ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Kiran M. Khandarkar*1, Mudrika Ahmed2, Jyotsna S. Meshram3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Zeolites have been used as an efficient and cost effective activating catalyst for the synthesis of furfural urea resin. The condensation of furfural with urea is conducted in aqueous and organic phase with natural as well as synthetic zeolite as catalyst. The catalysts are recycled and reused several times. This reaction is scalable to milligram scale and the methodology has resulted in an efficient synthesis with superior product. Herein benign, environ friendly, efficient and extremely fast procedure for the synthesis of untainted, superior furfural urea resin (FUR) have been demonstrated. The white FUR is characterized by 1H and 13C NMR, FTIR, X-ray diffraction and thermal analysis (DTA, TG, DSC).
Keywords |
| Natural Zeolites, stilbite, Furfural, Furfural-Urea resin, green polymerization. |
INTRODUCTION |
| Urea, a carbamide, is common organic chemical with versatile characteristics. It forms resins with aldehydes, most commonly formaldehyde, to produce urea-formaldehyde (UF) resins, which is used as a wood adhesive for many years, and is still the most widely used interior plywood and particleboard adhesive. But the urea treatment of wood ceased after a few years as the UF resins are susceptible to hydrolysis by water, and there is the potential of formaldehyde (toxic) release from the product. But, urea is an attractive chemical for wood improvement because of its non-toxicity, cheapness and ease of use. As a result, since the 1980s the inventors have been trying to find ways to overcome the disadvantages of UF treated wood. Furfural (C5H4O2) is aldehyde derived from plant material containing pentosans by acid hydrolysis. It is non toxic and used in agriculture both as a fungicide and a nematicide as it is nontoxic to either humans or the environment [1]. It would be very advantageous to have a furfural-urea resin (FUR) that is capable of being absorbed by wood in sufficient amounts to enhance the wood's mechanical properties and biodeterioration resistance. Furfural has lower vapor pressure than formaldehyde, so there are lower emissions. Furfural-urea resins are dark, crosslinked and waterproof. Adhesives and bonding are the largest markets for furfural resins [2]. Furan resins have found varieties of applications, such as bonding material for corrosion-resistant fiber and glass-reinforced plastic, which have excellent resistance to heat distortion and flame spread [3], membrane for water desalination [4], binder in core molding and friction materials [5], ion exchange [6], and antimicrobial resins [7]. FUR resins were also referred as wood stabilization compounds [8] and as a fertilizer having fungicidal assets [9]. There was work done on furfural-urea resins in the early 20th century [10, 11]. Urea condensed readily with furfural in acid catalyzed medium [12]. The acids used are mild in nature like acetic acid, formic acid, citric acid, lactic acid, maleic anhydride, phthallic anhydride and hydrochloric acid was also considered of concentration 0.036-0.007M [13]. Organic and mineral acids like proline [14], acetic acid [15], p-dodecylbenzenesulfonic acid [16] and some Lewis acids [17, 18], Bronsted acid catalysts [19] and Lewis base catalysts [20] have been reported to catalyze FUR synthesis. They often suffer from the drawbacks of long reaction times, toxicity, difficulty in product separation and harsh reaction