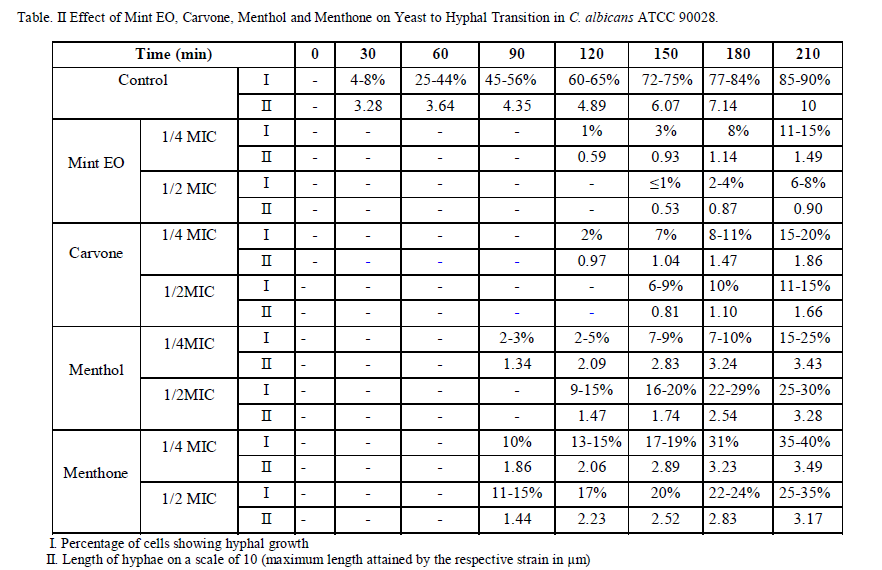
| The control cells in all cases showed a normal pattern of growth, with a lag phase of 4 hours and an active exponential phase of 8-10 hours before attaining the stationary phase. (Fig. 1a-b) shows a decrease in growth in the presence of increasing concentrations of Mint EO in both Candida species. Mint EO completely terminated the growth of fungal cells at its MIC value. The effect was almost equally as pronounced as with the negative control FLC (10μg/ml). Candida cells treated with sub-MIC values showed a depressed growth curve, with clearly differentiated phases. The higher concentrations (MIC & 2xMIC) clearly suppressed the exponential phase. The sub-MIC (MIC/2, MIC/4) treated C. tropicalis showed a delayed exponential phase, while the higher concentrations showed a suppressed growth curve with undifferentiated phases. (Fig. 1c-d) shows inhibition of growth by Carvone in a concentration dependent manner. The cells treated with sub-MIC concentrations showed a delayed exponential phase. The MIC and MFC of Carvone showed complete inhibition of growth. After 24 hr incubation, the cell population (observed microscopically) was highly reduced in comparison with the other test cultures and the control. Menthol, at sub-MIC values showed delayed growth, while the MIC and MFC showed complete inhibition of growth for both the Candida species (Fig. 1e-f). Similarly sub-MIC values of Menthone (2.1 and 4.2 mg/ml) showed depressed growth curves with clear differentiated phases (Fig. 1g-h) while MIC and MFC values (8.4 and 16.8mg/ml) completely terminated the growth of Candida. The Mint EO and its three components showed clear zones of inhibition (ZOI) which increased with increasing concentrations. At MIC (0.225mg/ml), Mint EO showed a ZOI of 9mm diameter which increased to 14mm when the concentration was increased to 5xMIC against C. albicans ATCC 90028 (Fig. 2). Carvone (0.25mg/ml) produced a ZOI of 8mm, which increased to 12 mm when the concentration was increased to 1.25mg/ml. The ZOI increased from 6mm to 10mm on increasing the concentration from 0.5mg/ml to 2.5mg/ml in case of Menthol. Menthone embedded discs produced a ZOI of 5 mm at 4.2mg/ml which increased only to 7 mm at 21mg/ml. (Table I) gives the values for the diameter of ZOI for all the test compounds in FLC susceptible and resistant Candida strains. All compounds tested here inhibited the growth of Candida strains in solid media, although less actively in comparison to FLC (10μg/ml). No significant differences were observed in susceptible and resistant strains. The order of inhibitory activity of the compounds tested at their respective MIC values was Mint EO > Carvone > Menthol > Menthone. Candida strains were tested for proteinase activity in the presence and absence of sub-MIC concentrations (1/2 and 1/4MIC) of Mint EO, Carvone, Menthol and Menthone. (Fig. 3) summarizes the data for proteinases activity calculated for all the strains tested in the present study. It was observed that at low concentrations of 1/8MIC, Mint EO and its constituents did not have any significant inhibitory effect on proteinase secretion (data not shown) by the cells. At higher concentrations of ½ MIC and ¼ MIC, Mint EO inhibited 44% and 34% proteinase secretion, respectively. Carvone inhibited proteinase secretion upto 40% and 29% at ½ MIC and ¼ MIC values, respectively in C. albicans. Our results show that Mint EO & Carvone inhibit proteinase secretion maximally in all the strains tested which corroborates well with the other results. Menthol inhibited proteinase secretion by 28% at ½ MIC & 16% AT ¼ MIC while Menthone showed around 13-17% inhibition at sub-MIC values. Secretion of phospholipases is another important virulence factor in Candida. Hence Candida strains were also tested for phospholipase activity in the presence and absence of sub-inhibitory concentrations of the Mint EO, Carvone, Menthol and Menthone. (Fig. 4) summarises the data for all the strains tested in the present study. It was observed that ½ MIC of Mint EO and Carvone inhibited 57% and 38% phospholipase secretion, while the same concentration of Menthol & Menthone could inhibit only 14% & 9% of enzyme activity on solid media. At lower concentrations of ¼ MIC, Mint EO, Carvone, Menthol & Menthone inhibited phospholipase secretion activity by only 47%, 33%, 9% and 6%, respectively. At very low concentrations (below ¼ MIC), Mint EO and its constituent compounds had negligible effect on both proteinase and phospholipase activity (data not shown). Yeast to hyphal transition is strongly dependent on pH besides other factors. Synchronized stationary phase G1 singlets were grown in Lee’s