ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Khushboo Srivastava1, Niharika Shringi2, Vijay Devra3, Ashu Rani4*
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
A simple method based on alkaline extraction followed by acid precipitation and acid dissolution has been developed to produce pure amorphous silica from perlite. The reaction parameters such as molar ratio of NaOH/SiO2, reaction time and reaction temperature are varied for obtaining maximum silica conversion. About 70.6 % pure precipitated silica from perlite has been achieved in closed system at 120 ºC within 60 min. XRF, BET surface area, XRD, FTIR and SEM techniques are used to characterize the physico-chemical attributes of materials. The present research includes a cost benefit process under optimized conditions for produce wealth out of waste
Keywords |
| Perlite, Silica conversion, Silica gel, Affecting factors. |
INTRODUCTION |
| Technologies related to production method and applications of pure precipitated silica have been studied and developed throughout the world in great variety of ways. Commercially available precipitated silica is manufactured by the fusion of high purity caustic soda and silica sand in furnaces at temperatures of 1300-1500 ºC and higher to produce solid glass [1]. Tetraethylorthosilicate (TEOS) is also widely used for production of commercial silica. But these processes are very expensive and energy intensive [2]. Recently, a large amount of precipitated silica has been extracted from various types of bio waste such as rice husk ash [3], rice hull ash [4] and bagasse ash [5] etc. Several techniques such as acid leaching or by gasification of rice hull with a pilot flame in a modified fluidized bed as well as by burning the rice hull ash at high temperature have been reported for the extraction of silica [6, 7]. The global market and demand for specially silicas (silica gel, precipitated, fumed and colloidal silica) reached and estimated value of $1.7 million. Through the year 2002, the global market for specially silicas is expected to grow at a rate of roughly 4.0 % a year exceeding $2 billion in that year [3]. Highly purified precipitated silica has been widely used in many applications including production of nanomaterials, in reinforcement of rubbers and plastics, in thickening and thixotrophy of coatings and paints, printed inks, plastics and cosmetics, as desiccants, stabilizer, adsorbent, food rheology modifier and as carrier of pesticides and catalysts [8]. The present study has been undertaken to evaluate systematically and quantitatively the influence of various physical and chemical factors on the extraction of pure precipitated amorphous silica from perlite, an abundant solid waste. Perlite containing high silica and alumina content can be an economically viable raw material for production of pure precipitated silica. It is an acerbic, siliceous, and volcanic glass lava containing crystal water, falling into the category of igneous rock [9]. China has the third largest in reserves of perlite in the world. The available world production of perlite in 2010 in respect of principal countries has estimated at 3.32 million tonnes [10]. The results permit a rational approach to the development and production of highly purified precipitated silica to satisfy individual requirements. This study employs a report on the manufacture, affecting factors and characterization of silica made from perlite using alkali solubilisation followed by subsequent precipitation and dissolution with different acids at lab scale. |
II. EXPERIMENTAL |
