ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
P. Balashanmugam1*, S. Santhosh1, H. Giyaullah1, M.D. Balakumaran1, P.T. kalaichelvan2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The development of appropriate processes for the synthesis of nanoparticles is an important aspect of nanotechnology. In this study, fungus mediated synthesis of silver nanoparticles using Microporus Xanthopus has been done. The dried macro fungi were powdered and boiled for 20 minutes to get the extracts. The extracts were treated with silver nitrate and kept in the dark conditions for 48 hrs, for the appearance of colour change. The Plasmon peak was observed at 425 nm. These nanoparticles were examined for the stability for a period of 2 months and were further characterized by SEM and FTIR. SEM analysis revealed the spherical nature of silver nanoparticles and size distributed in the range of 40 nm. However further studies regarding XRD, SEM, EDX, are confirmed to strengthen the existing findings. The synthesized silver nanoparticles have been investigated against the pathogenic bacteria Bacillus subtilis , Staphylococcus aureus, Pseudomonas aeruginosa , Escherichia coli , Klebsiella pneumoniae and shigella sp for their antibacterial activity and minimum inhibitory concentration
Keywords |
| Mycosynthesis, Microporus Xanthopus, Silver nanoparticles, Stability, Antibacterial. |
I. INTRODUCTION |
| Nanotechnology is emerging field of science which involves synthesis and development of various nanomaterials and it has recently become one of the most active research fields in technology and engineering. From last decade, application of nanomaterial has been extensively increased and the high need leads to the bulk production. Classically the nanoparticles are produced by physical [1] and chemical methods, [2] as these methods are costly, toxic and non eco friendly; scientists are looking forward to synthesize low cost, non toxic, eco friendly nanoparticles. Recently, biosynthesis of nanoparticles using bacteria [3, 4], fungus and plants have emerged as a simple and viable alternative to more complex physical and chemical synthetic procedures to obtain nanomaterials. In nanoparticles where the number of atoms on the surface is comparable to the total number of atoms play a decisive role in their catalytic, optical and electromagnetic properties [5-8]. Catalytic properties make metallic nanoparticles a system of choice in many biological applications, such as drug delivery, biosensors, and antimicrobial agent [9, 10]. |
| Silver is medically considered as one of the most powerful elements due to its activity against mammalian tissues where it acts as an antiseptic agent [11]. Silver ion it’s metallic as well as ionic forms exhibits cytotoxicity against several microorganisms and hence used as an anti microbial agent [12]. Some microbes can accumulate silver by adsorption. Silver nanoparticles have been exploited for their unique properties and their vast applications in biomedicine. Now the anti microbial activity of silver nanoparticles has been well established and they are preferred to posses anti inflammatory, anti viral, anti platelet and anti fungal activity [13]. It was observed that the biomass suspension has a yellow color before reaction with the silver ions and brown color on completion of the reaction. The mechanism of leading to formation of silver nanoparticles is not definitely understood at the moment. This change in color is due to the excitation of the surface Plasmon vibrations in the metal nanoparticles, as suggested by [14]. Silver nanoparticles are the metal of choice as they have the capability to kill microbes effectively. The strong toxicity of silver against wide range of microorganisms is well known and silver nanoparticles have shown to be an effective antimicrobial agent. |