simplified media at two different external pH values of 4.5 and 6.5 to differentiate growth as buds and hyphae, respectively. In control cells (untreated) at pH 6.5, the germ tubes were visible after 30 min only. More than 80% cells could be seen undergoing yeast to hyphal transition after 210 mins. At the ends of 210 minutes, Candida strains exhibited variable increase in hyphal lengths depending on the concentration and type of the test compounds. All the compounds tested were found to inhibit yeast to hyphae transition considerably. (Table II) shows the percentage of cells forming germ tubes and the length of hyphae on the μm scale when exposed to Mint EO, Carvone, Menthol and Menthone. Mint EO was the most effective followed by Carvone, Menthol & Menthone in reducing both germ tube induction and hyphal length. Mint EO contains bioactive compounds including Carvone & Menthol and hence its efficacy appears to originate from their relative composition. |
 |
 |
| Incidentally, the oil used in the present study has a very high amount of Menthol & Carvone. At ½ MIC values of Mint EO, germ tubes could be seen only after 150 min of incubation when less than 1% cells showed morphological transition and hyphal length of only 0.53 while it was 6.07 in untreated cells on a scale of 10. After an incubation period of 210 min, when around 90% control cells showed transition with full hyphal length, the cells incubated with ½ MIC of Mint EO showed only 6-8% transition with hyphal length of only 0.90 on a scale of 10. The process of morphological transition itself seems to be affected to a great extent by the exposure of cells to sub-MIC values of Mint EO. Only around 8% cells were seen forming germ tubes even after 210 min. Further, at even lower concentrations of ¼ MIC, only 11-15% cells, respectively, showed morphological transition. Mint EO inhibits yeast to hyphal transition in Candida cells at pH 6.5, and hence interferes at the very initial stages of infection. Hyphal length was even more profoundly affected. While the cells continued to grow with time in all the two sub-MIC values of Mint EO in comparison to control, their length at each point of time was significantly reduced. Exposure of Candida cells to Menthol & Menthone did not affect the morphological transition as strongly as Mint EO & Carvone did. At ½ MIC of Carvone, 11-15% cells showed germ tube induction after 210 mins incubation. ¼ MIC of Carvone inhibited transition in about 75-80% cells only. With increasing concentration of Carvone, a decrease in the length of hyphae was clearly visible. Cells treated with ¼ MIC and ½ MIC of Carvone showed 81-83% reduction in hyphal length. Carvone seems to be the most promising test entity interfering with morphogenesis in Candida while Menthol & Menthone were effective but to a lesser extent. |
 |
| Mentha piperita is a perennial flowering member of the mint family. The herb has medicinal properties and has been used since antiquity as a digestive aid, for management of gallbladder and respiratory diseases [17-20]. Extracts of peppermint are used in many cosmetic products and in over the counter medicines [21], [22]. The antifungal potential of plant essential oils and their components has been described in several studies [7], [8], [23], [24]. Ocimum sanctum and its lead molecules have been shown to be good antifungal agents having multiple target sites in Candida [11]. Although the inhibitory activity of Mint EO and its major constituents against various pathogenic yeasts has been demonstrated earlier [1], [25-28], their effect on certain crucial virulence factors has not been verified. In the present study, we have demonstrated the fungicidal activity of Mint EO and its three lead biomolecules and studied their effect on the virulence of C. albicans (ATCC 10261, ATCC 44829, and ATCC 90028), C. tropicalis (ATCC 750) and C. glabrata (ATCC 90030). Mint EO was used only after analysing its constituents by GC-MS. Since the oil was characterized by high amounts of Menthol, Carvone and Menthone, studies were also done with these natural compounds. Although the quantity of Menthol was higher in this particular chemotype of Mint EO, Carvone showed greater antifungal properties than Menthol and Menthone. Our results show that both sensitive and resistant Candida strains were affected by Mint EO and its constituents. MIC90 of Mint EO was 225μg/ml against all the Candida strains used in the present study. Comparative analysis between MIC and MFC showed that MFC of the compounds Carvone, Menthol and Menthone was twofold higher than MIC in all the Candida strains while in case of Mint EO, MFC was the same as MIC. MFC may be a better predictor