conditions like large quantities of mineral or Lewis acids as activators on work-up hydrolyzed with generation of large quantities of corrosive and toxic waste by-products; the use of stoichiometric quantities of reagents that produce metal salts as waste with poor yields and/or production of mixtures of regioisomers with low selectivity, which limit its use in the synthesis of complex molecules. With the increase in temperature or concentration of acid catalyst the reaction time decreases. However, the stability of furfural is incredibly frail and acidic media or rise in temperature augments the instability [21], owing to which quality of product recedes producing dark colored FUR. |
| Considering the difficulty in acid catalyzing furfural-urea condensation and a growing demand for the development of environment friendly synthetic manipulations, in this paper we had given green catalysis approach by using natural zeolite. In a liquid phase reaction, presence of a solid acid catalyst, particularly a zeolite can enhance reaction rate, support reagents, entrain by-products and enhance product selectivity [22]. This work was dedicated to elucidate whether the furfural urea condensation reaction can effectively catalyzed with a green catalyst with improved rate & yield of reaction in both aqueous and heterogeneous medium. Our results shows that not only the yield of furfural urea condensation increased but quality of the product also enhanced by a green sustainable polymerization using zeolite, with complete characterization of FUR. |
RELATED WORK |
| Furfural and furfuryl alcohol are incorporated in urea-formaldehyde (UF) resins for its modification as formaldehyde gets emitted from the wood products bonded with urea-formaldehyde (UF) resin. Beside the greater utilization of UF resin, formaldehyde emission resulting in potential health hazards and environment pollution [23], ebbed its demand in the market. These hazardous emissions of formaldehyde can impeded or completely escape by modifying UF polymers with Furfural [24-27]. |
EXPERIMENTAL |
| Urea furfural condensation was more feasible in acid catalyzed medium. The aldehyde group of furfural undergoes condensation with two urea and link the two urea substrate with the release of a water molecule. The rate of the reaction increases in acidic condition but at the expense of quality of the product (FUR). Therefore in the present work we have developed a zeolite catalyzed approach having green skill, high reaction kinetics and great quality of product FUR, as illustrated in scheme 1. |
 |
| Reaction in aqueous phase: The synthesis was carried out in a flask applied with a magnetic stirrer at room temperature. Urea (1 mole) was dissolved in 10 ml distilled water and furfural (1 mole) was added. When furfural was partially dissolved, 0.5 g zeolite was incorporated into the solution. A white solid began to precipitate in the solution after stirring for 10–15 min, depending on the zeolite used, and some minutes later no liquid could be observed. An excess of water was added to remove un-reacted urea and isolate the product by vacuum filtration. The product was washed several times with water and the incorporated zeolites were removed and reused. The bright white solid product was vacuum dried with yield achieved between 60 and 80%. |
| Reaction in organic phase: |
