ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Channaoui sanaâ1, Blaghen Mohamed1, Mestour ilham1 and Errachidi Faouzi2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The study of the Blue Cibacron (BC) treatment process through an experiment factorial design allows us to determine a function ƒ such that (Y= ƒ(X1, X2, …, Xn)) existing between magnitude Y which is the decrease of coloration (called response), and variables Xi , which are biomass (m), pH and Temperature (called factors). A complete factorial design (23) was made in order to determine the factors and their interactions which have a statistically significant influence on the studied response. Temperature has a significant effect on the reduction of coloration; by contrast, pH and biomass (m) does not have a significant effect. On the other hand, the interactions between the three factors have significant effects on the decrease of coloration.
Keywords |
| Blue Cibacron, Coprinus cinereus, temperature, pH, biomass, EFD and optimization. |
INTRODUCTION |
| Synthetic organic dyes are extensively used in textile industry. During manufacturing, an estimated 10-15% of these dyes are lost in the effluents, rendering them highly colored and polluted [1]. Release of these dyes into the environment has become a major concern in wastewater treatment since some of them or their metabolites may be mutagens or carcinogens [2, 3]. |
| Cibacron Blue, also called Procion Blue HB or Reactive Blue 2, is one of the most reactive synthetic dyes used in the textile industry [4]. In addition to the triazine group acting as reactive group, the Cibacron Blue (CB) presents an anthraquinone group as chromophore (Fig.1) dyeing natural fibers, such as cellulose, through covalent bonds [5]. Moreover, this polynuclear monochlorotriazine dye was shown to bind most types of proteins [4, 7]. |
| The major environmental problem associated with the use of this type of reactive dyes is due to their inefficient fixation to the fibers. Therefore, significant losses occur during the manufacture and processing with dyes being discharged as effluents into publicly owned water treatment plants. |
 |
| Considerable research efforts have been devoted to optimize color removal from effluents of textile industry. Most of the chemical and physical methods proposed for treating dye wastewater have been not widely applied because of the high cost and the secondary pollution that can be generated by the excessive use of chemicals [7-8]. This has led many workers to search for the use of microorganisms, such as bacteria or fungi, for the degradation of these compounds in wastewater treatment systems. Anaerobic bacteria [9-10] carry out most biological degradations of textile dyes. Generally, textile dyes are resistant to bacterial attack under aerobic conditions [11-12]. Furthermore, bacterial enzymes involved in dye degradation have been shown to be very specific [13]. |
| White rot fungi are however efficient in biodegradation of recalcitrant compounds like xenobiotics, lignin and dyestuffs by their extracellular ligninolytic enzyme system [14-15], so they offer significant advantages over than bacteria. Their extracellular enzyme systems including Mn dependent peroxidase (MnP), Mn independent peroxidase (P), lignin peroxidase (LiP) and laccase (L) being non specific can attack a wide variety of complex aromatic dyestuffs [16, 17]. Since the enzymes are extracellular, the substrate diffusion limitation into the cell, generally observed in bacteria, is not encountered. White rot fungi do not require preconditioning to particular pollutants, because enzyme secretion depends on nutrient limitation, nitrogen or carbon, rather than presence of pollutant. The extracellular enzyme system also enables white rot fungi to tolerate high concentrations of pollutants [18]. |
| Major improvements in the methods of bioremediation attributed generally to the development of efficient strains are also closely related to parameters such as cultural conditions environment in which the microorganisms are exposed and which affect the performance of treatment [19]. |
| This research has been designed to integrate the perspective of improvement of BC discoloration and it aims at determining the best values of factors and the best combinations between physicochemical factors (pH & T°C) susceptible to reach a decrease coloration in order to improve the efficacy of the process. In this context, the present work, initially, aims modeling and optimizing of physicochemical parameters Blue Cibacron discoloration by the strain Coprinus cinereus grown on minimal medium. |
MATERIALS AND METHODS |
1 Materials |
| The fungus Coprinus cinereus used in this study was isolated from effluents contaminated dyes of an industrial sector of the city of Casablanca (Morocco). It has been identified based on morphological and biochemical characterization of the isolates grown on nutrient agar PDA (potato dextrose agar)[20] and by using taxonomic keys described by[21]. |
| In this study two culture media were used : Potato Dextrose Agar (PDA), used in mycelium transfer and minimal medium (MM) containing [0.6 g l-1 K2HPO4, 0.1 g l-1 MgSO4, 0.6 g l-1 (NH4) 2SO4, 0.5 g l-1 NaCl, 20 mg l-1 CaCl2, 1.1 mg l-1 MnSO4, 0.2 mg l-1 ZnSO4, 0.2 mg l-1 CuSO4 and 0.14 mg l-1 FeSO4 (pH adjusted to 7 with 1M HCl)] used to assess BC discoloration. |
2. Evaluation of the discoloration. |