| The present study has focused on following steps: |
| 1. Synthesis of silver nanoparticles and proceeding with the fungi showing good stability without any precipitation. |
| 2. Characterization studies were done using SEM EDAX, FTIR and XRD. |
| 3. Antimicrobial activity and Minimal Inhibitory Concentration was carried out against six pathogenic bacteria. |
II. EXPERIMENTAL WORK |
| In this study five of the macro fungi namely Lentinus squarrosulus , Microporus xanthopus , Pycnoporous sanganeus, Polyporus grammocephalus, Daedalea sp. were collected from the forest Ponda hills, Goa, India in the month of November 2012. The samples were cleaned thoroughly using distilled water to remove dust particles adhering the surface of these fungi. The washed macro fungi were then kept in Shadow condition for complete drying, at a period of 4 to 5 days. The dried macro fungi were cut into small pieces and were powdered into fine particles. 4 g of the Powdered fungi was added to 100 mL of distilled water and boiled for 20 min at 55°c.The Crude extracts were filtered through Whatman filter paper and the filtrates were stored in Conical flasks at 4°c for later use. |
| In a this production scheme of silver nanoparticles, 2 mL of the extract was added to 8 mL of glass distilled water making the solution 10 mL in a test tube. For synthesis of silver nanoparticles, AgNO3 1mM solution of the final concentration was mixed with 10mL of filtrate in test tubes and kept in the dark conditions for 48 hrs. The filtrate without treating with silver nitrate was maintained as control in another set of test tubes for the experiment. The first step involves trapping of the Ag+ ions onto the surface of the fungal cells. It can be realized by electrostatic interaction between Ag+ and a negative charged carboxylate groups on the cell surface. |
| The antibacterial assays were done on bacterial organisms like Escherichia coli (E.coli) Klebsiella pneumoniae, Bacillus subtilise, Shigella.sp. Pseudomonas auriginosa, and Staphylococcus aureus by well diffusion method. The well diffusion test was carried out by using Muller Hinton Agar. The inoculum was prepared in sterile nutrient Broth and the tube was incubated at 370C until the turbidity was reached up to 0.5 McFarland standards [NCCLS (National Committee for Clinical Laboratory Standards), M100-S12 Performance Standards for Antimicrobial Testing: Twelfth Information Supplement, 2002]. The Muller-Hinton agar plate was inoculated with 5μl of inoculums by spreading the swab over the plate. Then, agar was punched with the help of sterile borer to create 6 mm well.40μl of fungal extract ,1mM of silver nitrate 40 μl ,40μl of Synthesized silver nanoparticles and 40 μl of antibiotic (Streptomycin) were added in respective wells. Streptomycin was used as a positive control. All plates were incubated at 37°C for 24 hrs. Zone of inhibition was measured after incubation with Hi-Media scale. The experiments were performed in triplicate. |
