ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
D. I. Sharif1, S. M. Monsur Musa2, Rajiv Kumar Sah2, Sabiha Rahman2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Electricity can be generated from naturally occurring electro potential differences through the burial of an anode in sediment and the immersion of a cathode in the overlying body of water. Such a set-up is called a Sediment microbial fuel cell (SMFC) or benthic MFC. Mangrove forest sediment of ―Sundarbans‖ was used to construct SMFC and an open circuit voltage was measured over 40 days. Voltage was found to increase over 14 days after which the voltage remained steady for about 7 days and then declined. The highest voltage of 610 mV was obtained from a single SMFC on the 24th day. Comparative analysis using earthen pot as a proton exchange membrane showed the voltage to increase gradually over an initial period of time, with a highest increase in 80 mV over SMFC without earthen pot. An analysis of different cathode material showed that the highest voltage was obtained from carbon rod over carbon brush and stainless steel as cathode material. Several SMFC were added to make a series connection, which gave enough voltage (3.8 volts) to turn-on a LED bulb and a scientific calculator. The study suggests that Sundarban soil sediment can be a potential source for further study in the development of SMFC
Keywords |
| Sediment Microbial Fuel Cell, Cathode, Earthen pot, Electricity generation, Voltage. |
I. INTRODUCTION |
| Microbial Fuel Cell (MFC) is a promising technology where microbes are used in the oxidation of organic substances for the production of electricity. The organic substrates utilized at the anode by microorganisms act as electron donors and generally range from simple carbohydrates, such as glucose [10], [4] and acetate [6] to more complex compounds, such as starch [9], wastewater [2] and sediment organic and inorganic constituents [3]. In a basic setup of MFC there is an anode chamber and a cathode chamber which is separated by proton exchange membrane (PEM). Microorganisms grow as biofilms at the anode and oxidize substrates producing electrons and protons. Protons migrate through an aqueous solution through the PEM to the cathode. Electrons that are generally donated through various mechanisms to the anode can pass to the cathode through an external wire. At the cathode compartment the electrons and protons are consumed and combined with oxygen to form water, thus a continuous flow of electrons generate electricity. Sediment microbial fuel cell (SMFC) is a relatively new technology that generally differs from the basic microbial fuel cell (MFC) in which oxidisable carbon compounds and other components in sediments and similar environments are utilized to produce power. In a SMFC, anode is placed in the sediment and the cathode is immersed in the overlying body of water. Microorganisms present in the sediment decompose the organic matter through metabolism thereby producing electrons and protons. The produced electrons are then extracted by the anode and passed to cathode through an external wire. The cathode plays a role of an electron sink to accept electrons and protons principally with the reduction of oxygen at the overlying body of water creating flow of electricity [1]. |