| The reaction was conducted in polar organic solvent like ethanol at room temperature in presence of zeolite as a catalyst in a closed vessel. 1 mole urea and 1 mole furfural were mixed carefully in minimum quantity (5 ml) of ethanol to guarantee the maximum interaction between reagents. The reaction mixture was covered and stirred with 0.5 g zeolite in ethanol, a white solid starts appearing after 5-10 min. The product was thoroughly washed with ethanol and dried in a vacuum rotary evaporator. A bright white FUR was obtained with 70–87% yield in 60-70 min depending upon the catalyst used. |
| Furfural (Merck) was vacuum distilled just before being used for syntheses. Urea (Merck) and other reagents were used without further purification. The product’s characterization was assessed by X-ray diffraction XRD using a PANlytical’s Xpert-Pro MPD diffractometer and K-Alpha1 Cu radiation (1.5418 Å); Infrared spectra were recorded on Bruker Alfa-T with direct sampling for analysis; 1H and 13C NMR spectra were recorded on a Cryo-magnet Spectrometer 400 MHz (Bruker) instrument using tetramethylsilane (TMS) as an internal standard with DMSO as a solvent. Chemical shifts are given in parts per million (ïÃÂä scale) and the coupling constants are given in Hz; thermal analysis was done by Mettler Toledo 851e device with TGA and DSC in presence of nitrogen while TGA in air. Samples were inserted at a temperature of 30°C and heated with a rate of 10°C min-1 to a maximum temperature of 1000 °C. The stare software Mettler Stare Default DB V9.20 was used for evaluation of the detected thermograms. |
RESULTS AND DISCUSSION |
| Condensation of Furfural and Urea was carried in different phases, with and without variety of heterogeneous catalysts like natural zeolite viz. Indian natural zeolite, such as Scolecite (CaAl2Si3O10 .3H2O), Stilbite (NaCa4 (Si27Al9)O72 .28(H2O)), Fluorapophyllite (KCa4Si8O20(F,OH)·8(H2O)), Mesolite (Na2Ca2(Al2Si3O10)3 ·8H2O) and synthetic zeolite with general formula - M2/nO[(AI2O3).(SiO2)X].y H2O where, M represents alkali metals sodium, potassium and calcium. These aluminosilicates are 3A, 4A, 5A and 13X with pore size 3Å, 4Å, 5Å and 10Å respectively. The alkali metal in aluminosilicate 3A and 4A is potassium, 5A is calcium and 13A is sodium. The zeolites were used in macro granular form for ease in recollection. The catalyst can be reused several times, without appreciable loss of activity. The recovered catalyst was washed with ethyl acetate, then dried at 70°C prior to use for next run in model reaction and activated at 350°C after five consecutive runs. The chemical compositions of natural and synthetic zeolites in the weight percentage of the composition are described in Table 1. |
 |
| Synthesis: |
| The yield of urea furfural condensation product was higher with the use of catalyst or medium in an acidic range i.e. in pH 2 to 6. The reaction in the heterogeneous phase occurs from the surface toward the centre of the solid particles. As the reaction proceeds, the particles formed agglutinate and their external layer which is more exposed to air, turns harder and browner. Thus, the isolated product does not exhibit a homogeneous color; some parts are darker than others. There are two facts which should be considered in order to understand the change in color of the urea-furfural resin. The first one, which is common to both procedures, is the presence of small quantities of a dark product arising from the oxidation of furfural [28]. The second one, which is applied to the catalyzed the process, is the additional contribution of the colored species produced by the interaction of furfural with acid [29]. In fresh solution urea is acidic and becomes alkaline upon standing, also it interacts with hydrogen ion [30] and both pH and pK values of weak acids increases in its presence [31]. Thus the use of acid as a catalyst increases the yield of the product with a diminished quality due to oxidation of furfural. Many of the mild acid, acid analogs, Lewis acid and even HCl of concentration 0.036 to 0.007 mole/l used gave the same dark colored furfural urea resin. |
