ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
X. Zhu1, J. Gao, J. Lang, H. Zhang, Y. Zhu2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Di-(2-ethylhexyl) phthalate (DEHP) is an endocrine chemical, and has been widely used as a plastizer in polymer materials. In the present paper, DEHP was directly extracted from polymer films with micro liquid phase on a carbon paste electrode composed of graphite powder and deep eutectic solvent, and monitored by open circuit potential method. The micro extraction process follows an exponential association function with the apparent first order rate constant of 6.35×10-4 s-1. The potential responds to the amount of DEHP in the polymer films was described as the Nernstian equation with 31.0 mV/decade of slope, and used in the detection of DEHP. Five polymer film samples were tested by this method, the reasonable results were obtained
Keywords |
| Liquid phase micro extraction; Open circuit potentialïÿýïÿýDi-(2-ethylhexyl) phthalate; Carbon paste electrode; Deep eutectic solvent |
I. INTRODUCTION |
| Di-(2-ethylhexyl) phthalate (DEHP) is the most commonly used plasticizer such as polyvinylchloride (PVC), rubber, cellulose and styrene, which are used in number of packaging materials and tubings for production of foods and beverages. It had been found that DEHP is an endocrine chemical and damage the liver, kidney, even cause cancers [1- 5]. So the detection of DEHP in environment and in polymers and the polymer products is a very important task in environmental protection, industries and researches. The commonly recommended methods for the analysis of DEHP are chromatography related methods such as gas chromatography, high performed liquid chromatography [6-8]. |
| In the most cases, DEHP as a plasticizer distributed in a solid polymer cannot be determined directly, so it needs a suitable sample preparation method in common. As we know, liquid-phase micro-extraction (LPME) is a green sample preparation method for a kind of substances from complex sample matrices. Several kinds of LPMEs have been developed [9-11], and combined with chromatography and electrochemical methods [12-13]. Carbon paste electrode is a two phases system including a solid phase of graphite powder as the electron conducting material, a organic phase of silicone oil as the binder to adhere graphite powder particles together into a paste and prevent it releasing into contacting solution. The carbon paste electrode is contacting with aqueous solution including analytical targets, and composed into a three-phase detecting system[14-15]. The graphite powder and the organic liquid can also be considered as solid phase micro extractant and liquid phase micro extractant in electroanalytical chemistry [13,16]. DEHP is an electrochemical inactive species, therefore the common used amperometric methods cannot be used, but potentiometric methods such as open circuit potential, or other potential measurement are suitable for the detection after extraction. Open circuit potential method not only serves in the detection of substance with or without electrochemical activity [17-18] with higher sensitivity and applicability, but also gives out the dynamic information about chemical process occurred at the electrode surface [5]. Another important point is the open circuit potential measurement only needs two electrodes one working electrode for response to extraction process and a reference electrode for potential reference, so that it can also simplify electrochemical measurement system. |
| In order to get the fast responds of the electrode to the extraction process, the electrode must be contact tightly to the polymer film on a glass plate or the polymer plane surface with a thin layer of extractant liquid. The thin layer extractant liquid must be the same liquid as that composed of carbon paste with conductance in some extent, and contacts with the two electrodes. So a new carbon paste electrode holder is need to be designed with the two electrode contact with each other through the carbon paste and the thin layer organic liquid, as well as the reference electrode. In order to meet the needs of all these requirements, a room temperature liquid salt, deep eutectic solvent (DES) composed of urea and choline chloride solids mixture [ [19-20] was chosen as the organic liquid for carbon paste electrode buildup and liquid phase micro extractant [21]. In the present paper, the liquid phase micro extraction and determination in different polymer samples of DEHP was studied by open circuit potentiometry, some interesting results were reported here. |
II. EXPERIMENTALS |
| A. Instruments and reagents |
| The electrochemical experiments were carried out on CHI620B electrochemical working stationïÿýïÿýChina, CHI Co.ïÿýïÿýwith two electrodes system, a home made carbon paste electrode as working electrode, and home made silver/silver chloride electrode with DES liquid as the reference electrode, and all the potentials reported here were respect to this reference electrode. |
