ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Shyamala Viswanathan1 and Thangaraju Nallamuthu2*
|
| Corresponding Author: Thangaraju Nallamuthu, |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Seaweeds are available in large quantities; they appear to be the most suitable raw material for commercial exploitation. The major aim of the present study was to examine the physiochemical and biochemical properties of sodium alginate in different brown seaweeds of Colpomenia sinuosa (Derbes & Solier, 1851), Lobophora variegata (Lamouroux)Womersley,1967), Chnoospora implexa (J. Agardh, 1848), Padina gymnospora(Kützing) Sonder, 1871), Sargassum tenerrium (J. Agardh, 1848)and Dictyota dichotoma. Sodium alginate was produced by the method described in Haug et al (1974) and other properties were studied following standard methods. The highest yield of sodium alginate and maximum water content was recorded in C. implexa respectively. The maximum ash content was recorded in P. gymnospora (23.01%). Gelling and melting temperature was noted as 42º C and 85 º C, 82 º C and 70 º C respectively. The maximum carbohydrate was recorded in C. implexa (93.21%), protein content in L. variegata (7.17%) and the lipid content in D. dichotoma (6.12%). The study of their physiochemical properties, together with their biochemical composition, reveals their suitability to be a good source for human consumption. Further studies are necessary for isolation and characterization of the active molecules, which can be used to treat various oxidative stressrelated diseases.
Keywords |
| Brown seaweeds, Sodium alginate, Biochemical analysis and physiochemical analysis |
INTRODUCTION |
| Seaweeds have been consumed in Asia since ancient times, but to a much lesser extent in the rest of the world. Edible seaweeds are a rich source of dietary fiber, minerals, and proteins. Cell walls from marine algae characteristically contain sulfated polysaccharides, which are not found in land plants and which may have specific functions in ionic regulation. Seaweed dietary fibers are particularly rich in the soluble fractions, which in red seaweeds are mostly composed of sulfated galactans, such as agar or carrageenans. In brown seaweeds, soluble dietary fiber polysaccharides are alginates, fucans, and laminarans; the insoluble fibers are essentially made of cellulose. |
| Nowadays, Marine plant resources are attracting more and more attention as a raw material for the production of phytochemicals such as Alginic acid, agar-agar, carrageenan, iodine and the like, which are widely used in several industries involved in the manufacture of certain food materials, fertilizers and pharmaceuticals. Brown algae are a premising object in the food industry and prophylactic and clinical medicine. The word alginate is a generic term, meaning the various derivatives of alginic acid that either occur naturally in certain brown seaweeds (alginophyter),are produced from the natural derivatives. Alginate is the major structural component of the brown algal cell and mainly consists of β-D mannuron acid and α-L glucuronic acid units. In a wide range of industrial application, alginates are essential component as thickening, gelling or stabilizing agents (18 & 21).and in some cases, immunostimulatory agents (13). The preparation of alginic acid prevents chemically induced cancerogenes’s and ulcerogenes’s. The wide range of pharmacological effect of alginic acid is evidence of the nanospecific mechanism of its influence. The alginophytes growing in temperate climate, within a temperature range of 20°C or below it, are having good yield and viscosity, but those in tropical climate, though yield well, bas low viscosity. The brown seaweeds such as Sargassum sps and Turbinaria have been exploited from Gulf of Mannar at a rate of 3.128t dry weight during 1988 for industrial purpose. Since 1966, Sargassum wightii has been commonly harvested for commercial use. Generally the material is preserved in liquid preservative like sulphur dioxide, which can eliminate the bacterial contamination [28]. Improvement of viscosity of sodium alginate by pre- treating the raw material in hydrochloric acid has successfully been attempted [10]. Pre-treatment of the material not only increases the quantity of the product but changes its physical property to a marked extent. |
| Alginates, extracted from brown seaweed with an acid and an alkali, are used in a wide range of applications, particularly in the food, industrial, and pharmaceutical fields because of their water holding and gel forming capacities and ability to form and stabilize emulsions [22]. Extracts of alginate play a key role in the food industry, food, textiles, health and cosmetics. In the food industry, alginates were used to stabilize mixtures dispersions and emulsions, which increase viscosity and forms gel, such as jam and jellies [29]. Alginate can be used in the manufacture of soft capsules and is consumed as a beverage for lowering blood sugar level. In the textile industry, alginate was used as an additive for textile dye [17]. |