| The present work gives solution to the problem with reduced reaction time, superior white FUR without use of environmental hazardous catalyst. It consists of use of natural as well as synthetic zeolite as a catalyst in polymerization of urea with furfural. The facile zeolite catalyzed reaction conditions involve addition of urea and furfural in aqueous or organic phase at room temperature with stirring. The time required for urea furfural condensation in presence of range of natural and synthetic zeolite catalyst with their production yield is given in Table 2. |
 |
| All the FU-condensation products obtained via natural as well as synthetic zeolite catalyst were of high purity. Quality of FUR obtained from each zeolite was equal with high yield but crystallinity differs with the phase employed. Amongst the zeolite considered, stilbite gives higher yield in both the phases as compared to others and thus the FUR produced through it was considered for further examination. |
| IR spectroscopy: |
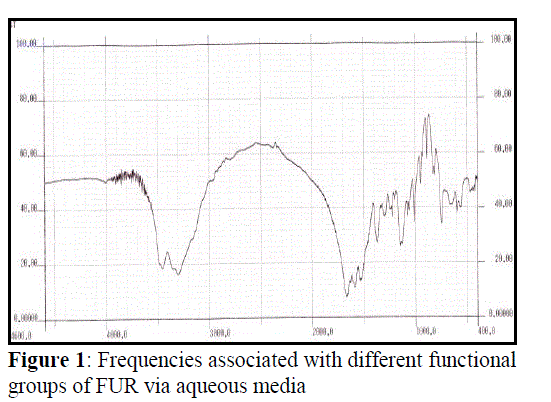
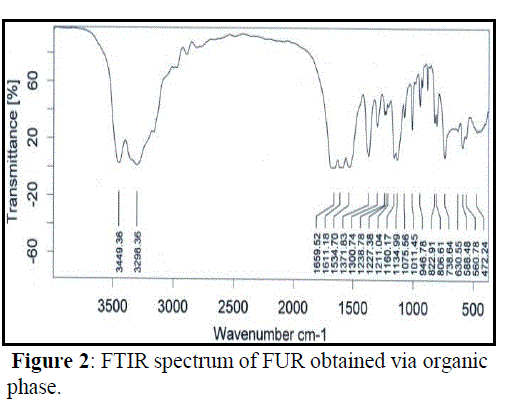
| IR spectra of FUR obtained by the two synthesis methods were recorded as shown in Figure 1 and Figure 2. Both the spectrum were found to be equal, deducing the frequency of the main absorption bands and therefore their IR spectrum is elucidated concurrently. The νas (N–H) and νs (N–H) appear at 3349.36 and 3298.36 cm−1 respectively. These two bands appear at 3500 and 3350 cm−1 in urea (NH2 stretching absorbance). The observed shift in these two bands in urea furfural resin compared to their positions in urea reveals the conversion of primary amine to the secondary, forming the HN-C urea furfural linkage with ν (C–N) at 1160.17 cm−1. The stretching absorbance ν (C=O) is observed at 1659.52 cm−1 infers the amide group and a sharp stretching absorbance band at 1011.45 cm−1 indicates C-O stretching for cyclic C-O-C linkage of furfural. |
 |
| 13C and 1H NMR spectroscopy: |
| NMR spectra of all the FUR samples synthesised via aqueous and heterogeneous phase as well as via catalyzing various zeolites were corresponding to the same substance. 13C NMR spectrum as described in Figure 3 shows the signal assigned to a CH aliphatic carbon at 110.05 ppm (CH). Signals corresponding to furan rings are observed at 77.26 ppm for (CH)/C3; 77.58 ppm, (CH)/C4; 77.78 ppm, (CH)/C5 and 77.91 ppm, C (quaternary)/C2. |
| Only one signal is observed at low magnetic field, 159.91 ppm, for C/C=O, indicating that all carbonyl groups in CP present the same chemical shifts or that they are very close. |
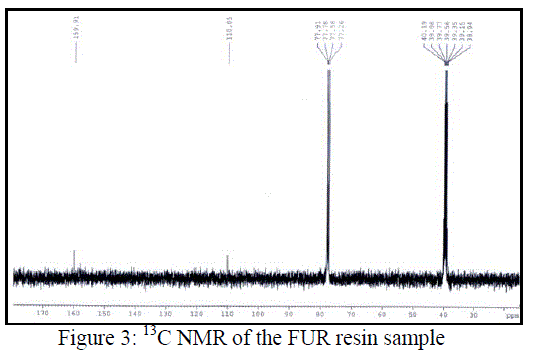 |
| In 1H NMR spectrum Figure 4, the wide singlet signal with unit intensity at 5.54 ppm whose intensity diminishes markedly with addition of D2O, is assigned to –NH. Since protons from NH– groups are in the same chemical environment and involve in polymeric linkage they resonate together with higher intensity at 5.26 ppm. The intensity of this signal also decreases in the presence of D2O. Moreover, signals of –CH proton neighbour to NH groups is observed as a deshielded singlet at 7.391 ppm. Proton on carbon adjacent to oxygen of the furan ring is observed as doublet between 7.708 and 7.704 ppm with coupling constant 1.6 Hz. The other aromatic protons of furan ring are obtained between 6.878 to 6.28 ppm. |