| Various sediments and soils of marine [5], rice plants [16], and mangrove sediment [14] have been used in electricity production from SMFC. Mangrove forest sediments have been shown to be a promising natural inoculum for MFC [14]. Mangrove forest sediments are known to harbor a variety of bacteria which are supported by the organic sedimentary content of the mangrove forest, local algal mats and mangrove tree wastes. The environmental conditions such as high soil salinity, frequent tides, and the anoxic conditions in the soil can also provide biodiversity which can define a microbial population capable of living in the anodic compartment of a MFC. Small scale SMFC systems evaluated primarily in the laboratory indicate power densities for typical graphite plate anodes on the order of 10–50 mW m−2 [11]. Besides selecting the sedimentary or soil for use as an inoculum in SMFC an increase in power output and cost reduction have been made possible through modifications of the SMFC configuration, such as using cheap materials as bioanodes and cathodes, use of earthen pot as proton exchange membranes, continuous supply of organic matter etc [15]. Increasing the performance of SMFCs is important to generating practical levels of power for undersea devices. Sustained operation of many sensors and communications systems require power in the range of 0.6 mW to 20 W. The use of mangrove forest sediment from âÃâ¬Ãâ¢The SunderbansâÃâ¬Ãâ as inoculum in SMFC has been still unexplored. The purpose of this work is to investigate voltage generation form the tropical mangrove forest sediment using various modifications to design a relatively cheap small scale SMFC to power up small devices at homes in rural areas having electricity scarcity. The present study will analyze voltage produced from tropical mangrove forest sediments using earthen pot as cheap proton exchange membrane and by using various cheap cathode materials in the construction of SMFC. |
II. MATERIALS AND METHODS |
| A. Construction of SMFC |
| Soil sample from âÃâ¬Ãâ¢SunderbansâÃâ¬Ãâ was collected from two different locations and used in the SMFC. Two different trials termed tria1 and trial 2 represented two different locations collected at different times of the year. The sample was collected from 8 inch deep from the surface of the ground-layer. In addition, the sample was not further modified before using in MFC chamber so that they can act as natural. Carbon brush of pure quality were used as electrodes e.g., anode and cathode. Each anode and cathode was connected with one end of a copper wire of 15-inch length by the use of epoxy resin. In the experiment SMFC was constructed by placing the anode in the sediment contained in a beaker and the cathode immersed in the overlying body of water. In each beaker, about 400 ml of sediments were placed and the anode was immersed into the sample about 4 inches from the surface. The cathode was placed in the aerobic upper layer containing water which was obtained after the sedimentation of the soil content in the beaker. The upper moist layer was constantly maintained at 1.5 inches above the sediment layer by the addition of extra water sample when necessary. Control experiments were performed using sterile sediments as the procedure mentioned above. |
