ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Priyanka Das1, Alok Kumar Srivastav2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Andrographis paniculata Nees.,a member of the family Acanthaceae is a plant commonly used by the local people, for the treatment of common cold, liver diseases, snake bite and some skin infection. The aerial part of the plant was screened for their phytochemical properties and antimicrobial activity. Antimicrobial activity of leaf extract of Andrographis paniculata, was studied using different solvent like chloroform, methanol, petroleum ether and water against bacterial strains like Bacillus subtilis, Escherichia coli by disc diffusion method. Methanolic extract showed maximum inhibitory action against Escherichia coli and Bacillus subtilis respectively. Results of the phytochemical screening showed that steroids, phenols, terpenoids, alkaloids, saponins, flavonoids were the active compounds present in the plant. The plant is traditionally used for the treatment of antibacterial, antioxidant, antidiabatic, antipyretic, hepatoprotective and several infectious diseases ranging from malaria to dysentery. The medicinal value of this plant is due to the presence of active ingredients viz andrographolide and neoandrographolide which are derivatives of diterpenoids. It is a potent scavenger of a variety of reactive oxygen species including superoxide anion, nitric oxide, hydrogen peroxide. The anti-oxidant activity was determined by DPPH (2, 2-diphenyl-1-picrylhydrazyl) method. Total antioxidant capacity were determined spectrophotometrically
Keywords |
| Phytochemical Extraction, Andrographis paniculata, Kalmegh, Anti-Bacterial, Anti-Oxidant, Anti- Pyretic, Anti-Diabetic Activity |
I. INTRODUCTION |
| HERBAL MEDICINE AND ITS IMPORTANCE |
| Nature has been a source of medicinal agents for thousands of years and since the beginning of mankind. The application of medicinal plants especially in traditional medicine is currently well acknowledged and established as a viable profession. Medicinal plants are an important source for the therapeutic remedies of various ailments(10). Scientific experiments on the antimicrobial properties of plant components were first documented in late 19th century(9). There is a growing interest in correlating the Phytochemical constituents of a medicinal plant with its pharmacological activity10). |
| ANDROGRAPHIS PANICULATA |
| Kalmegh (Andrographis paniculata) commonly known as “king of bitter” belonging to family Acanthacea, is an important annual medicinal herb widely distributed in Madhya Pradesh, India. It is herdy and erect herb which grows mainly as undershrub in tropical, moist deciduous forest. It is one of the most widely used plant in Ayurvedic formulations(14). It is used to overcome sannipata type of fever, difficulty in breathing, hemopathy burning sensation, cough, skin diseases, fever, ulcer and worms. It is also useful in acidity and liver complaints(1). |
| MORPHOLOGY |
| Andrographis paniculata grows erect to a height of 30– 110 cm in moist, shady places. The slender stem is dark green, squared in cross-section with longitudinal furrows and wings along the angles. The lance shaped leaves have hairless blades mea ruing up to 8 centimeters long by 2.5 wide. The small flowers are borne in spreading racemes. The fruit is a capsule around 2 centimeters long and a few millimeters wide. It contains many yellowbrown seeds. It is also known as Bhui-neem, meaning "neem of the ground", since the plant, though being a small annual herb, has a similar strong bitter taste as that of the large Neem tree. |
