ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Razi Ahmad1,MeryamSardar2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Microbial resistance represents a challenge for the scientific community to develop new bioactive compounds. In the present study antibacterial effect of TiO2 nanoparticles (<25nm) on Escherichia coli strain was investigated. The MIC50 value for the TiO2nanoprticles was found to be 200 μg/ml for E. coli, which was confirmed by the Colony-forming unit (CFU) of the culture with and without TiO2 nanoparticles inoculated on agar plate. The Inhibition was further confirmed using Disc diffusion assay. It is evident from the zone of inhibition that TiO2 nanoparticles possess potent bactericidal activity. Further, growth curve study of E.coli in the presence and absence of TiO2 nanoparticles has been done, which shows effect of sub-inhibitory and inhibitory concentration of TiO2 nanoparticles against E. coli till 20 hours of incubation. Nanomaterials are known to inactivate cellular enzyme and DNA by binding to electron donating groups such as carboxylates, Amides, Indoles, Hydroxyles, Thiols etc. They cause little pores in bacterial cell walls leading to increased permeability and cell death.
Keywords |
| TiO2 nanoparticle, E. coli, Disc diffusion assay, Minimum inhibitory concentration, Antibacterial effect |
INTRODUCTION |
| Nanobiotechnology, an emerging field of nanoscience, utilizes nanobased-systems for various biomedical applications. Nanoparticles have a high specific surface area and a high fraction of surface atoms; have been studied extensively because of their unique physicochemical characteristics including catalytic activity, optical properties, electronic properties, antibacterial properties and magnetic properties (Zhao and Stevens, 1998; Crabtree et al., 2003; Krolikowska et al., 2003; Catauro et al., 2004).As an important component in the development of nanotechnology, nanoparticles have been extensively explored for possible medical applications. Nanoparticles provide a particularly useful platform, demonstrating unique properties with potentially wide-ranging therapeutic applications (Gao et al. 2004). Antibiotic resistant bacterial strains with different mechanisms are found continually and thus new drugs are required (Streit et al. 2004). Therefore, the finding of new antimicrobial agents with novel mechanisms of action is essential and extensively pursued in antibacterial drug discovery (Coates et al. 2002). Microbial resistance to antibiotics is a world-wide problem in humans and animals. It is generally accepted that the main risk factor for the increase in the antibiotic resistance is an extensive use of antibiotics. This has lead to the emergence and dissemination of resistant bacteria and resistance genes in animals and humans (van den Bogaard and Stobberingh 2000). The antibiotic resistance of pathogens can be a result of several different factors. Resistance among bacteria is continuously increasing. Nanotechnology is expected to open new avenues to fight and prevent disease using atomic scale tailoring of materials. Recently it has been demonstrated that metal oxide nanoparticles exhibit excellent biocidal and biostatic action against Gram-positive and Gram-negative bacteria (Lopez Goerne et al. 2012). TiO2 has three crystalline phases: anatase, rutile and brookite. Thermodynamically, rutile is the most stable form. Anatase has attracted much attention owing to its application in photovoltaic cells (Fujishima and Donald 2000) and photocatalysts (Gratzel, 2001) and for its antimicrobial properties. TiO2 nanoparticles have become a new generation of advanced materials due to their novel and interesting optical, dielectric, and