| The regents used in this study including urea, choline chloride, DEHP, All of which were analytical pure (purchased from Shenyang Chemical Co.). Graphite powder (200# chromatographic pure) and methyl silicone oil (chromatographic pure) were purchased from Shenyang Chemical Co. All solutions were prepared with DES prepared from urea and choline chloride solids by hand. |
| The deep eutectic solvent (DES) was prepared by mixing urea and choline chloride solids (the weight ratio was about 1:2) in a small glass beaker with a glass bar into a liquid. |
| B. Fabrication of basic carbon electrodes |
| The solid carbon electrode was prepared with epoxy resin, polyamide resin and Graphite powder (200#) mixed at weight ratio of 8:2:2 into a paste, the paste were filled into a clean glass tube with inner diameter of about 5mm. A glass capillary (about 0.5 mm in diameter) was inserted and passed through the paste along with inner wall of the glass tube before the paste solidified, and a piece of copper wire was inserted into the paste from the same end of the glass tube with the capillary near the centre of the tube as the lead of the working electrode. After the carbon paste solidified in air for 1 hour, the capillary was taken out carefully from the glass tube and left a chamber for the reference electrode. After the electrode solidified in air for about one week, a small cavity (about 1 mm in depth) was tipped out from the another end of electrode as the room for the carbon paste. |
| C. Preparation and modification of carbon paste electrode |
| The carbon pastes were the uniform mixture of graphite powder and DES liquid as the binder at the weight ratio of 2:1, and filled into the basic electrode cavity tightly, polished on a piece of glassy paper into mirror, and served as the working electrodes. |
| D. Preparation of reference electrode |
| A piece of capillary with diameter of 0.5 mm was cut out at one end and a piece of platinum wire was sintered into the capillary at this end, polished on a sand-paper into a smooth end. DES liquid was filled into the capillary from the other end, and inserted a silver chloride covered silver wire into the capillary, set the capillary into the reference through the reference electrode channel and reach the surface of the electrode. |
| E. The sample measurement procedure |
| A small drop of DES liquid was put on the polymer surface, and set the newly prepared carbon paste electrode stand in the drop, and left small amount of liquid DES between the electrode surface and polymer surface. After about 10 min, then perform the open circuit potential experiment for 6000s, the potential-time curve was obtained. |
III. RESULT AND DISCUSSION |
| A. The open circuit potential curve of DEHP |
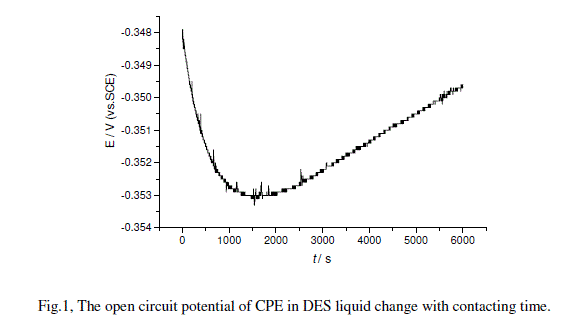
| A new prepared electrode was set up in one drop of DES liquid on a clean glass plate, and open circuit potential experiment was then performed. The recorded potential-time curve was shown in Fig.1. The potential sharply decreases with time from initial point of -0.348 V to the lowest point of -0.353 V at 1500 s, and then slowly increased almost linearly to -0.350 at 6000 s. Even the potential changes were not very much, but it is the responds of the electrode to the molecules and ions liquid. The CPE was mode of DES and graphite powder, so the chemical potential of DES in CPE is lesser than that of chemical potential of DES in DES liquid, and CPE shows a lower potential than the reference electrode of silver/silver chloride electrode. |
| The measured potential deference between CPE and reference electrode was a negative potential at the initial point. DES liquid is composed of urea and choline chloride. Choline includes an anion of chloride and a quaternary cation. Compared with quaternary cation, the chloride anion has smaller size and larger mobility, and diffuses to the electrode surface faster than the quaternary ion. So a negative charged layer first setting up at the CPE electrode surface. The first part of the curve can be regressed into a quadratic equation as |
| -4 1/ 2 -6 2 -4 =-0.34174-2.7209.10 +2.7233.10 E t t;R =0.9889,SD=1.4272.10(1) |
 |
| In this regression equation, the second term has a larger coefficient than the third one, which means the process was mainly controlled by the non-linear diffusion of ions to the electrode surface. The second part of the curve can be regressed into another quadratic equation as, |
| -5 1/ 2 -6 2 -5 E=-0.35242-7.6054.10 t +1.4603.10 t;R =0.9962,SD=6.6123.10 (2) |
| Compared with the equation (1), the coefficient of the second term is 50 times smaller, but the third coefficient is 2 times larger. This result indicates the nonlinear diffusion process was replaced by the linear diffusion, which related to the re-orientation of the ions. This curve well describes the formation of double layer at the carbon paste electrode surface contacting with pure DES solvent, and the distribution of ions of chloride and quaternary cation. |
