ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
M.P.D.Prasad1, V.Sridevi2*, N.V.Surendra babu3, O.V.S Reddy4, N.Harsha5
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Citric acid is a tri carboxylic organic acid is the most important acid produced by fermentation used in the food and pharmaceutical industries. The present work deals with the determination of optimum growth conditions for citric acid production by submerged fermentation by using strain of A.niger MTCC662 respectively. Isolation, screening and mutated the A.niger strain induced by UV irradiation, diethylsulphate and Co60 by providing different types of media and the submerged fermentation was carried out under various growth parameters like temperature, pH and media of Sorghum malt as substrate. Strain improvement studies for maximum production of citric acid obtained after the fermentation. Maximum yield was obtained from mutant type of A. niger MTCC 662 than wild strains were about 12.0g/100ml and 8.2g/100ml. It could be concluded that the A.niger MTCC662 mutant strains have produced high yield than wild strain of A.niger MTCC662 cultures.
Keywords |
| A.niger MTCC 662, submerged fermentation, Temperature, pH, Strain improvement. |
INTRODUCTION |
| Citric acid is a tricarboxylic organic acid, soluble in water with pleasant taste, and is the most important in food and other industries. Citric acid derives its name from the Latin citrus, the citron tree, the fruit of which resembles a lemon. Refined sugars such as glucose and sucrose are the most commonly used substrates for commercial production of citric acid by fermentation process; however they are expensive and can be replaced by various cheap and abundant substrates like agroindustrial wastes or by-products including starch, cellulose and lignocellulose material[1]. Surface method and submerged process are widely recognized for the production of citric acid. The surface process though commercially profitable for many years, is labored intensive and inefficient in its use of space. The submerged process has become the method of choice in the industrialized countries[2], because it is less labour intensive, yields higher production rate, and uses less space. Citric acid is used in food, Confectionery and beverages, in pharmaceuticals and industrial fields. Citric acid forms a wide range of metallic salts including complexes with copper, iron, magnesium, manganese and calcium. These salts are used as sequestering agents in industrial processes and as anticoagulants in blood preservation. Citric acid is used as plasticizer in plastic films and as an antioxidant in oil and fats. Sorghum is produced in agro-based countries like India. Surplus production of the above substrates and their availability prompted us to utilize them in the present study. The damaged grains and unused flours can be diverted for the production of a value-added product like citric acid which has tremendous application. The present work is focused on these substrates which are rich in starch. Instead of hazardous acid hydrolysis and expensive enzymatic treatment, the flours are hydrolyzed by natural malting process which is economically feasible and enhances the nutrient quality of the malts for fermentation process. Since the citric acid concentration produced by wild strains is too low for economical processes, strain improvement was carried out in order to develop