| The microbial activity of silver nanoparticle was checked by determination of Minimum Inhibitory Concentration. Bacterial suspension was prepared and swabbed on the medium then wells were made on the plates. Different concentrations of the silver nanoparticles (10, 20, 30, 40 μl) were added to different wells. All the plates were incubated at 37°C for 24 hrs in order to determine the inhibitory growth of the silver nanoparticle on particular pathogen. The MIC was defined as the lowest concentration of the nanoparticles that inhibited the visual growth of the test organisms. |
| The reduction of silver ions from AgNO3 to silver nanoparticles was monitored by measuring the absorbance as a UVvisible spectrum of the reaction mixture on a HITACHI U-2900 UV-Visible spectrophotometer. The absorbance was recorded from 300 to 700 nm for detection of mycosynthesized silver nanoparticles. The lyophilized sample was subjected to FTIR Spectroscopy analysis. 10mg of the sample was mixed with 200 mg KBr (FT-IR grade) and pressed into a pellet. The sample pellet was placed into the sample holder and FT-IR spectra were recorded in the range 4000- 450 cm–1 in FT-IR spectroscopy at a resolution of 4 cm-1. The spectrum for FTIR was scanned on a Perkin Elmer Fourier Transform Infrared Spectrophotometer at a resolution of 4 cm–1. After lyophilisation of the purified silver nanoparticles, the structure, composition, and average size of the synthesized silver nanoparticles were analysed by scanning electron microscopy (HITACHI SO-6600, Japan). Energy-dispersive x-ray (EDX) spectroscopy analysis for the confirmation of elemental silver was carried out for the detection of elemental silver. A scanning electron microscope (HITACHI SO-6600, Japan) was used to record the micrograph images of synthesized Ag-NPs. The silver nanoparticles solution thus obtained was purified by repeated centrifugation at 10,000 rpm for 20 min followed by redispersion of the pellet of silver nanoparticles into 10 ml deionised water, after freeze drying the purified silver nanoparticles were analysed by XRD Using SEIFERT JSO- DEBYEFLEX 2002 model. XRD help to determine the crystalline domain size. |
III. RESULTS AND DISCUSSION |
| After screening among the five fungal extracts Microporus xanthopus (Figure 1) fungal extract showed good stability and rest of the four mushrooms showed aggregation and no stability. It is well known that silver nanoparticles exhibit yellowish - brown colour in aqueous solution due to excitation of surface Plasmon vibrations in silver nanoparticles. [15]- [17] |
 |
| Silver nitrate is used as reducing agent as silver has distinctive properties such as good conductivity, catalytic and chemical stability. My experiment work is focused on Mycosynthesis of nanoparticles from Microporus xanthopus, two tubes one containing the filtrate of the Microporus xanthopus fungal extract (control) and the other with fungal extract treated with AgNO3. The absorption spectrum of the brown silver nanoparticle solution prepared with the proposed method showed a surface Plasmon absorption band with a maximum of 425 nm, indicating the presence of spherical silver nanoparticles. The two peaks in (Fig 2) shows that the fungal extract had no Plasmon absorption band, whereas the synthesized nanoparticles had a Plasmon absorption band of 425 nm after 48 hrs. |
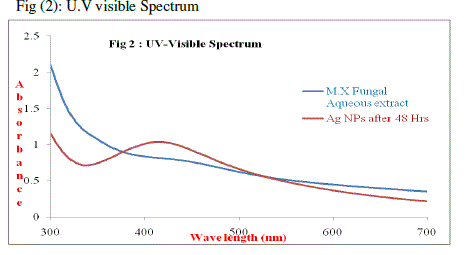 |
| The confirmation of formation and stability of the silver nanoparticles in the Silver nitrate treated sample was monitored by using UV–Vis spectral analysis, (Fig 3) for which after completion of reaction (0-48 hrs) aliquots of the reaction sample were removed and subjected to UV–Vis spectroscopy measurements. At 0 hrs there was no peak observed, at 8 hrs a small peak started between 390 to 400 nm, after 12 hrs a there was an increase in peak to 413 nm, |