| The British chemist Stanford first described alginate (the preparation of “Alginic acid” from brown algae) with a patent dated 12th January 1881[25]. After the patent was further discussed in papers from 1883 [26]. Stanford believed that alginic acid contained nitrogen and contributed much to the elucidation of its chemical structure. The different sp consumed has a great nutritional value as source of protein carbohydrate, minerals and vitamin. The seaweed has been utilized mainly as raw material for extraction of phycocolloids as alginate from brown seaweed agar and carrageen are from red macro algae. It is generally agreed that all sp of the Phaeophyceae contain alginates. In recent year several seaweed industries manufacturing agar and sodium alginate have come up in India [1]. In seaweeds polysaccharides are the primary components of both the cell walls and the intercellular matrix. The biological functions of alginates in the plants include prevention of desiccation, maintaining of integrity of cells, and providing of mechanical strength and flexibility of the algal tissue. Ion-exchange functions are also important [24]. In biotechnology alginate gel beads are used as a matrix to entrap molecules of biological significance. Alginates can be used in the manufacture of soft capsules and in consumed as a beverage for lowering blood sugar level. In the textile industry alginate was used as an additive for textile dye [17]. |
MATERIALS AND METHODS |
| Collection Seaweeds |
| The six different species of brown algae (Colpomenia sinuosa, Lobophora variegata, Chnoospora implexa, Padina gymnospora, Sargassum tenerrium and Dictyota dichotoma) were collected from Rameswaram coast, Gulf of Mannar. The collected seaweeds were washed, shade dried and grounded into fine powder and stored at 4ºC for further use. |
| Extraction of Sodium Alginate |
| The extraction of alginate that had been develop according to the method of [14] were first leaching with 2% CaCl2 for 1 hr, second leaching with 5% HCl for 30min at 30 to 40 ºC, and treated with 40% of formaldehyde for 2hr to cross link phenolic compounds , the samples were then washed with deionised water and extraction with 5% Na2Co3 for 48 hr. this was then filtered through muslin cloth and precipitated in ethanol as sodium salt, neutralized with 0.5M / L, NaOH and precipitated again until pH 2.5 with 0.5 mol/L HCl. The precipitate was washed with acetone and dried overnight a 60 ºC. The yield of alginate was extracted as percentage/ dry weight. |
| Yield |
| Extraction yield was expressed as weight percentage of alginate with weight of the seaweeds samples [2]. |
| Physiochemical properties of sodium alginate |
| Ash content |
| The ash content was obtained by gravimetric method according to [2] by burning the sample in a hot air oven at a temperature of 200 ºC till it become ash. |
| Water content |
| The water content was obtained by gravimetric method according to [2] by drying the sample at a temperature of 105ºC. |
| Gelling and Melting temperature |
| The gelling and melting temperature of alginate was found using thermometer according to [27]. |
| Biochemical analysis of sodium alginate |
| Estimation of protein |
| The protein present in the sodium alginate was estimated by Biurette method had been developed by [23]. Protein was calculated by using BSA as standard and expressed as mg/g protein. |
| Estimation of Carbohydrate |
| The carbohydrate content was estimated by phenol- sulphuric acid method by [8]. Carbohydrate content was calculated by referring to a standard D- Glucose and the results have been expressed as mg/g sugar. |
| Estimation of Lipid |
| The lipid was estimated by using Chloroform – Methanol according to [9]. |
RESULT |
| Yield of sodium alginate |
| The highest yield of sodium alginate was obtained from C. implexa (29.15%) and L. variegata (27.57%) compared to other four different seaweeds. |
| Physiochemical properties of sodium alginate |
| Water Content |
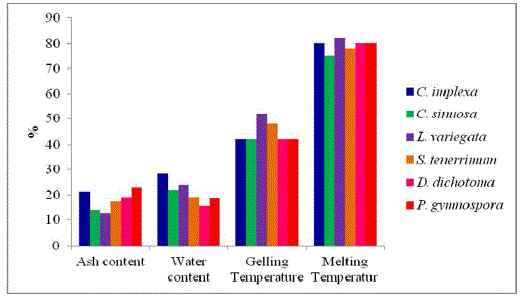
| The maximum water content (Fig. 1) was recorded in C. implexa (28.35%) and L. variegata (24.10%). The minimum water content was recorded in D. dichotoma (15.62%). |
 |
| Ash Content |
| Sodium alginate of P. gymnospora (23.01%) and C. implexa (21.53%) showed highest ash content (Fig. 1) whereas the lowest content was recorded in L. variegata (12.78%) when compared with other seaweeds respectively. |
| Gelling and Melting Temperature |
| The four species has a gelling point (Fig. 1) of 42ºC ± 2º C and L. variegata (52 ºC), and S. tenerrium (48ºC). The melting point was observed as 82, 78, 80, 70, 85 and 80 for C. sinuosa, L. variegata, C. implexa, P. gymnospora, S. tenerrium and Dictyota sp. |
| Biochemical analysis of sodium alginate |
| Protein |
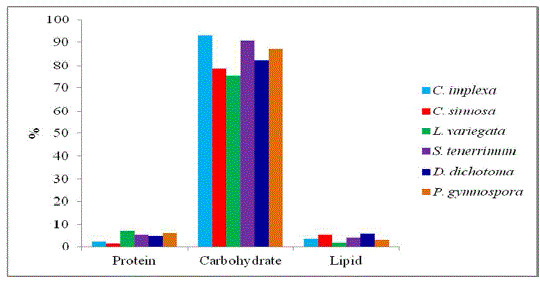