| PHYTOCONSTITUENTS |
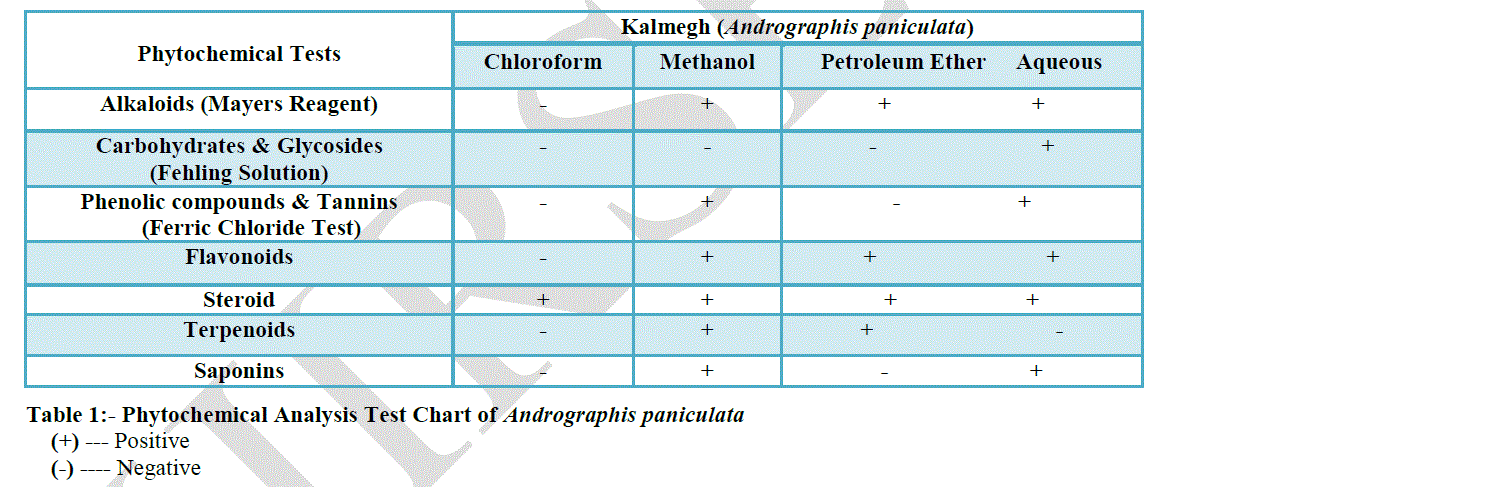
| Medicinal plants contain physiologically active principles that over the years have been exploited in traditional medicine for the treatment of various ailments(2). Andrographis paniculata contains diterpenes, lactones and flavonoids. Flavonoids mainly exist in the root, but have also been isolated from the leaves. Aerial parts contain alkanes, ketones, and aldehydes and the bitter principles in the leaves were due to presence of the lactone andrographolide named kalmegin. Four lactones Chuaxinlian A (deoxyandrographolide), B (andrographolide), C (neoandrographolide) and D (14-deoxy-11, 12-didehydroandrographolide) were isolated from the aerial parts. The leaf and stem extracts were assayed for the presence of glycosides, flavonoids, gums, steroids, terpenoids, tannins, saponins and phenolic compounds(7). The ethanol, acetone, methanol, petroleum ether and chloroform extracts of Kalmegh were screened for the presence of secondary metabolites(7). |
II. RELATED WORK |
| The Plant is widely used in Ayurvedic and homeopathic systems of medicines. The medicinal value of this plant is due to the presence of active ingredients viz. Andrographolide and neoandrographolide which are derivatives of diterpenoids (Anil Kumar et al. 2012). |
| The Phytochemical research and quantitative estimation of the percentage yields of chemical constituents of the plant studied that the leaves and stem were rich in flavonoids, gums, mucilages, saponins, and tannins. They were known to show medicinal activity as well as exhibiting physiological properties (Sofowara et al,1993). Antibacterial assay was carried out by Agar well diffusion method (Perez C et al 1990). The MIC method was applied on extracts that proved their high efficacy against microorganisms by the disk diffusion (Kirby-Bauer) method. The highest dilution of a plant extract still retains an inhibitory effect against the growth of a microorganism is known as MIC (Misra et al, 1978). The inhibitory facts of Andrographis paniculata on the test micro-organism may be due to the presence of the above Phytochemical components. The various Phytochemical compounds detected are known to have beneficial importance in medicinal science. Phenols are said to offer resistance to diseases and accounts for most of the anti-oxidant activity in plants. Flavonoids show anti-allergic, anti-inflammatory, anti-microbial and anti-cancer activities (Okeke et al, 2001). |
| Antioxidant activity of A.paniculata and its constituents have been reported by various investigators which involve nicotine-induced inhibition of mitochondrial electron chain complexes and the resultant increase in nitric oxide (NO) in different parts of rats’ brain was prevented by simultaneous treatment with water and ethanol extracts of A.paniculata, the water extract exhibited greater anti-oxidant activity than the ethanol extract (Das et al, 2005). |
| A.paniculata has been reported as having antibacterial, antifungal, antiviral, choleretic, hypoglycaemic and adaptogenic effects (Kavanagh et al, 1963). Four lactones – chuanxinlian A (deoxyandrographolide), B (andrographolide), C (neoandrographolide) and D (14-deoxy-11, 12-didehyroandrographolide) were isolated from the aerial parts (Chang et al, 1986). |