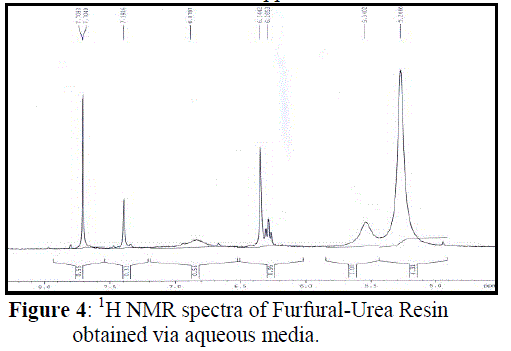 |
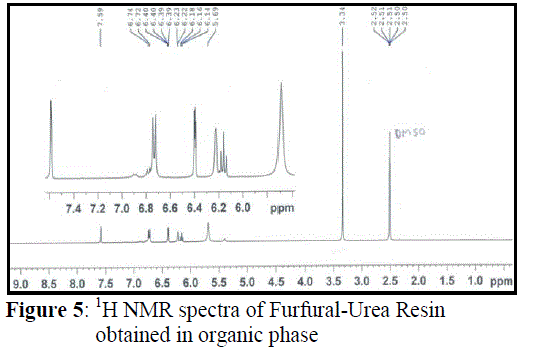
| The 1H NMR spectrum of FUR obtained via organic phase is similar to the spectrum of FUR obtained via aqueous media (Figure 4) and is described in Figure 5. The wide singlet signal at 5.69 ppm is assigned to –NH proton. Since protons from –NH groups are in the same chemical environment, they resonate together between 6.18-6.14 ppm with 8 Hz J value. The intensity of these signals also decreases in the presence of D2O. Signals of –CH proton neighbour –NH groups obtained as doublet at 6.74 and 6.72 ppm and the coupling constant value has same value of 8 Hz indicating presence of NH as an adjacent group, confirming the CH-NH linkage of Furfual-Urea. Signals corresponding to the furan ring are observed between 7.59-6.22 ppm. The proton on the carbon next to the oxygen in furan ring is observed as a singlet at 7.59 ppm due to involvement in hydrogen bonding and proton on adjacent carbon to it appears as double doublet between 6.40-6.39 ppm. |
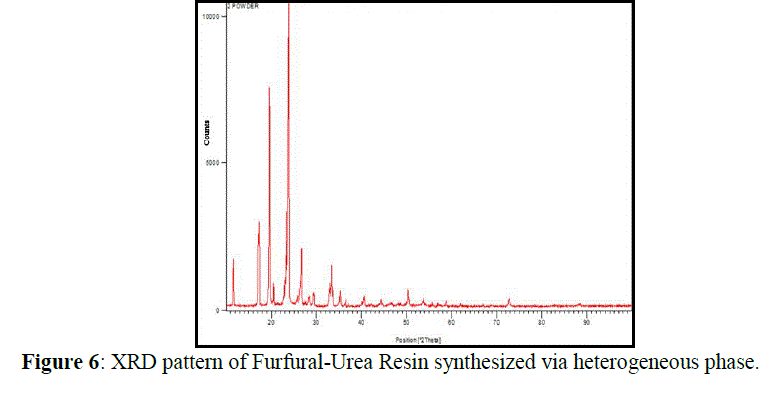
| X-ray diffraction: |
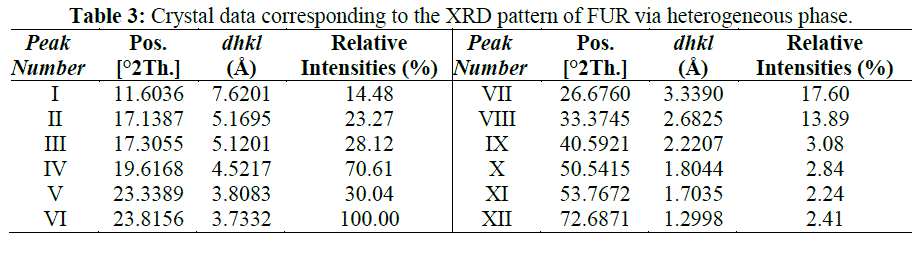
| Figure 6 show the XRD patterns of the FUR prepared in heterogeneous phase. According to these patterns, the higher crystallinity (the smaller peak width) is observed in the FU resin obtained from the reaction in the heterogeneous phase. Table 3 collects the crystal data (interplanar distances and relative intensities) corresponding to the XRD pattern of FUR sample obtained in a heterogeneous phase. |
 |
| The reaction in the aqueous phase always produces low crystalline products independently of the U: F ratio used. It seems that the higher reaction rate in the heterogeneous phase when compared to the aqueous one inhibits the formation of a well-crystalline material. In the pattern of UF-polymer sample, the reflection at 20.5 2(θ) is present; the main reflection at 23 2(θ) is somehow shifted to 23.8 2θ value; the reflection at 6.5 2(θ) disappears, and the intensity ratio of the doublet at 11.5 2(θ) changes. Such a result is not surprising since polymorphism is not rare within organic compounds. The most outstanding conclusion from XRD data is that independent of the preparation methods employed, the condensation of urea with furfural yields a crystalline product of low molecular weight and defined stoichiometry. Moreover, the syntheses in the heterogeneous phase renders a more crystalline and pure product. |
 |
| Thermal analysis: |
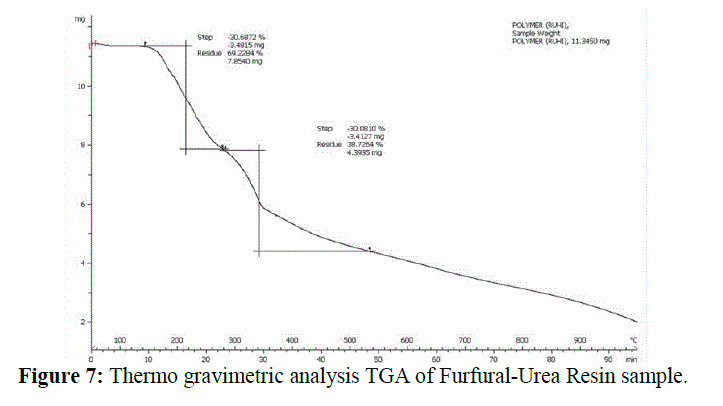
| Thermo gravimetric analysis: |
| `Thermal analysis of FUR was done by analysing sample for thermo gravimetric analysis TGA and differential scanning calorimetry DSC, registered in N2 atmosphere, and differential thermal analysis DTA recorded in air. TGA values indicates that UF resin is stable only till 175 °C, as only up to this temperature it is present as a solid mass with a loss of 5% between 50 and 100 °C, which is attributed to sample’s humidity. A further major loss in the mass of the sample was observed at 335 °C as shown in Figure 7. |
 |
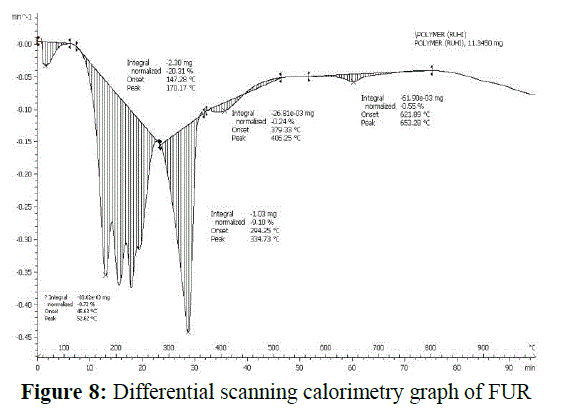
| Differential scanning calorimetry |