photo-catalytic properties from size quantization (Alivisatos, 1996). Now a day’s Titanium dioxide (TiO2) is widely utilized as a self-cleaning and self-disinfecting surface coating materials, titanium dioxide has a more helpful role in environmental purification due to its photo induced super-hydrophobicity and antifogging effect (Fujishima and Honda, 1972). These properties have been applied in removing bacteria and harmful organic materials from water and air, as well as in self-cleaning or self-sterilizing surfaces in medical centers (Tojo et al. 2008). Some antimicrobial agents are extremely irritant and toxic and current researches are focused on formulate new types of safe and cost-effective biocide materials (Kato andKudo, 2002). On the other hand, nano structured reservoirs made of inorganic oxides like TiO2, and synthesized by the sol-gel process, have been demonstrated to be biocompatible and suitable supports for a wide variety of compounds (Peterson et al. 2007). Therefore, in this study the effect of commercially available Anatase TiO2 nanoparticles was investigated against E.coli. |
II. MATERIALS AND METHOD |
| Luria Agar and Luria broth were purchased from HiMedia. TiO2 nanoparticles with a size approximately <25nm were procured from sigma. All other reagents used were of analytical grade. A. Antimicrobial Effect of TiO2 in Liquid media Stock suspension of TiO2 nanoparticles with concentrations of 1 mg/mL was prepared by suspending them in distilled water and the solution was sonicated for 5 minutes to get the homogenous suspension. A stock suspension was kept in UV for 30 min for sterilization. Overnight Culture of the E. coli was added to Luria broth with different concentration of TiO2 nanoparticles respectively and incubated at 37°C for 16 hours.To study the bacterial concentration, The O.D. values were taken at 600 nm. B. Colony-Forming Unit 100 μl of the serial dilution 107 of the culture with and without TiO2nanoparticles were inoculated on agar plate and wasspreaded over it. The plates were incubated for 18 – 24 hours and observed for growth. Viable count per 100 μl for a dilution was obtained. C. The Agar Diffusion Test or Bauer-Kirby Test The antimicrobial activity of TiO2 nanoparticles was evaluated against E. coli. 0.2 ml of fresh cultures of organism was inoculated into 5 ml of sterile Luria broth and incubated for 3–5 h to standardize the culture to McFarland standards (106 CFU/ml). 100 μl of revived culture added on agar media and poured on three replicate plates. Wells were made having a diameter of about 8 mm, 50 μl samples of TiO2 nanoparticles were added in one well and 50 μl antibiotic (ampicillin) was also added in a separate well as standard. The petriplates were incubated at 37ºC for 18 hours. D. Antimicrobial effect of the TiO2 in solid Media The Bacterial culture (100 μl) was added in agar Media and poured on three replicates petriplates. After the media was solidified the six wells were made in each plate and different amounts ( 20 μg, 40 μg, 60 μg 80 μg and 100 μg) of TiO2 nanoparticles were added in these wells and one of the well was without TiO2naparticles in each plate. After 18 hours of incubation, each plate was examined and measured for the diameters of the zones of complete inhibition including the diameter of the wells. E. Growth curve study of E. coli Freshly grown bacterial inoculum (100 μl) was incubated in the presence of different concentrations of TiO2 nanoparticles in 100ml media and the flasks were incubated at 37°C temperature and 150 rpm. The growth rate is indexed by measuring optical density (OD) at 600nm after every two hours. The readings obtained were plotted and comparative studies were performed between control withand without TiO2nanoparticles. |