| The protein content ranged from 1.4% to 7.17% (Fig. 2). The maximum protein content was found in L. variegata (7.17%), followed by P. gymnospora (6.16%), whereas C. sinuosa (1.4%) showed minimum protein content respectively. |
 |
| Carbohydrate |
| Sodium alginate of carbohydrate content (Fig. 2) ranged from 82.42% and 93.21%. The maximum carbohydrate content was recorded in C. implexa (93.21%) and L. variegata (75.56%) recorded the minimum value, whereas C. sinuosa (78.54%), S. tenerrium (90.67%), D. dichotoma (82.42%) and P. gymnospora (87.21%) respectively. |
| Lipid |
| The maximum lipid content (Fig. 2) was recorded in D. dichotoma (6.12%) and the minimum lipid content was recorded in L. variegata (1.76%) compared to other seaweeds. |
DISCUSSION |
| Water content |
| The water content was influenced by the presence of ethanol used in the extraction process. Ethanol has ability to bind water from alginate solution, so it would reduce the water content. Parthiban et al., 2012 recorded maximum water content showed in brown seaweeds are Sargassum tenerrium, Padina boergesenii and Dictyota dichotoma 20.94%, 19.68% and 16.41% respectively. In [19] recorded the average water content in Sargassum and Padina as 12.7%. But the present study showed higher water in C. implexa (28.35%) and L. variegata (24.10%) than the earlier report. |
| Ash Content |
| The amount of mineral salt could differ in each alginofit. The presence of ash content showed that there were mineral salt. Mineral salt could be found on surface and in thallus. Conditions of hydrology and hydrochemistry on the habitat also influence the ash content. In the present study, P. gymnospora (23.01%) has higher ash content than the other seaweeds. |
| Protein |
| The higher protein content was found in L. variegata and lower in C. sinuosa. Quantitative analysis ranged from 1.4% to 7.17%. Similarly Parthiban et al., (2012) reported more protein content in brown alga S. tenerrium from tuticorin coast. In [7] recorded highest protein content in brown alga from Gulf of Mannar region. Protein content varied among different genera and also in different species of the same genus [6]. This change may be of spatial or temporal in nature. However, it is largely attributed to the surrounding water quality [5]. |
| Carbohydrate |
| The carbohydrate content of brown seaweeds ranged from 75.56% to 93.21%. The maximum carbohydrate content was recorded in C. implexa and L. variegata found the minimum content. Similarly, parthiban et al., (2012) recorded higher carbohydrate in S. tenerrium as 90.67% respectively. In [7], reported higher carbohydrate in the Saccharina longicruris, Ascophyllum nodosum and fucus vesiculosus as 99.1%, 89.6% and 84.1% respectively. In [13] also reported similar kind of results that the green seaweed is having high carbohydrate than the red and brown alga. In [3] recorded higher carbohydrate in S. tenerrium (35.27%). |
| Lipid |
| The lipid content of seaweeds ranged from 1.76% to 6.12%. The maximum lipid content was recorded in D. dichotoma and L. variegata recorded the minimum content. Parthiban et al (2012) reported maximum lipid content in D. dichotoma. Similarly, maximum lipid content in the green seaweed Enteromorpha intestinalis (7.13%) was recorded. In [20] also recorded the highest lipid content in green alga Ulva fasciata. Mostly brown seaweeds have low lipid content than green seaweeds. |
CONCLUSION |
| The search for new metabolites from marine organisms has resulted in the isolation of more or less 10,000 metabolites [11] many of which are endowed with pharmadynamic properties. According to [15], only 10% of over 25,000 plants have been investigated for biological activity. In addition, there are number of seaweeds with economic potential [4]. Our study indicates that all the six different seaweeds would be good source of raw materials. Hence, the biochemical properties are sodium alginate; Carbohydrate, Protein, Lipids and Ash content were influenced by environmental parameters. The study of their physiochemical properties together with their biochemical composition reveals their suitability to be a good source of food for human consumption. This was confirmed by other reports, indicating that environmental conditions at on biosynthetic pathways. It can be concluded that seaweeds can be utilized as a source of natural biochemical compounds like sodium alginate. Bioactive compounds found in seaweeds await a major breakthrough for a variety of food and medical applications. Further research effort should be targeted at the isolation of new bioactive natural compounds useful as natural antioxidant, pharmaceutical agents or dietary supplements. |
ACKNOWLEDGEMENT |
| Authors are grateful to Prof. R. Rengasamy, Director, Centre for Advanced Studies in Botany, University of Madras, Maraimalai Campus, Guindy, Chennai- 600 025, Tamil Nadu, India for providing laboratory facilities and for his constant support and encouragement during the course of study period |
References |
|