| The minimal medium used in each experiment is added, after sterilization for 20 minutes at 121 °C, by Cibacron blue so as to obtain a final concentration of 25mg/liter. Then, each flask was inoculated with 0,1 or 0,2 mg / ml of biomass (dry weight equivalent). These are incubated in an incubator at 30 or 37 °C according to the experimental design. Discoloration was evaluated by reading the absorbance at 612 nm against the template (untreated medium). |
3. Experimental Design |
| Many factors can significantly influence the discoloration of BC. The bibliographical data have advised us to study three factors: biomass concentration (m), pH, and temperature (T°C). This study was performed according to factorial experiment design where the calculation of the coefficients of the polynomial model has been accomplished through the method of “least squares” with the use of coded variables. In fact, the act of replacing the natural variables by coded variables allows for the same domain of variation for each factor (between –1 and +1) and hence being able to compare the effect of factors among themselves. The lowest level is coded –1 while the highest level is coded +1 (Table 1). |
 |
4. Statistical Analysis |
| The statistical calculations (calculation of coefficients, T-test, analysis of variance, curves were done using the JMP software. |
EXPERIMENTAL RESULTS |
| On the basis of table 3, the mathematical model is written as follows and has the form: |
 |
| We also noted that the interactions between Temperature and pH (T(°C)*pH), temperature and biomass (T(°C)*biomass), between pH and biomass (pH*biomass) and between Temperature and pH and Biomass (T(°C)*pH*Biomass) have significant effects on the discoloration. pH and temperature factors can not be ignored in the mathematical model because their interaction is high so the mathematical model is retained. |
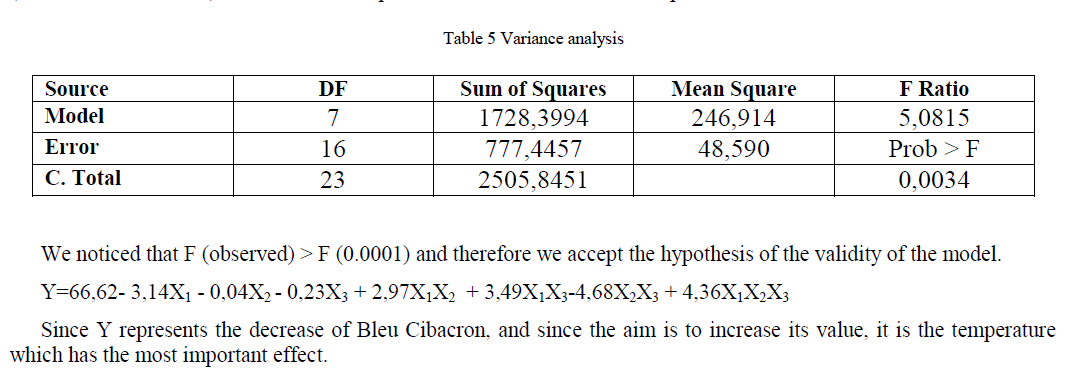
| Y=66,62- 3,14X1 - 0,04X2 - 0,23X3 + 2,97X1X2 + 3,49X1X3-4,68X2X3 + 4,36X1X2X3 |
| Variance analysis (Table 5) which aim is to compare the sum of differences squares due solely to regression (therefore to the model) with the sum of squares of the residues with the help of the F test. |
 |
| Limited to statistically significant words, we can adopt the mathematical model to find the relationships between the factors studied in pairs to draw the isoresponse (Contours plot) curves. This allows to decipher the interactions of the factors in pairs. These allowed us to draw isoresponse curves connecting temperature and pH, temperature and biomass and pH and biomass (Figures 2, 3 and 4). To trace the isoresponse curves, we used the JMP software that facilitates the task. |
• Study of the interaction of pH and temperature |
| To study the fading under the experimental conditions of pH and temperature, the factor X3, corresponding to the biomass (m) may take discrete values (-1, +1) corresponding respectively to the values of 0.1mg/ml and 0.2mg /ml concentration of biomass. Figure 2 shows the responses obtained in the experimental area bounded by the temperature (T) and Hydronium potential (pH). From this figure, we see that the increase of both factors simultaneously improve discoloration (blue curves) and decrease reduces the phenomenon of discoloration (yellow and red curves). |
| Both physicochemical parameters (pH & Temperature) explain in a clear theirs effects on enzyme activities involved in BC treatment process. The enzymatic equipment affected include Mn dependent peroxidase (MnP), Mn independent peroxidase (P), lignin peroxidase (LiP) and laccase (L) [16, 17]. The study of their interaction gives an idea about the ability to automate the process. |
 |
| The study of the interaction between biomass and temperature is the evaluation of the effect of the biomass, which plays the role of biocatalyst on the stability of endo-enzymes and transfer of exo-enzymes for degradation of BC [16, 17]. The results illustrated in Figure 3 show that the biomass may be stable source of the two types of enzymes which are positively correlated to biomass. |
• Study of the interaction between pH and biomass m |
| For the study of discoloration under the experimental conditions of pH and biomass, X1 factor corresponding to the temperature, can take discrete values (-1 and +1) which are respectively values are 30°C and 37°C. Figure 4 displays the isoresponses curves in the experimental zone bordered by the pH and the biomass (m). From this figure, we notice the existence of a symmetric reduction (red curves) on either side of the diagonal (-1,+1) (blue curves). These results show that the pH behavior differs from that of the temperature. Thus, the temperature factor could play an important role in controlling discoloration Cibacron Blue. |
 |
| Interaction study between biomass (m) and pH goal evaluating potential hydronium effect on peroxidases enzymes biosynthesis involved in BC bioremediation. The results shown in Figure 4 show that neutral pH (pH = 7) and low biomass (0.1mg/ml) are favorable for maximizing the discoloration of BC. Thus, it can be inferred that a limited fraction of peroxidase enzymes may be involved in this study. |
CONCLUSION |
| The overall results of this preliminary study, using the full factorial design shows that the optimal conditions for obtaining the best yields are fading as follows: |
| - A temperature of 37 ° C, |
| - A pH of 7, |
| - A biomass of 0,1mg/ml. |
| The temperature factor could play an important role in controlling discoloration Cibacron Blue. We intend in this research, further optimize the discoloration of Cibacron Blue studying nutritional factors, such as the nitrogen source and the source of phosphate. Subsequently we intend to combine the physical, chemical and nutritional factors in a single plane of experience to master discoloration Cibacron Blue. |
References |
|