mutants of parent strain for increased production of the products[3]. However, strain development from wild strains to mutants depends mainly on the process of mutagenesis (physical and chemical mutagenesis). Although, traditional random strain mutagenesis is time-consuming and lacks specificity and precision due to the absence of adequate molecular information i.e., there is no opportunity to expand the gene pool, it is still a very practical approach[4], as it is quite simple and rapid method when compared to targeted mutagenesis. Developments of mutant strains which can synthesize higher concentration of citric acid within a short fermentation time and capable of growing at lower pH are preferred. Finally the yield of citric acid was further enhanced by optimizing the fermentation parameters like temperature, pH, incubation time, substrate concentration and nitrogen source and several other organisms were found to accumulate citric acid including strains of A. niger, A. awamori, A. fonsecaeus, A. luchensis, A. phoenicus, A. wentii, Acremonium sp., Aschochyla sp., Botrytis sp., Eupenicinium sp., Mucor piriformis, Penicuium etc., Among many microorganisms that have been exploited for citric acid production, A. niger still remains as the promising species. |
II. MATERIALS AND METHODS |
| Isolation: Samples were collected from sugar cane waste molasses from local industry Empee Sugar and Distilleries, Naidupeta, Nellore district (A.P). |
| Screening of fungal cultures: Dye Method: The A. niger was screened qualitatively in Petri plates containing Czapek-Dox agar medium with Bromocresol green as an indicator. |
| Preparation of medium: Sorghum (Sorghum bicolor) is the cereal crops produced throughout the world, and are very important to the Asian countries. Hence the raw material is available perhaps at a cheaper cost. The rate conversion of carbohydrates into fermentable sugars is high in sorghum. The conversion rate of starch into simpler sugars in these substrates is around 80-88%. |
| Malting and mashing of substrate: Instead of expensive enzymatic hydrolysis and hazardous acid hydrolysis the natural and safe malting and mashing process is adopted to convert the complex substrates in to readily utilizable simple substrates. |
| Estimation of reducing sugar: The reducing sugars in the malted and mashed grains of sorghum estimated by DNS (Dinitro salicylic acid method). The reducing sugars further qualitatively estimated by paper chromatography with standard reducing sugars. |
| Estimation of citric acid: Citric acid from the fermentation broth is quantitatively estimated by acetic anhydride, pyridine methods [5] using spectrophotometer at 405 nm. |
| Strain improvement studies: Mutagenesis is mainly through physical mutagen by UV irradiation, chemical mutagen by diethyl sulfonate (DES) and a mixture of both (UV and DES) mutagens in combination with certain time intervals of exposure. |
III. RESULTS AND DISCUSSION |
| A successful process depends upon appropriate strain, culture medium and optimization of fermentation parameters. In the present work, cultural conditions such as sugar concentration, time profile of citric acid synthesis, incubation time, temperature, pH, nitrogen source were optimized with the parent isolate and mutant strain of sorghum malt substrate. |
Isolation of strain: |