| In SMFC with earthen pot, the earthen pot was filled with sediment and the anode was inserted in to the sediment as described above. The earthen pot was then placed inside the beaker. The distance of the area between the beaker and the earthen pot was 1.5 cm. The area between the beaker and the pot was filled with water sample from the sediments which was maintained at a constant level above the sediment sample (Fig.1). |
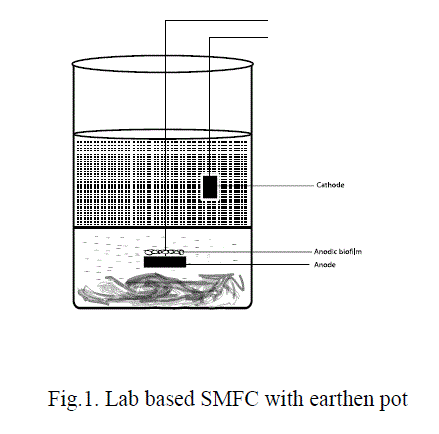 |
| B. Measurement of Electricity |
| The open circuit voltage was measured by a multimeter and reading was taken every 2 days over a period of 40 days. From the different voltages (mV), various currents (mA) regarding to these voltages was measured by a multimeter. The surface area of the Carbon brush used as anode was calculated with the help of slide-calipers. The area of the anode was 37.48×10-4m2. The current density (mA/m2) was detected by dividing current (mA) by surface area of the anode. Moreover, measurement of the power (P) was obtained by multiplying the value of current with voltage and extrapolated as the power density (PD) in mW/m2. All the constructed SMFC was connected in a series connection to test the practical implication of the SMFC to turn-on a LED bulb and a scientific calculator. |
III. RESULTS AND DISCUSSION |
| A. Voltage Produced from Sundarban Soil |
| The whole experiment was performed for about 40 days using carbon brush electrode as anode. Two different trials were used to represent different locations of soil sample. Fig. 2 shows variation in voltages (mV) from trial 1 and trial 2 over time. The voltage was found to increase sharply initially over 2 weeks and then reached a steady period of 4-6 days after which it gradually decreased. Maximum voltages were reached within 22-25 days and were 610 mV and 485 mV for trial 1 and trial 2 respectively. Such values were found to be consistent with other findings from other authors [8]. The starting voltage in both trials was different. Voltages obtained from trial 1 were low and remained low over the entire period compared to trial 2. This could well represent the physiological conditions and microbiological content of two different soil types. However the overall trend in voltage increase was similar. Control experiments performed using sterile Sundarban soil did not show any increase in voltage (result not shown) suggesting that the voltage increase was due to natural microbial consortia. Moreover, when sterile soil was enriched with scrapings of anodic bacteria the voltage was found to increase but did not increase as much as natural soil also suggesting that and natural physiological condition of the soil can affect voltage generation besides the natural microbial consortia of soil. |
| B. Voltage Produced Using Earthen Pot as Proton Exchange Membrane |
| Several studies have shown that earthen pot could be used as a cheap proton exchange membrane [12]. In order to reduce the cost in the production of SMFC, earthen pot could be used as cheap alternative to expensive proton exchange membranes. Proton exchange membranes in most cases aid by improving voltage production by facilitating the transfer of protons at the cathodic compartment of an MFC. When earthen pot was used as a proton exchange membrane (Fig. 2) the voltage increased over an initial period in both trials compared to configurations without earthen pots. The average difference in voltage in SMFC with earthen pot over SMFC without earthen pot was 80 mV with the maximum voltage from trial 1 and trial 2 being 570 mV and 650 mV respectively (Fig. 2). Results suggest that earthen pot could be used to slightly increase voltage but may need further optimization for significant increases over extended periods. |