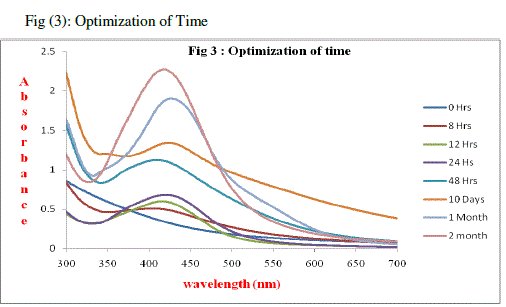 |
| After 24 hrs time interval the peak shifted to 418nm, by 48 hrs the peak was observed as 425 nm, after ten days the sample was again observed and the peak was stable at 424 nm, the same value was constant for 1month and finally after 2 months, the U.V spectrum was taken and we got a peak at 422 nm. It is clearly observed that the peak at all the different time duration (i.e., 8hr,12 hr ,24 hr,48hr, 10 day, 1 month,2 months) had by the peak range between 390 and 422 nm in every successive reading. The above results conclude that the silver nanoparticle activity was started at 12 hrs and it was ended on 48 hrs and after that its activity was stable for a period of 2 months. |
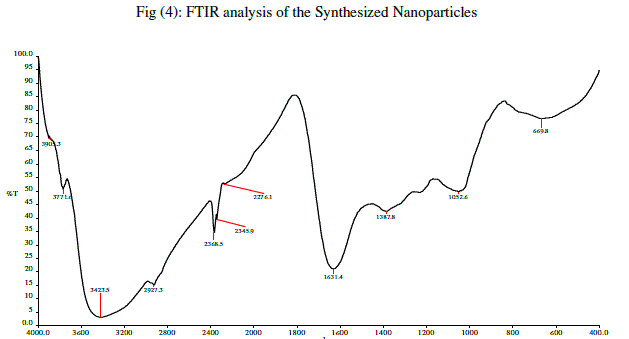 |
| In (Fig.4) Silver nanoparticles showed the presence of eleven bands at 3905.3 ,3771.6 ,3423.5 ,2927.3 ,2368.5 ,2345.9 ,2276.1 ,1631.4 ,1387.8 ,1052.6 and 669.8 cm–1 (Fig.4). The band at 3423 cm–1 has been identified as O–H stretch (alcohols, a broad, strong band), which arose due to carbonyl stretch and. The bands at 2927 and 1631 developed for C–H stretch (alkenes) and C=C (alkenes) stretch stretching respectively and were commonly found in the proteins indicating the presence of proteins as ligand for Ag-NPs, which increases the stability of nanoparticles synthesized. The representive spectra of nanoparticles obtained manifests absorption peaks located at about 3843..68 cm-1(N-H group of amines) 3597.73 cm-1 (OH-Group of phenols), 2080.65 cm-1 aromatic CH stretching and 767.16 cm-1 (CCl). Therefore from the FTIR results it’s clear that the synthesized nanoparticles were surrounded by proteins and amino acids which may be responsible for the stability of the Silver nanoparticles. From the analysis of FTIR studies we assume that the functional groups of alcohols, aldehydes and carboxylic acids present in the sample may be responsible for the reduction of silver nitrate to silver nanoparticle. |
| Mcyosynthesized silver Nanoparticles were subjected to the SEM analysis in which the SEM photos of Silver nanoparticles (Fig. 5) clearly shows that in the room temperature synthesized sample of Microporus xanthopus, the size diameter of the nanoparticles lie between 30 nm to 50 nm, and the shapes were observed as spherical. The inset in the (Fig. 5) shows that the most of the particles are found to be in spherical shape and averagely distributed in a 40 nm range. The Silver Nanoparticles of spherical shapes will contribute to the variation of the size as can be understood by SEM image. |
 |
| The energy dispersive X-ray analysis (EDX) reveals strong signal in the silver region and confirms the formation of silver nanoparticles. The element analysis of the silver nanoparticles was performed using EDX on the SEM. The freeze-dried silver nanoparticles were mounted on Aluminium foil. (Fig 6) shows the EDX spectrum of spherical nanoparticles prepared with this bioreduction method. The peaks around 3.40 keV correspond to the binding energies of AgL. The results indicated that the reaction product was composed of high purity silver nanoparticles. |
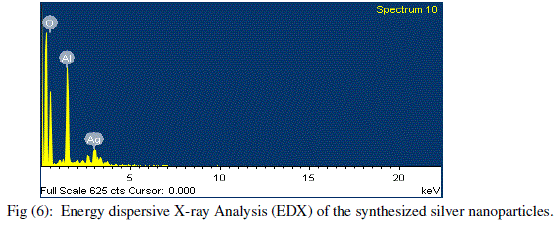 |
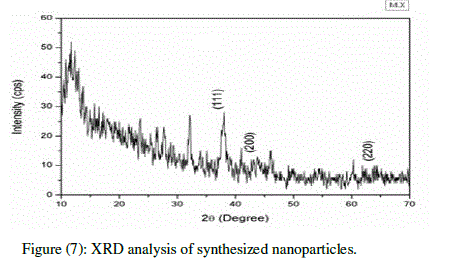
| The dry powders of the silver nanoparticles were used for XRD analysis. The diffracted intensities were recorded from 10 âÃâæ to 70 âÃâæ at 2 theta angles. The diffraction pattern in (Fig 7) corresponds to pure silver metal powder. Subsequent analysis of Silver nanoparticles precipitate by powder-XRD revealed four peaks at 38° (111), 44° (200) and 64° (220). |