| Four different A. niger strains were isolated from the molasses waste and by single colony isolation technique. Futher the single potent isolate for citric acid production were screened by dye method followed by submerged fermentation by shake flask method [6]. |
Effect of sugar concentration |
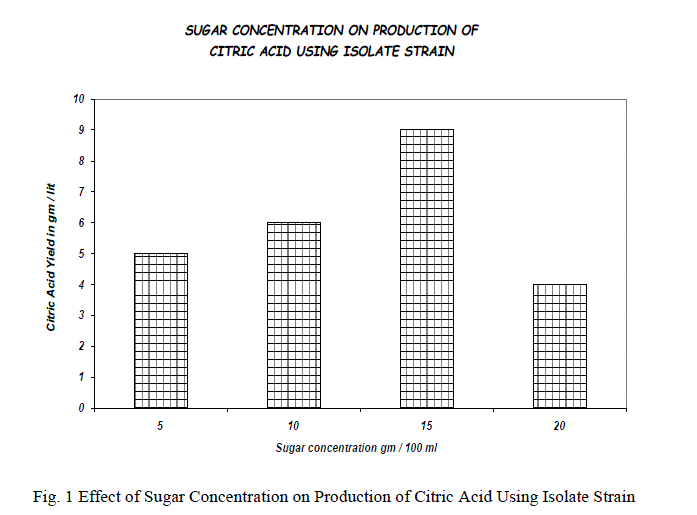
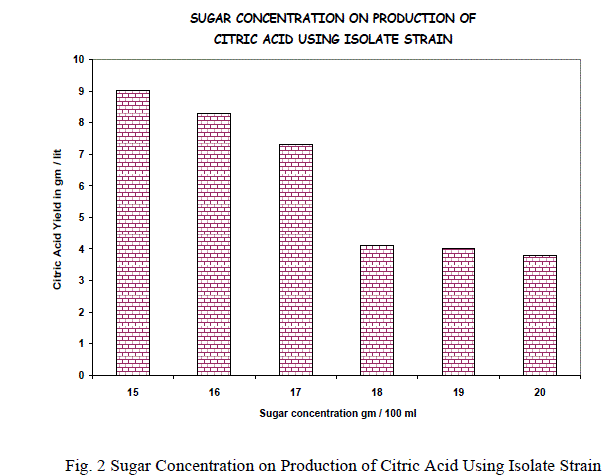
| The initial sugar concentration has been found to determine the amount of citric acid produced by filamentous fungi A.niger. The experiment was performed at 5%, 10%, 15%, 20% of reducing sugars and maximum yield was obtained at 15% above which there is decrease in the yield in that 15-20% of reducing sugar concentrations and found that there is a decrease in the citric acid production after 16% of reducing sugar. Reduction in the citric acid formation was observed when the sugar concentration of the medium is increased; it may be due to the order growth of mycelium, which results in increase viscosity of the medium. The results are shown in the fig.1&2. |
 |
 |
Optimization of malting and mashing conditions: |
| Malting is the limited germination of the sorghum grain under very carefully controlled conditions. There are three phases in malting namely, steeping, germination and kilning. Steeping encourages germination to start, germination prepares the conversion of the starch to sugars, and kilning stops the germination to ensure that very little of the starch is hydrolysed. Conversion of starch to maltose occurs in the mashing process. The steeping conditions (time and temperature) for sorghum were optimized and maximum yield of reducing sugars were obtained at 18 h of time and 31oC temperature for sorghum. The reducing sugar concentration was more with NaOH steeping rather than with distilled water or KOH. The germination process was carried out for 3 days for optimum yield of reducing sugars. In order to convert more amount of starch, mashing process was carried out with different ranges of temperature (550C-650C) and the yield obtained was maximum at 590C for sorghum. Further there was two fold increases in the reducing sugars when the mashing process was carried out with commercial enzyme like Biogluconase. |
Optimization of media conditions for maximum production of citric acid Incubation temperature: |
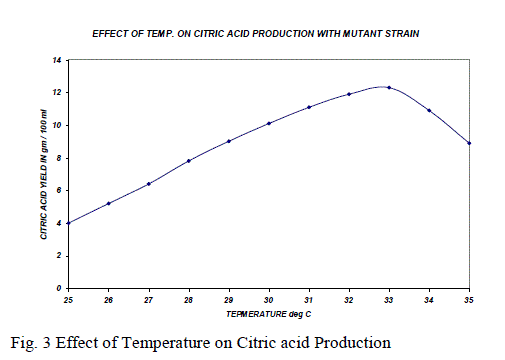
| The production of citric acid at different temperatures (250C-350C) was studied. Maximum production of citric acid was obtained when temperature of the medium was at 330C and it is shown in the Fig. 3. The temperature of the fermentation medium is one of the critical factors that have direct effect on the production of citric acid. When the temperature of the medium was low, the enzyme activity was also low, giving no significant impact on the enhancement of citric acid production. But when the temperature of the medium was increased beyond 330C, the biosynthesis of citric acid was decreased. It may be due to that the high temperature can cause denaturation of the enzyme citrate synthase and accumulation of other by products such as oxalic acid. A temperature of 40oC was most favorable for oxalic acid production while citric acid accumulation was completely inhibited at this temperature [7]. |
 |
Effect of pH: |
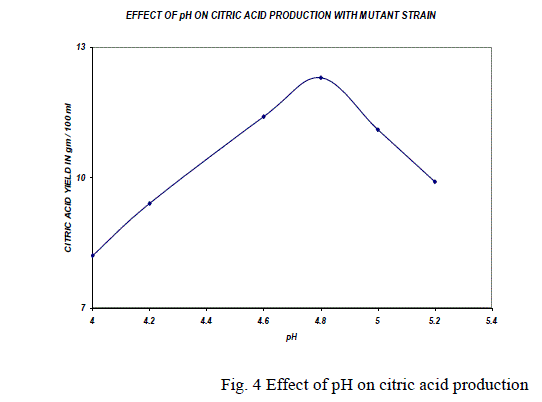