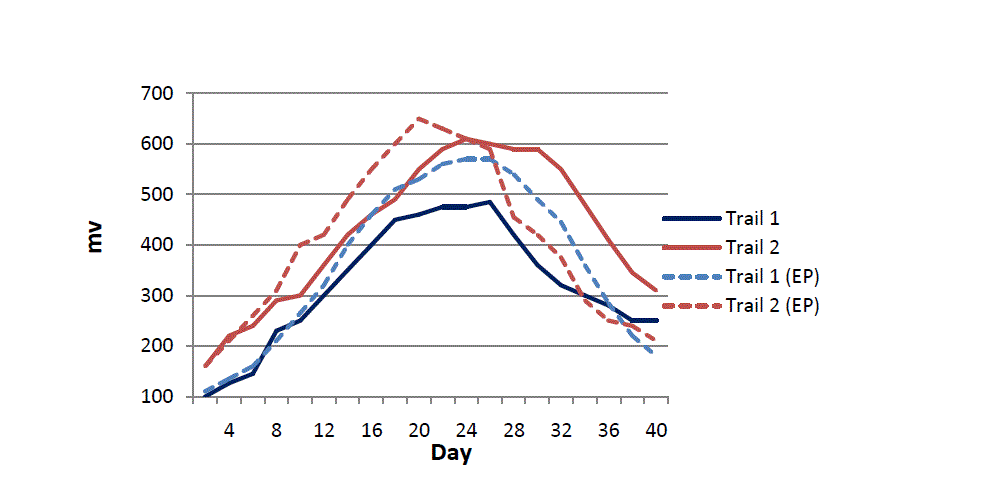 |
| C. Comparison of Different Cathodes |
| In this study carbon brush was used as an anode and cathode due to its wide use as efficient electrodes in microbial fuel cells [7] and also due to its reduced cost. However, different cathodes were tested to investigate their suitability in voltage generation. Carbon brush was compared to that of stainless steel and carbon rod. Among them Carbon rod gave the highest voltage in MFC compared to other cathodes (Fig. 3). Stainless steel gave the lowest voltage indicating it to be a poor cathode in MFC configuration. |
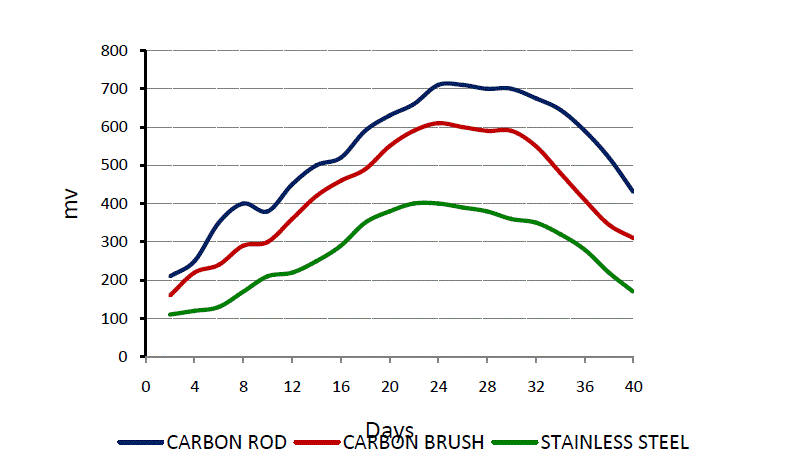 |
| D. Current Density Vs Power Density |
| Fig. 4, illustrates the variation of current density (CD) with power density (PD) found from the SMFC chamber. The highest current density and power density was obtained after 2 weeks which was 150 mA/m2 and 125 mW/m2 respectively. The values represent projected surface area of anode. Although the power density obtained may not scale directly with size, reports have shown that power generation scales almost linearly with anode size up to about 1–2m2 [11]. |
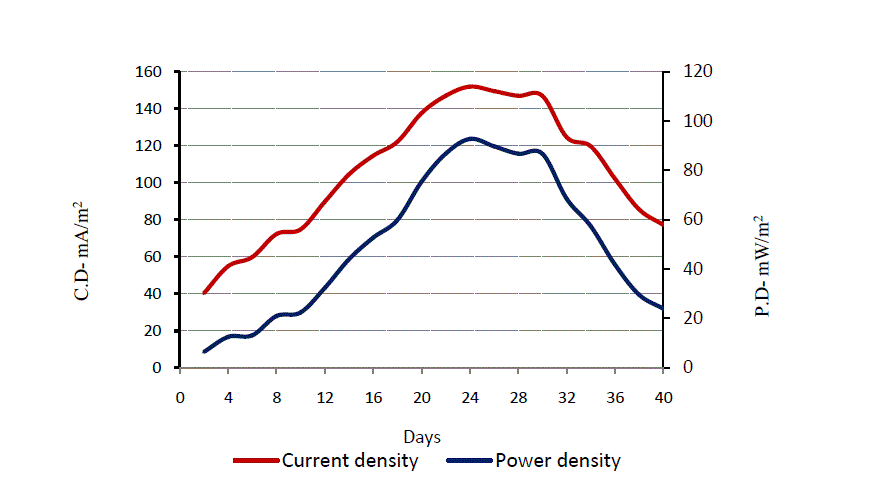 |
| E. Functional Application of SMFC |
| Nine SMFC plants were joined together, after 20 days in a series connection and then the voltage output was measured by a multimeter. A small LED lamp and a scientific calculator were used to see whether the constructed SMFC could power up such devices. A voltage of 3.8 volts and 4.7 mA was found, which was sufficient to power up the LED bulb and scientific calculator (Fig. 5). However, the total voltage found was not equal to the voltage of individual SMFC’s in a series. Such has been reported in single chambered MFC connected in a series by other authors [13]. |
 |
IV. SUMMARY |
| The findings of this experiment indicate that sediments like mangrove forest sediment can be used as a potential inoculum for bioelectricity generation to power small devices. Mangrove forest sediment always contains a wide range of organic matter and microorganisms, which may result in high electrogenic activity. The highest voltage obtained from this type of SMFC configuration is consistent with other findings reported elsewhere. The materials used in the construction of SMFC are very cheap, available and is easy to assemble. Though further improvements this set up can be well suited to the socio-economic condition of rural environment in the countries like India and Bangladesh. Future studies may involve analysis of the microbial biofilm produced on the anode surface and identification of novel electrogenic bacteria. The reduction in SMFCs size can be studied by reducing the number of SMFCs in a series connection by studying the effect of multi anode configurations. Although it has been possible to light up sensors the output is still further away from practical power generation. |
References |
|