| Perlite was obtained from Indian Chemical Pvt. Ltd., Kotdwar. All chemicals were purchased from S. D. Fine Chem. Ltd., India. A. Preparation of highly purified precipitated amorphous silica from perlite Fig.1 represents the flow diagram for production of precipitated amorphous high purity silica from perlite. Perlite was thoroughly washed with distilled water to remove adhering soil and dust and then dried at 110 ºC for 12 h. After that, it was calcined at 800 ºC for 3 h. For producing sodium silicate solution, the reaction mixture of perlite (10 g) and NaOH solution was stirred in two types of systems: (i) open (ii) closed system. In the first one, the reaction was carried out in open borosil beaker under atmospheric pressure while in closed system, the reaction mixture was placed in an air tight Erlenmeyer flask under stirring at 600 rpm. The solution was filtered through Whatman No. 41 filter paper and the residue was washed with 20 ml boiled distilled water. The filtrate and washings were allowed to cool to room temperature and titrated with 5 N H2SO4 with constant stirring. The pH of the solution was monitored and titration stopped at pH 7. A soft white gel was formed and aged for 6 h. After aging, the slurry was washed by vacuum filtration using distilled water to remove sodium sulphate and then dried at 110 ºC for 12 h, weighed. For removing other mineral contents such as Al, Na etc. the dried material was refluxed with 1 N HCl at 120 ºC for 2 h and then washed repeatedly using deionised water to make it acid free. Now, the obtained pure precipitated amorphous silica was dried at 110 ºC for 12 h. The solid residue obtained during filtration was washed with distilled water and dried at 110 ºC for 12 h. B. Quantification of silica conversion (wt. %) The silica conversion with lesser amount of Al, Na content (wt. %) was determined by mass balance (Fig. 1) using following formula: - SiO2 conversion (wt. %) = [P – R / P] x 100 Where P = total wt. of perlite, R = wt. of solid residue consisting water insoluble unreacted silica. The reaction parameters such as reaction time (0 to 60 min), molar ratio of NaOH/SiO2 (0.6 to 2.4) and reaction temperature (60-120 ºC) were optimized during silica extraction. |
 |
| C. Regeneration Regeneration is the step where calcium hydroxide reacts with sodium sulphate to form calcium sulphate and sodium hydroxide. As per the reported procedure, NaOH is regenerated upto 90 %. The regenerated NaOH solution is used for digestion of the fresh perlite [11]. The obtained calcium sulphate can be used for other laboratory work. D. Characterization Techniques The chemical components of the samples were analyzed by X-ray fluorescence spectrometer (Philips PW1606). The BET surface area was measured by N2 adsorption-desorption isotherm study at liquid nitrogen temperature (77 K) using Quantachrome NOVA 1000e surface area analyzer. Powder X-ray diffraction studies were carried out by using (Philips X’pert) analytical diffractometer with monochromatic CuKα radiation (k = 1.54056 Å) in a 2θ range of 0-80º. The particle size of the samples was determined from the X-ray diffraction data using Debye-Sherrer formula. Cs = 0.9 λ / β cosθ Where Cs is the particle size, λ is the wavelength of the incident X-ray beam, β is the full width at half maximum (FWHM) of the X-ray diffraction peaks and θ is half of the angle 2θ corresponding to the peak [12]. The FTIR study of the samples was done using FTIR spectrometer (Alpha-T model, Bruker, Germany) by mixing the sample with KBr pellet mode (in 1:20 wt. ratio) in the range of 4000-400 cm-1. The detailed imaging information about the morphology and surface texture of the sample was provided by SEM (Philips XL30 ESEM TMP). |
III. RESULT AND DISCUSSION |