III. MATERIALS AND METHODS |
| MATERIALS REQUIRED |
| Microorganisms |
| E.coli Culture, Staphylococcus aureus, MTCC (Microbial Type Culture Collection) |
| Animals Used |
| Swiss Albino Mice (25 gm), Wister Albino Rat (150 gm) |
| Chemicals Required |
| 95% Ethanol, Distilled water, Nutrient Broth (NB), Agar, Nutrient Agar Media (NAM), Culture, Herbal Drug powder (Kalmegh), Chloroform, Methanol, Petroleum ether, Fehling solution A & B, Ferric chloride, Mayer’s reagent (Mercuric Chloride, Potassium Iodide), Ninhydrin solution, DPPH (Diphenyl picryl Hydrazine), Sodium Hydroxide, Biuret Reagent, Conc. Sulphuric Acid, Acetic Acid, Dilute Hydrochloric Acid, Alloxan, Paracetamol. |
| Instruments Required |
| Soxhlet Assembly (J-Sil, 50/42, Borosil glass), Vacuum Rotary Evaporator (Scientech), Digital Balance (Denver, Germany), Hot Air Oven (Scientech, 325 L), Laminar Air Flow Chamber Horizontal, Incubator (Scientech), Cyclo Mixer (REMI), Antibiotic Zone Scale, Digital Tele Thermometer. |
METHODOLOGY |
| PHYTOCHEMAICAL EXAMINATION OF DRUG |
| Phytochemical examinations were carried out for all the extracts as per the standard Methods (Brain & Turner 1975, Evans 1996). |
| 1. Plant Constituents ----- Alkaloids |
| Test / Reagent Used ----- Mayers Reagent |
| 2. Plant Constituents ----- Carbohydrates & Glycosides |
| Test / Reagent Used ----- Fehling Solution |
| 3. Plant Constituents ----- Phenolic Compounds & Tannins |
| Test / Reagent Used ----- Ferric Chloride Solution |
| 4. Plant Constituents ----- Flavonoids |
| Test / Reagent Used ----- Alkaline Reagent Test |
| 5. Plant Constituents ----- Phytosterols |
| Test / Reagent Used ----- Liebermann Burchard’s Test |
| 6. Plant Constituents ----- Terpenoids |
| Reagent / Test Used ----- Acidic Reagent Test |
| 7. Plant Constituents ----- Saponins |
| Reagent / Test Used ----- Foam / Froth Test |
| ANTI-BACTERIAL ACTIVITY BY DISC DIFFUSION METHOD |
| The agar disc diffusion method was employed for the determination for anti bacterial activities of the extract of A.paniculata. All bacterial cultures were first grown on nutrient agar plates at 37ºC for 24 hours. Few colonies of similar morphology of the respective bacteria were transferred to nutrient broth and incubated until adequate growth. The inoculations of the respective bacteria were streaked on to the nutrient agar plates. Sterile filter paper discs (5mm) (Watt’s man no.1) were punched and impregnated with petroleum ether, methanol, chloroform and aqueous extracts of A.paniculata. These were then placed on the nutrient agar plates inoculated with S.aureus and B. subtilis. The plates were then allowed to stay for incubation at 37ºC for 24 hours. The assessment of antibacterial activity was based on the measurement of diameter of inhibition zone (mm) formed around the disc. |
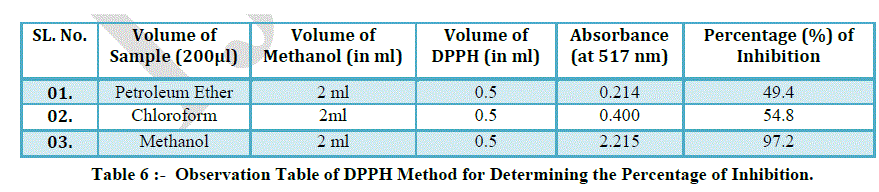
| ANTI-OXIDANT ACTIVITY USING DPPH (Diphenyl picryl Hydrazine) METHOD |
| PREPARATION OF REAGENT |
| DPPH Reagent ----- 4 mg of DPPH was taken & dissolves in 100 ml of Methanol. |
| METHOD |