of therapeutic response than MIC, especially in immunosuppressed patients [29]. Menthone was the weaker antifungal while Mint EO and Carvone proved to be the most effective with low MIC and MFC values. Candida isolates having an MIC≥ 64 mg/l for FLC were considered resistant. The zones of inhibition formed on solid agar media by exposing the Candida strains with MIC and sub-MIC values of Mint EO and its constituents further demonstrated the fungicidal activity of these compounds while FLC showed fungistatic activity. The zones produced around the discs in diffusion assay were completely clear, an indication of potential fungicidal activity, whereas in contrast FLC showed a turbid halo, an indication of its fungistatic nature. Virulence characteristics of Candida species include adherence to host tissues, morphological changes, and secretion of hydrolytic enzymes like phospholipases and proteinases [30]. The present work demonstrates that at sub inhibitory concentrations, Mint EO and its lead molecules, significantly inhibit cell transition from yeast to hyphae which is considered to be the more pathogenic form, thus preventing the initial stage of the infection process. Germ tube induction is essential for the invasion of host tissues in the Candida infection process and our results show efficacy of these natural oil components against the virulence attributes of Candida. In control cells, the germ-tube induction proceeds with the increase in incubation time period. C. albicans is capable of producing proteinases [31], [32] and phospholipases [33] that hydrolyse peptide bonds and phospholipids, respectively and cause tissue invasion and damage. Mint EO and its lead molecules have a significant inhibitory effect on proteinases and phospholipases which suggests that these natural compounds can prevent the hydrolysis of host tissue when it is colonized with C. albicans. These compounds not only prevent the transition of the yeast to a more pathogenic hyphal form but also check the invasion of fungal cells into the host tissues. As phospholipases and aspartyl proteinases of C. albicans are considered the important virulence factors [34], the lowered secretion of these enzymes may indicate the less virulent nature of Candida strains, in the presence of Mint EO and its lead molecules. In the absence of test compounds, secretion of hydrolytic enzymes was greater. Candida albicans and other Candida species cause both superficial and systemic infection. Mint EO and its lead molecules used in the present study have the ability to kill C. albicans at their respective MIC values. In addition the therapeutic agent should be insoluble or at least sparingly soluble in these secretions. All the four compounds used here are only moderately soluble in aqueous environment. Our results showed that all the test entities have the ability to reduce the transition of C. albicans from yeast to hyphal form and also decrease the secretion of proteinase and phospholipase at subinhibitory concentrations. Hence these essential oil components have the potential to act as topical agents or rinsing agents when applied to a susceptible infection site at high concentrations which are high and can be maintained. The toxicity of these essential oil components was studied by in vitro haemolytic assay. These compounds are significantly less toxic and showed less than 10% hemolysis while FLC at the same concentration showed 100% hemolysis. Human erythrocytes useful for studying toxicity of compounds, as they are readily available, their membrane properties are well characterized, and their lysis can be easily monitored by measuring the release of hemoglobin. Hence, the in vitro hemolytic assay is a feasible screening tool for investigating in vivo toxicity to host cells [35]. Mint EO and its lead molecules Carvone, Menthol and Menthone were studied for hemolytic activity and the results indicate that these test compounds were significantly less cytotoxic than the conventional antifungal drug FLC. |
IV. CONCLUSION |
| conclusion, Mint essential oil components, Carvone, Menthol and Menthone are structurally related fungicidal bioactive compounds having low MIC values and negligible cytotoxicity. Mint essential oil and its three lead compounds (Carvone, Menthol and Menthone) not only reduce the transition of C. albicans from yeast to the invasive and more pathogenic hyphal form at sub-inhibitory concentrations but has also a significant effect on the production of the hydrolytic enzymes secreted by the fungal cell during infection. These compounds have a great therapeutic potential and further studies will lead to the development of new antifungal drugs. |
References |
|