| The newly prepared CPE electrode was set up in a drop of DES solution including 1.00 mg/g DEHP, and open circuit potential experiment was performed as described before. The obtained potential-time curve was substrate shown in Fig.2A.The open circuit potential at initial point of -0.3552 V sharply decreases to the lowest point of -0.3583 V at 200 s and slow increases to -0.3349 V at 6000 s. All the potentials are negative due to the same reason as described before. |
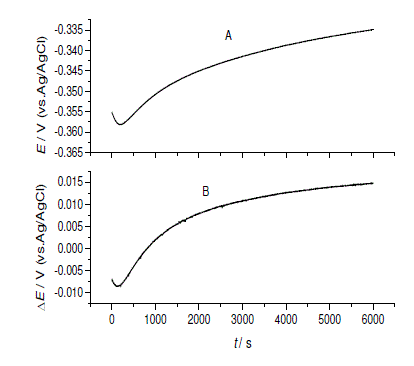 |
| Fig.2, A. the open circuit potential of CPE in DES solution including 1.00 mg/g DEHP change with contacting time, and B. the open circuit potential difference between of CPE in DES solution including DEHP and without DEHP. |
| In order to reduce the influence of the background and obtain the real responds of the CPE to the DEHP in the solution, the potential difference between DEHP in DES solution with DEHP and in DES was plotted against the contacting time as shown in Fig.2B. |
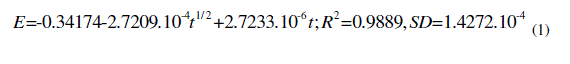 |
| This equation can also serve as the dynamic equation of the CPE response to sample solution of DEHP, from which the apparent first-order extraction rate constant was obtained as 6.35×10-4 s-1. |
| B. The influence of DEHP concentration in DES |
| The one drop of DES solutions with different amounts of DEHP was put on a clean glass plate. The electrode was stood in the solution on the plate. The open circuit potential experiments were performed as before. The potential difference at 6000 s was plotted against the amount of DEHP in the solutions, and a linear line was obtained as shown in Fig.3. The line was regressed into a linear equation as, |
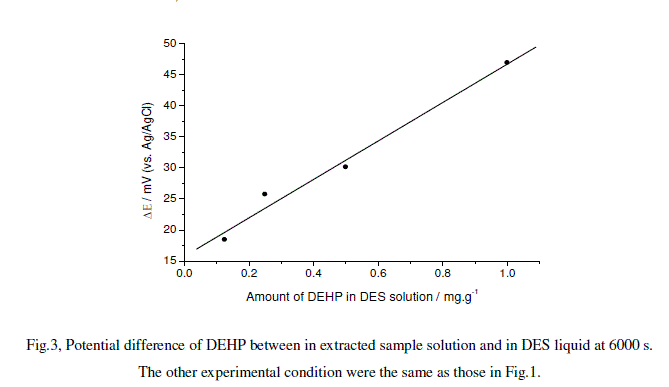 |
| Fig.3, Potential difference of DEHP between in extracted sample solution and in DES liquid at 6000 s. |
| The other experimental condition were the same as those in Fig.1. |
| From thermodynamics, the chemical potential difference of DES solutions before and after extraction of DEHP from the polymers can be expressed as, |
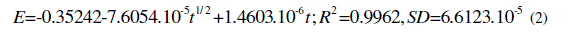 |
| compared with equation 4, we found n=2. This result means the equlibrium potential difference follows Nernstian equation, and can be used to determine the amount of DEHP extracted from the polymer into DES solution. |
| C. The micro extraction and determinations of DEHP in different plastic films |
| DEHP as a common plastizer was added in many plastic products especially in the plastics of PVC. Some plastic films or plates were chosen as the samples for the test of the method. A plastic film was put on a glass plate, and one drop of DES liquid was dropped on the film surface to extract the DEHP for 10 min, and then perform the open circuit potential experiments[11]. The potential difference at 6000 s was used to calculate the amount of DEHP extracted from the plastic film by the equation (4). The calculated results were plotted in Fig.4. |
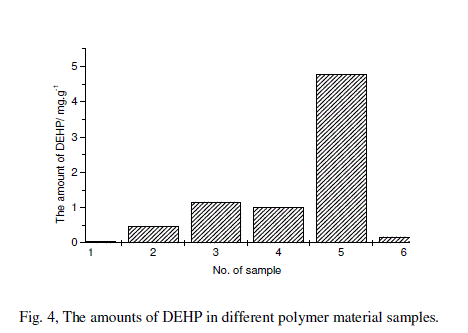 |
| 1, food fresh-keeping packing PE-film; 2, bread packing bag; 3,moon cake packing box; 4, 1.00 mg/gstandard solution; 5, apparent cloth packing box;6, unapparent instrument packing box. |
| These results indicate that PE-film for food fresh-keeping film and unapparent instrument packing box show no DEHP at all, because they are made of PE do not need to additives. The apparent cloth packing box is need to apparent and soft so it was added too much amount of DEHP, it is similar to the moon cake packing box. |
IV. CONCLUSION |
| In this paper, a liquid phase micro extraction on carbon paste electrode with DES and open circuit potential methods was combined for the detection of DEHP from polymer samples. The results were summarized as the followings. (1) DEHP can be extracted into micro liquid phase in carbon paste electrode composed of urea and chlorine chloride, and follows an exponential association dynamic function; (2) the amount of DEHP extracted from polymer products can be estimated from the equilibrium potential difference between sample and background at 6000 s, which was a linear relationship with the concentration of DEHP in the DES solutions; (3) The analysis results show that the amount of DEHP in PE-film of food fresh-keeping packing was the lowest one, but the amount of DEHP in apparent cloth packing box was the highest one among the 5 samples, which are reasonable from the materials and commercial usages of DEHP. |
ACKNOWLEDGMENT |
| The author would like to acknowledge the financial supports of the Chinese National Science Foundation (20875063), Liaoning education minister Foundationïÿýïÿý2004-c022ïÿýïÿýand Shenyang Sciences and Technology Bureau Foundation (2007-GX-32). |
References |
|