| 11 clean test tubes were taken and ascorbic acid solution was added to each of the test tubes in an increasing amount from 0.2, 0.4,….. The eleventh test tube was kept blank with no ascorbic acid. Then methanol was added to make the final volume to 2 ml. Then 0.5 ml of DPPH solution was added to each of the test tubes. The test tubes were allowed to stand for the reaction to occur for 10 min in dark conditions. Finally the readings were noted down by the help of UV VIS SHIMADZU 1800 Spectrophotometer at 517nm. In case of extracts obtained from herbal sample same procedure was used .20 μl of the samples were taken & volume was made to 2 ml with methanol. 0.5 ml of DPPH solution was added to each of the test tubes and it was allowed to stand for reaction for 10 min in dark conditions. Reading was noted down on UV VIS SHIMADZU 1800 Spectrophotometer at 517nm. |
| Determination of percentage inhibition of DPPH Activity by using following formula : |
| % Inhibition of DPPH Activity = A-B/A *100 |
| Where, |
| A = Optical Density (O.D.) of the blank |
| B = Optical Density (O.D.) of the sample |
| BIOLOGICAL ACTIVITY |
| ANTIPYRETIC ACTIVITY OF ANDROGRAPHIS PANICULATA |
| Animal Care and Handling |
| The experiment was carried out on albino rats of 4 months, of both sexes, weighing between 100 to 170 gm. They were provided from Truba Institute of Pharmacy, Bhopal, (M.P.). The animals were acclimatized to the standard laboratory conditions in cross ventilated animal house at temperature 25±2°C relative humidity 44 –56% and light and dark cycles of 12:12 hours, fed with standard pallet diet and water ad libitum during experiment. The experiment was approved by the institutional ethics committee and as per CPCSEA guidelines (Approval no. 1196/a/08/CPCSEA). |
| Chemicals |
| Paracetamol injection and Typhoid vaccine were purchased from Rajshree medical store, Bhopal. All other chemicals used for this study were of analytical grade. |
| Pyrexia Induced in Rats by Typhoid Vaccine |
| The room temperature was maintained at 30ºC. Only animals with a body temperature of at least 38ºC (Max) were taken into the test. Typhoid-Paratyphoid A,B vaccine (0.1 ml) was injected i.p. of rats of each group. Standard paracetamol (I.V.) was injected 30 minutes before administration of Typhoid-Paratyphoid A, B vaccine. The rectal temperature of each animal was recorded initial and at the interval of 60 minute after treatment using treatment using tele thermometer up to 2 hours(3) |
| EXPERIMENTAL DESIGN |
| In the experiment, a total of 12 mice were used. The mice were divided into 6 groups comprising of 2 animals in each group as follows: |
| Group : Control |
| Group I : Mice received Paracetamol (10mg/kg, p.o.) only 1 day around 1 hr before measurement of body temperature by the help of digital tele thermometer. |
| Group II: Mice received Methanol Extract of A. paniculata, (100mg/kg p.o.) once daily for 2 days. |
| Group IV: Mice received Chloroform Extract of A. paniculata, (100mg/kg p.o.) once daily for 2 days. |
| Group V: Mice received Petroleum ether Extract of A. paniculata, (100mg/kg p.o.) once daily for 2 days. |
| Group VI: Mice received Aqueous Extract of A. paniculata, (100mg/kg p.o.) once daily for 2 days. |
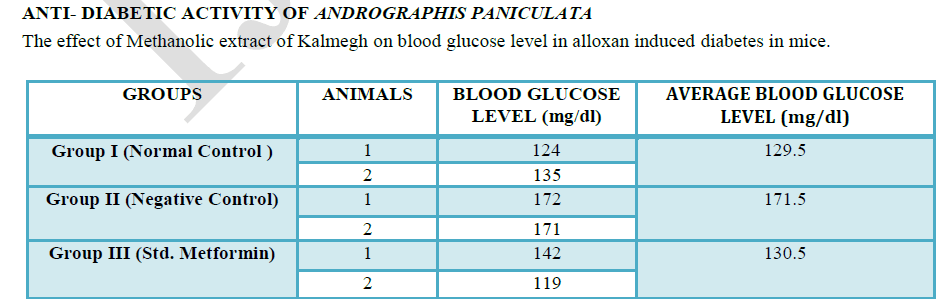
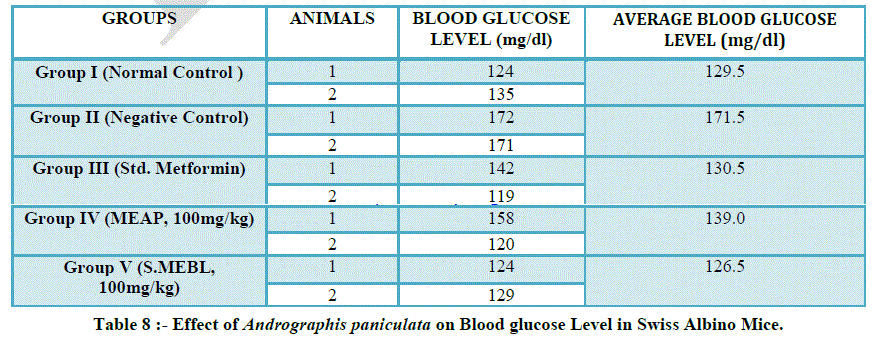
| ANTI-DIABETIC ACTIVITY OF ANDROGRAPHIS PANICULATA |
| Animal Care and Handling |
| The experiment was carried out on Swiss albino mice of 4 months, of both sexes, weighing between 25 to 30 gm. They were provided from Truba Institute of Pharmacy, Bhopal, (M.P.). The animals were acclimatized to the standard laboratory conditions in cross ventilated animal house at temperature 25±2°C relative humidity 44 –56% and light and dark cycles of 12:12 hours, fed with standard pallet diet and water ad libitum during experiment. The experiment was approved by the institutional ethics committee and as per CPCSEA guidelines (approval no. 1196/a/08/CPCSEA) |