| The DSC of FUR was carried out using 11.345 g FUR sample with a heating rate of 20 ïÃâðC/min as shown in Figure 8. Two defined endothermic peaks are observed in DSC. The first endothermic peak at 52.62°C was caused by the elimination of water of crystallization. The first major endothermic peak at 170.17 °C corresponds to the melting point of UF resin which is accompanied by 20.31 % of mass loss. After which a furthermore 30% overall mass loss brings the second endothermic peak at 334.73 °C associated with 9.10 % of mass loss. Broad melting peak, because of the size distribution of crystallites, indicates the partial crystalline nature of the polymer. Furthermore the enantiotropic solid-solid transition, which rarely occurs, is reversible and gives rise to bell-shaped (Gaussian) peak specifying the fine crystalline powder nature of polymer. |
 |
| Differential thermal analysis: |
| These exothermic effects are also observed in the presence of air according to DTA, suggesting that they correspond to an oxidative degradation process. As the temperature increases the FUR sample eventually reaches its melting temperature at 171.55 °C and decomposition temperature at 339.68°C. The large exothermic effect produced by the oxidation, i.e., combustion of sample residues is observed in DTA around 500 °C as shown in Figure 9. |
| FUR obtained via aqueous and heterogeneous media, both are compared through their thermo gravimetric analysis as shown in Figure 10. FUR synthesized via heterogeneous phase represented the melting point by 12.9% weight loss at somewhat higher temperature i.e, 218 ± 0.5 °C, but low crystalline FUR samples obtained by synthesis in aqueous medium gave melting points at 177 ± 0.5°C by losing 5.41% weight. Furthermore, FUR synthesised in a heterogeneous phase showed initial weight loss at 75°C in TGA which apparently appears as melting at first sight. This weight loss at 75°C in fact is caused by the elimination of water of crystallization (mass loss of 18.6 ïÃÂÃÂg). With the loss of water of crystallization from the FUR obtained in heterogeneous phase, the degradation of FUR become rapid upto its melting point at 218 °C and then decomposes but FUR via aqueous phase decomposes only after its melting at 177°C. These decomposition patterns may help in deciding the crystalline nature of heterogeneously synthesized FUR, as with the loss of crystalline water the degradation gets rapid due to loss of crystallinity. |
 |
CONCLUSIONS |
| The condensation of furfural with urea under the zeolite catalyzed experimental conditions of this work yields a pure white product independent of the solvent used with a reduced reaction time and enhanced yield. The quality, yield and purity of Furfural-urea resin (FUR) obtained in this work using such environmental friendly catalysts were not attained earlier and it is an evolutionary green technique developed providing FUR without any dark color. Any intermediate product possibly will react quickly and would not be detectable. IR and NMR spectral studies confirm the FUR formation. The XRD, TGA and DSC analyses conclude that FUR is crystalline and this property depends on the syntheses and experimental conditions employed, that is, it diminishes as reaction medium changes from heterogeneous to aqueous. A crystallization temperature of FUR is obtained at 102 1 °C. Amongst the natural and synthesized zeolite employed as catalyst, stilbite provides highest yield in both the mediums. Looking at the quantity and quality of product and the eco-friendly, simplicity, recyclability and pace of the catalyst used, the protocol described in this paper is dynamic in green synthesis of high quality Furfural-urea resin. |
References |
|