| The production of citric acid was studied at different pH (4.8 to 6.0) levels. Maximum yield was obtained (9.3g/100 ml) when the pH was maintained at 5.4 with the A. niger and maximum yield 12g/100ml with the mutant strain at pH 4.8 and it is shown in the Fig. 4. The maintenance of a favorable pH is very essential for the successful fermentation of citric acid. Decrease in pH caused reduction in citric acid production [8]. At low initial pH the growth of the mycelium in the medium is very poor. |
 |
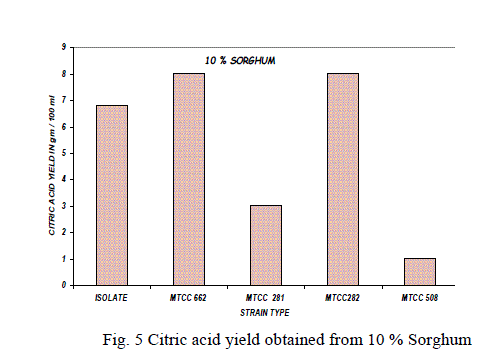
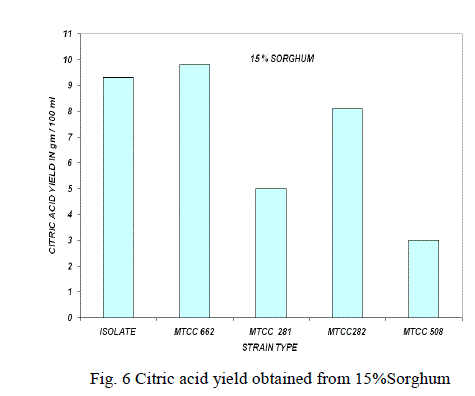
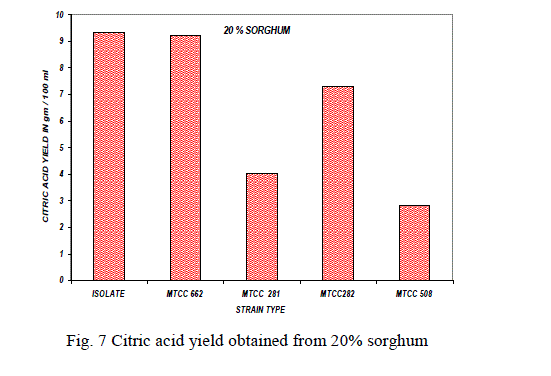
Substrate concentration: |
| The effect of substrate concentration was also studied with 10%, 15%, 20% of sorghum with parent isolate and mutant strains and the results were shown in the Fig. 5,6,7. The same experiment was also carried out with MTCC culture for comparison of efficiency of the parent isolate for citric acid production [9].The high yield of citric was reported at 15% of sorghum with A. niger isolate and mutant strain. At higher substrate concentration (above 20%), the medium became the viscous and the yield of citric acid decreased due to limited aeration in the culture broth. |
 |
 |
 |
 |
 |
Reducing sugar concentration |
| The effect of different sugar concentrations (12-20g/100 ml) [10] on citric acid production by A. niger isolate was carried out, and the maximum amount of citric acid (9.3g/100 ml) was obtained in the medium containing 15g/100ml(substrate concentration). The initial sugar concentration has been found to determine the amount of citric acid produced by filamentous fungi A. niger. Reduction in the citric acid formation was observed when the sugar concentration of the medium was increased. It may be due to the overgrowth of mycelium, which resulted in increased viscosity of the medium [11]. The increase of mycelia formation in the medium, there was reduction in the yield of citric acid. The sugar concentration higher than 15-20% led to greater amount of residual sugars [12], while a lower sugar concentration led to lower yields of citric acid owing to the accumulation of oxalic acid in the culture broth. |
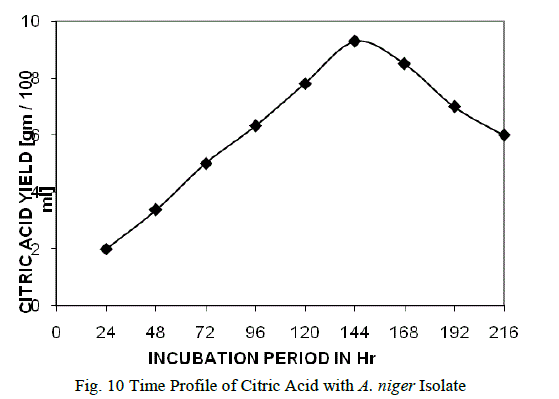
Time profile of citric acid: |
| The fermentation was carried out with different malts of sorghum from 24 h to 216 h. Maximum yield of citric acid was obtained after 144 h (7 days) and the results are shown in the Fig. 10. The optimum time of incubation for maximal citric acid production varied both with the organism and fermentation conditions. In the submerged fermentation of citric acid, the production started after a lag phase of one day and reached maximum at the onset of stationary phase or late exponential phase. Further increase in incubation period did not enhance citric acid production. It may be due to the age of the mycelium and depletion of sugar contents in the cultural broth [13]. Our findings are more significant as compared to the pervious workers because reduction in incubation period would reduce the cost of citric acid production. |
 |
IV. CONCLUSION |
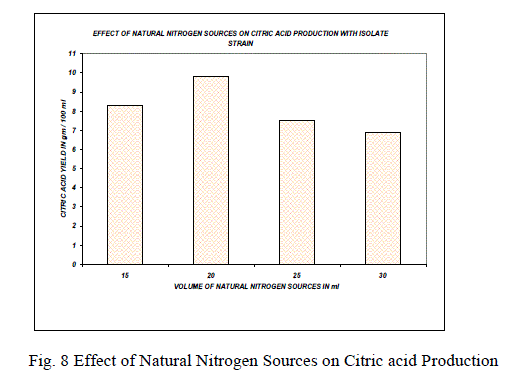
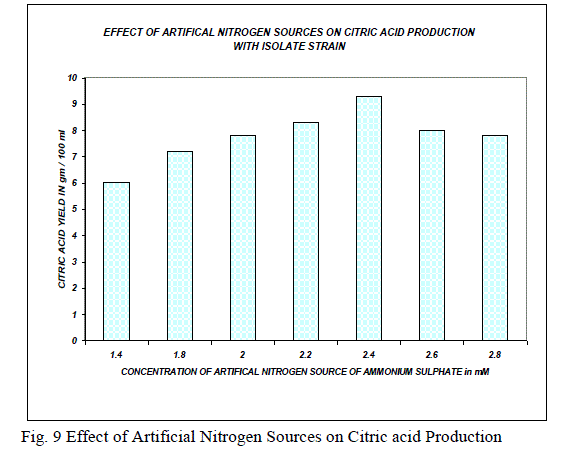
| The optimization of environmental conditions was experimented by taking temperature, pH, nitrogen source, substrate concentration and incubation time. The suitable conditions for the production of citric acid by using A.niger mutant strain MTCC 662 were temperature 330C; pH 4.8 nitrogen source 2.4 mM, substrate concentration 15 g/ml, incubation period 144 hrs high yields was obtained. |
References |
|