| A. The physico-chemical characteristics of perlite The chemical composition of raw perlite was as determined by XRF (in wt. %): SiO2 (74.9%), Al2O3 (12.6%), Fe2O3 (0.8%), CaO (0.6%), MgO (0.1%), TiO2 (0.1%), Na2O (4.6%), K2O (4.7%) and trace elements (4.0%). The L.O.I. (loss on ignition) was 2 wt. %. It can be evidenced that the perlite contains 73.9 wt. % silica with light grey color due to presence of carbon particles and small amount of other elements considered as impurities which turns into white-pink color with 74.3 wt. % silica content after calcination at 800 ºC for 3h. Thermal treatment at high temperature leads to the removal of water and the organic matter [13]. The surface area and average particle diameter of raw perlite were 5 m2/g and 3.20 μm. B. Effect of reaction parameters Fig. 2 presents the influence of molar ratio of NaOH/SiO2 variation from 0.6 to 2.4 on silica extraction which indicates that the concentration of NaOH solution positively affects the silica dissolution since silica extraction is increased on increasing NaOH concentration. The dissolution process of silica in aqueous solutions is mainly due to hydrolysis of Si-O-Si bonds, therefore silica configuration has strong influence on this process. As a comparison, quartz silica is the most stable form while amorphous silica is the most soluble [14, 15]. Extraction with more concentrated NaOH does not improve the silica yield. |
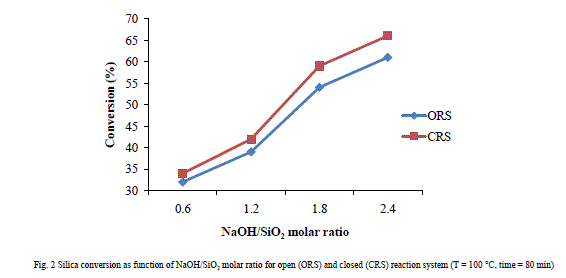 |
| Fig. 3 exhibits that silica conversion into sodium silicate in closed vessel results better than open system on increasing reaction time (0 to 80 min) at 100 °C temperature. The time of “0 minute” corresponds to the time necessary for reaction system to reach the desired temperature. |
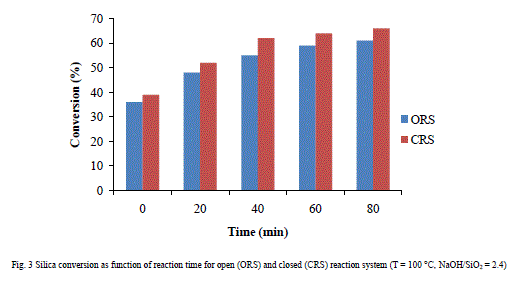 |
| The influence of reaction temperature on silica extraction is shown in Fig. 4. It is observed that the silica conversion increases with rising temperature, reaching 71.4 % at 120 °C in 60 min. Further increasing the reaction temperature, the conversion reaches at constant value in lesser time. |
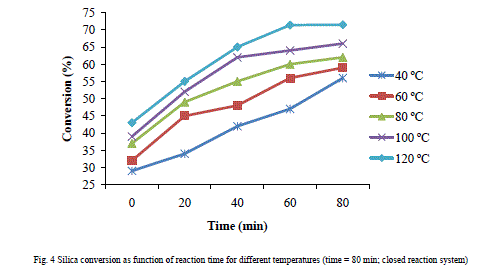 |
| C. Characterization of pure extracted silica from perlite The XRF analysis shows that the obtained precipitated silica from perlite is found to be white fine solid with 70.6 % pure silica content having ≈ 98 m2/g specific surface area in the range of 0.3-1 μm particle size. As shown in Fig.5, the X-ray diffractogram of extracted silica confirm amorphous characteristic along with broad humps centred between 10-35º (2θ). Amorphous substances display an atomic arrangement that is either random or has very short-range order [4, 7]. |
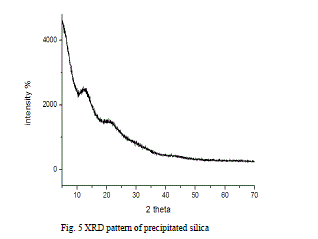 |
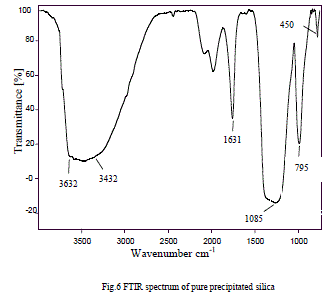
| The major chemical groups present in extracted silica are identified by the FT-IR spectra shown in Fig. 6. The broad band between 3645-3400 cm-1 indicates the presence of isolated and surface Si-OH groups. The predominant adsorbance peak at 1085 cm-1 is due to asymmetric stretching vibration of siloxane bonds (Si-O-Si) and the corresponding symmetric stretch at 795 cm-1. The peaks at 1085, 795 and 450 cm-1 are attributed to the vibration modes amorphous silica gel network [16, 17]. |
 |
| In Fig.7 (a & b), the SEM image of raw perlite indicates irregular shaped crumbled particles whereas the SEM micrograph of extracted pure silica demonstrates globular shaped highly porous particles of different shapes and sizes in agglomerated form. Such type of large gelatinous mass is due to the presence of amorphous silica gel skeleton. |
 |
IV. CONCLUSION |
| This study reveals a simple low energy chemical process for the generation of high purity amorphous silica from perlite without using any organic additive. The yield of extracted siica from perlite was 70.6 % with ≈98 m2/g surface area in closed reaction system using NaOH/SiO2 = 2.4 molar ratio at 120 ºC within 60 min. The used chemicals can be regenerated making it an environmentally benign, cost effective and atom efficient process and thus finds a novel route to utilize abundant waste perlite in future. |
ACKNOWLEDGEMENT |
| The authors are thankful to Dr. D.D. Phase and Er. V.K. Ahire for SEM analysis and Mr. Mukul Gupta for XRD conducted at UGC DAE-CSR Lab Indore. XRF analysis was conducted at Punjab University, Chandigarh. The financial support was provided by Department of Science and Technology, Rajasthan, India, project sanction no. P- 7/DST/R&D/11435-38. |
References |
|