III. RESULTS AND DISCUSSION |
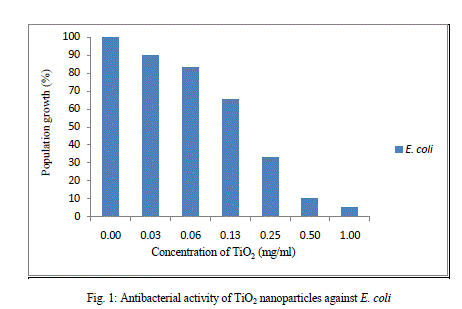
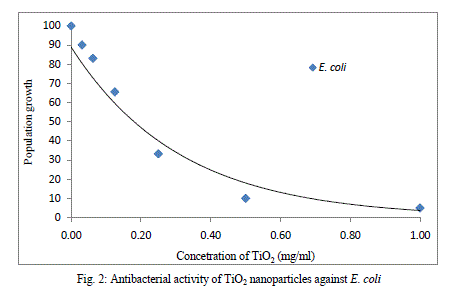
| Nanomaterials exhibit strong inhibiting effects towards a broadened spectrum of bacterial strains. According to several studies, it’s believed that the metal oxides carry the positive charge while the microorganism carry negative charge, this causes electromagnetic attraction between microorganism and the metal oxides which leads to oxidization and finally death of microorganism (zhang and chen 2009). Figure 1 show the population growth of bacteria with increasing concentration of TiO2nanoprticles.s |
 |
 |
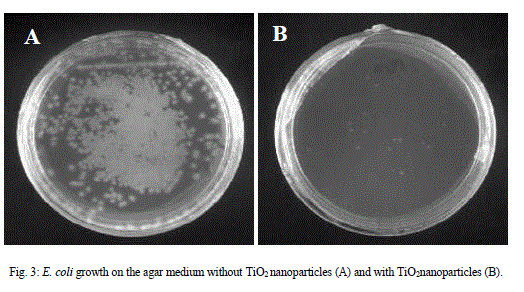
| MIC50 was defined as the minimum amount of the drug/compound to inhibit 50% of E. coli growth which was confirmed by measuring turbidity. MIC50 of TiO2 nanoparticle against E. coli was determined and found to be 200μg/ml. Also it was confirmed by the CFU of the culture with and without TiO2nanoparticles was inoculated on agar plate. Colony-forming unit (CFU) is an estimate of viable bacterial numbers. The purpose of plate counting is to estimate the number of cells present based on their ability to give rise to colonies under specific conditions of nutrient medium, temperature and time. Figure3 shows that with the TiO2 nanoparticles the number of viable colony decreases as compared to the control. |
 |
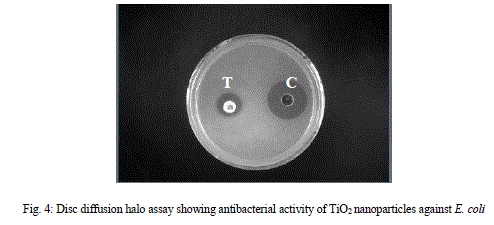
| For testing the TiO2 nanoparticles susceptibilities of E. coli, we used disc diffusion assay. As it is evidence from the results,cells were highly sensitive to all tested concentration of TiO2 nanoparticles, which was also confirmed from the size of the zone of inhibition. TiO2 nanoparticles have been found to be a potent bactericidal compound, although not as much as the established antibacterial compound such as ampicillin but can be studied for superficial application as well as for combinational therapies. |
 |
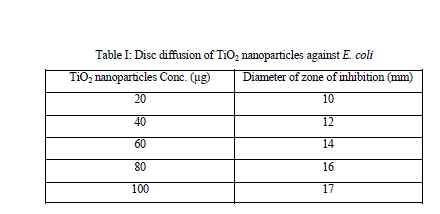 |
| For testing the susceptibilities of E. coli, we used Disc diffusion assay, and it was seen that as concentration of TiO2 nanoparticle has been increased, simultaneous increase of diameter of zone of inhibition was observed (Table1). It is evident from the zone of inhibition that TiO2 nanoparticles possess potent bactericidal activity. |
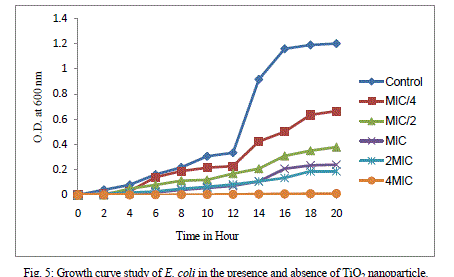 |
| Growth curve study of E. coli in the presence and absence of TiO2 nanoparticles has been done, which shows effect of sub-inhibitory and inhibitory concentration of TiO2 nanoparticle against E. coli till 20 hours of incubation. In control cell lag phase ends up to 4 hour after which log phase starts which ends up to 16 hours. However when cells were treated with TiO2 nanoparticles, there occurred delay in the lag phase. At MIC concentration lag phase ends up to 12 hour. There occurred delay in lag phase on exposure to different concentration of TiO2 nanoparticles and thus a prominent decrease in growth of E. coli was observed. |
IV. CONCLUSION |
| TiO2 nanoparticles shows inhibitory effect on the growth of E. coli strain which was confirmed by above parameters. Hence TiO2 nanoparticles can be considered as potent antibacterial compound, but not as much as the established compound such as ampicillin. As they are eco-friendly, so the antibacterial properties of TiO2 nanoparticles can be further explored in future on other bacterial strains, so that these nanoparticles can be used in various industrial and medical applications. |
ACKNOWLEDGMENT |
| The financial support provided by ICMR, Government of India to Razi Ahmad in the form of SRF is greatlyacknowledged. |
References |
|