| The peaks were compared with X-ray diffraction database. The particle size histogram obtained silver nanoparticles shows broad distribution of particle size. The size particle ranged from 30-50 nm and the average particle size comes out to be 40nm. The X-ray diffraction patter obtained for silver nanoparticles synthesized by fungal extract shows that the silver nanoparticles are crystalline in nature. |
 |
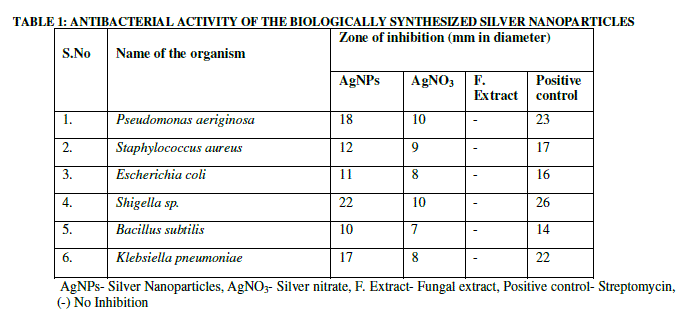
| The antibacterial effects of biologically synthesized silver nanoparticles have been investigated against six pathogenic bacteria. The test was performed by loading the biologically synthesized nanoparticles into the well B followed by silver nitrate in well A , then Antibiotic(streptomycin) in well C and fungal extract in well D (Fig. 8), demonstrates zones of growth inhibition around the well loaded with silver nanoparticles. It was observed that clear zone of inhibition was shown in all the experimental bacteria (Table 1). |
 |
| This study shows that, Streptomycin has highest antibacterial activity among all the bacteria than synthesized silver nanoparticles. The highest zone of inhibition was shown in Shigella.sp., second mean zone of inhibition was found in Pseudomonas auriginosa, and lowest zone of inhibition was found in Bacillus subtilis in silver nanoparticles (Table 1). |
 |
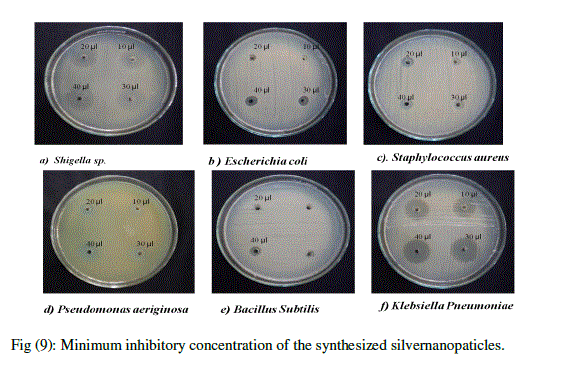
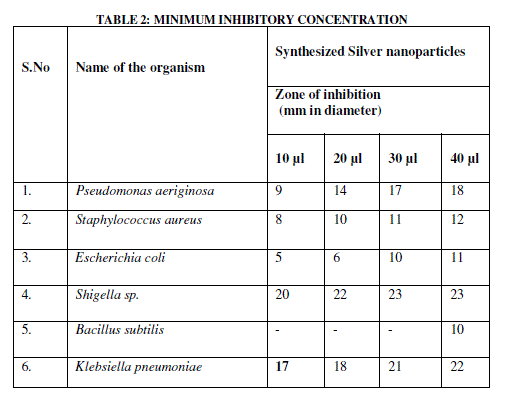
| The synthesized Silver nanoparticles were very effective in controlling the bacterial human pathogens. The Minimum Inhibitory Concentration was checked with experimental Pathogenic bacteria like Staphylococcus aureus, Shigella .sp. Bacillus subtilis, E.coli, Klebsiella pneumonia and Pseudomonas aeroginosa. The silver nanoparticle was used in different concentrations such as 10, 20, 30, 40 μl to check the Minimum Inhibitory Concentration. The silver nanoparticles had MIC of 20 μl on Pseudomonas aeroginosa and, Staphylococcus aureus. Shigella sp. and Klebsiella pneumonia had MIC of 10 μl where as Escherichia coli and Bacillus subtilis had MIC of 40 μl (Fig 9d), (Fig 9e), (Table.2). |
 |
 |
IV. CONCLUSION |
| In this research work the following results were obtained: |
| The synthesized silver nanoparticle had a good stability for a period of two months. The SEM and EDX image shows the average particle size as 40 nm. The XRD analysis confirmed the mean size of the silver Nanoparticles (111), (200) (220) and its crystal in nature. The silver nanoparticle shows more antibacterial activity against Gram- Negative Bacteria Shigella sp., and Pseudomonas aeroginosa. The Minimum Inhibitory Concentration also checked with experimental pathogenic bacteria. The Silver nanoparticles showed the MIC concentration recorded at 10 μl in shigella sp. except all the bacteria. From this it’s clear that the synthesized silver nanoparticle has great potential for applications in catalysis, biomedical, and pharmaceutical industries. |
V. ACKNOWLEDGEMENT |
| We wish to thank Prof. R. Rengasamy, The Director, Centre for Advanced Studies in Botany, University of Madras, Chennai, for providing the laboratory facilities. Our special thanks to Dr. M. Kumar Assistant Professor, Madras Christian College for analysing my fungal samples. We also thanks to NCNSNT, University of Madras, for providing Characterization studies and UGC herbal science scheme, New Delhi, for financial support. |
References |
|