| Induction of Diabetes by Alloxan: |
| The diabetes was induced by a single intraperitoneal injection of a freshly prepared solution of Alloxan monohydrate (120mg/kg b.w.). Hyperglycemia was confirmed on the third day of Alloxan injection. Mice with moderate diabetes having hyperglycemia (with blood glucose above 170 mg/dl) were taken for the experiment. |
| EXPERIMENTAL DESIGN |
| In this experiment, a total of 10 mice were used. The mice were divided into 5 groups comprising of 2 animals in each group as follows: |
| Group I: Normal control |
| Group II: Negative control mice received Alloxan 120g/kg, i.p. for inducing diabetes. |
| Group III: Mice received Metformin (30mg/kg,p.o.) for 5 days and Alloxan 120g/kg, i.p. on 1st day. |
| Group IV: Mice received Methanolic Extract of Kalmegh, (100mg/kg p.o.) once daily for 5 days and Alloxan 120g/kg,i.p. on 1st day. |
| Group V: Mice received Aqueous Extract of Kalmegh, (100mg/kg p.o.) once daily for 5 days and Alloxan 120g/kg, i.p. on 1st day. |
IV. RESULTS AND DISCUSSION |
 |
| ANTI-BACTERIAL ACTIVITY OF DRUG EXTRACT FROM SOXHLATE EXTRACTION METHOD |
 |
 |
| ANTI-BACTERIAL ACTIVITY OF SOME STANDARD ANTIBIOTICS |
 |
DISCUSSION AND CONCLUSION |
| The determination from the present study showed that the four extracts (choloroform, petroleum ether, methanol, and aqueous) of Kalmegh revealed anti-bacterial properties against the two bacteria S. aureus and E. coli. When the four extracts were compared with each other and with the standard antibiotic Penicillin G and Ofloxacin, the methanolic extract observed to have highest potential compared to that of chloroform, petroleum ether and aqueous extracts. The anti-bacterial activity of the methanolic extract of Kalmegh is attributed due to the combined effect of andrographolide and arabinogalactan proteins(11) . |
ANTI-OXIDANT ACTIVITY OF ANDROGRAPHIS PANICULATA |
| Phytochemical screening reveals that the major constituents of Kalmegh extract are phenolic compound, glycosides, alkaloid and flavanoid. Among these phenolic compounds which may be responsible for the activities of antioxidant. |
| DPPH Radical Scavenging Activity : |
| Kalmegh had significant scavenging effect on the DPPH free radical which increased with increasing concentration. The scavenging effect of sample was lower than that of Ascorbic acid. |
 |
DISCUSSION AND CONCLUSION |
| The results of this study clearly indicate that Kalmegh have high antioxidant activity and radical scavenging activity against various antioxidant systems in vitro. Kalmegh can be used as an easily accessible source of natural antioxidants and as a possible food supplement. In our present study we conclude that Kalmegh has good antioxidant property and could be attributed to the presence of flavonoids, alkaloids, tannins, saponin glycosides and phenolic compounds. |
| AEAP - Aqueous extract of Andrographis paniculata. |
| MEAP - Methanolic extract of Andrographis paniculata. |
| CEAP - Chloroform extract of Andrographis paniculata. |
| PeEAP - Petroleum Ether extract of Andrographis paniculata. |
 |
DISCUSSION AND CONCLUSION |
| Antipyretics are the agents which reduce the elevated body temperature. Regulation of body temperature requires a delicate balance between production and loss of heat, and the hypothalamus regulates the set point at which body temperature is maintained. In fever this set point elevates and a drug like paracetamol doesnot influence body temperature when it is elevated by the factors such as exercise or increase in ambient temperature(4). Experimental studies reveals that extracts of Andrographis paniculata (at dose 100mg/kg) produced an antipyretic action by decreasing the body temperature in the model of fever in rats. Both chloroform and aqueous extract of Andrographis paniculata (at dose 100mg/kg) exhibits higher anitipyretic activity as compared to petroleum ether and methanolic extract (100mg/kg). |
 |
 |
DISCUSSION AND CONCLUSION |
| Alloxan causes diabetes through its ability to destroy the insulin-producing beta cells of the pancreas (8,9) . In vitro studies have shown that alloxan is selectively toxic to pancreatic beta cells, leading to the induction of cell necrosis (6). The cytotoxic action of alloxan is mediated by reactive oxygen species, with a simultaneous massive increase in cytosolic calcium concentration, leading to rapid destruction of beta cells (12). |
| Experimental studies reveals that methanol extract of Andrographis paniculata (at dose 100mg/kg) produced an antidiabetic action by decreasing the blood glucose level in the model of alloxan-induced diabetes in mice. |
V. FUTURE PROSPECTS |
| Herbs have various salutary effects for promoting health and well being. Such herbs are called “tonics” for their ability to strengthen tone and regulate. India also has a venerable ancient tradition of herbal medicine and natural healing called Ayurveda, “science of life”. Andrographis paniculata is one such plant which is extensively used in herbal medicines due to its vast and time proven medicinal properties. The plant contains bitter glycosides, among them the major one is “andrographolide” found in higher quantum and proven to have various medicinal properties. Therefore, it is of great interest to carry out screening of this particular secondary metabolite in Andrographis paniculata in order to validate their use in folk as well as modern medicine and to reveal the active principle. |
| Many research organizations and Industries are pursuing research on exploring the flora like CIMAP, Himalayan Drugs etc. and many success stories are daily published. But the research should be carried out in a large scale and should be region specific so that new formulations can be prepared. Much work is also going on Polyherbal Formulation, in which many herbal drugs are scientifically mixed to get the synergistic effect. |
ACKNOWLEDGEMENT |
| I express my heartfelt thanks and sincere gratitude to my guide Mrs. Tasneem Ahmed (M.Tech-Biotechnology, IIT, Kharagpur), Co-ordinator of Blossom Pharma Biotech Institute and Research Centre, Bhopal (M.P.) for their immense support and guidance providing to me throughout the entire course of this project work. |
| I express my deepest gratitude to Dr. Sanjay Nagar, Head of The Department of Biotechnology, IPS Academy, Indore for his never ending guidance and direction through valuable suggestions along with enthusiastic encouragement through-out the period of my work and preparation of this project report. Last but not the least I would like to thank my parents for their constant support and encouragement in bringing this work to the present form. |
References |
|