e-ISSN: 2320-0812
e-ISSN: 2320-0812
Kamaldeep Singh*, Gurvinder Pal Singh, and Sandeep Kumar Sharma
Adesh Polytechnic College, Sri Muktsar Sahib 152026, Punjab, India.
Received Date: 08/06/2013; Accepted Date: 21/06/2013; Published Date: 28/06/2013
Visit for more related articles at Research & Reviews: Journal of Pharmaceutical Analysis
In the present work simple and sensitive RP-HPLC method has been developed for the quantitative estimation of Baclofen in bulk drug and pharmaceutical formulations. A rapid and sensitive RP-HPLC Method with UV detection (253 nm) for routine analysis of Baclofen in Bulk drug and Pharmaceutical formulation was developed. Chromatography was performed with mobile phase containing a mixture of Methanol: Water: Acetic Acid (100:100:0.5) with flow rate 1.0 ml/min The linearity was found to be in the range of 10-50 μg/ml with (r2=0.999) A known amount of the drug solution (80, 100 and 120 %) was added to the powder sample of the tablet formulation and subjected to the estimation of the drug for the recovery studies. There was a high recovery of Baclofen (100.03%, 100.18%, and 100.05%) Robustness of the proposed method was determined by analysis of sample by changes in different parameter like flow rate, detection and Column temperature using similar operational and environmental conditions, the tailing factor was found to be less than 2. The precision of the assay was determined in terms of intra and inter-day variation in the peak area for a set of drug solution (30 μg/ml) assayed three times on the same day and on three different days The intra and inter day variation in the peak ratio of the drug solution was calculated in terms of co-efficient of variation (CV) and obtained by multiplying the ratio of standard deviation to the mean with 100(CV=SD/MEAN X 100)All the parameters are compared with standard raw material of baclofen. The proposed method was validated in accordance with ICH parameters and applied for analysis of the same in marketed formulations. The proposed method was validated by determining sensitivity, accuracy, precision, robustness stability.
Analytical Method Development
Analytical method development plays an important role in the discovery, Development, and manufacture of pharmaceuticals. The number of drugs introduced into the market is increasing every year. These drugs may be either new entities or partial structural modification of the existing one. Very often there is a time lag from the date of introduction of a drug into the market to the date of its inclusion in pharmacopoeias. This happens because of the possible uncertainties in the continuous and wider usage of these drugs, reports of new toxicities, development of patient resistance and introduction of better drugs by competitors. Under these conditions, standards and analytical procedures for these drugs may not be available in the pharmacopoeias. It becomes necessary, therefore to develop newer analytical methods for such drugs. Various analytical technique (HPLC, UV-spectroscopy, HPTLC, Titration, Fluorescence spectroscopy) are used by Quality control laboratories to ensure the identity, purity, potency and performance of drug products. Most of the drugs in multicomponent dosage forms can be analyzed by HPLC method because of the several advantages like rapidity, specificity, accuracy, precision and ease of automation in this method. HPLC method eliminates tedious extraction and isolation procedures. Optimization parameters in HPLC are mainly partition coefficient, pKa and solubility of drug. Column selection is most important part of HPLC method development. Column selection depends upon polarity, pH range of buffer and amount of compound eluting out. It also depends upon the functional groups present in drugs. If the sample is polar then C8 or C4 columns are preferred to use.
Drug Profile
Description
Baclofen is a gamma-amino-butyric acid (GABA) derivative used as a skeletal muscle relaxant. Baclofen stimulates GABA-B receptors leading to decreased frequency and amplitude of muscle spasms. It is especially useful in treating muscle spasticity associated with spinal cord injury. It appears to act primarily at the spinal cord level by inhibiting spinal polysynaptic afferent pathways and, to a lesser extent, monosynaptic afferent pathways.
Structure
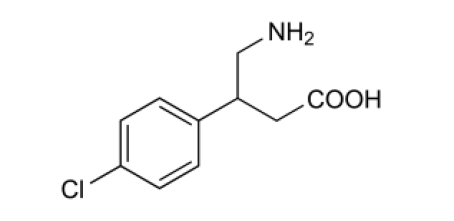
Chemical Structure of Baclofen
IUPAC : β-(Aminomethyl)-4-chlorobenzenepropanoic acid
Appearance : White or off white crystalline
Molecular weight: 213.67g/mol.
Molecular formula: C10H12CLNO2
Solubility : Easily soluble in water and methanol
Melting point range : 206-208oC
Pka 3.782
Storage: Stored in high-density polyethylene bottle (HDPE), with silica gel desiccant canisters or sachets, and polyester fiber packing material
Dose: Baclofen is taken orally, in either tablet according to the prescribing physician's, instructions. Baclofen is prescribed by physicians in varying daily dosages, usually ranging 5mg 3-4 times a day to be increased every 3-4 days up to a maximu m of 80mg per day [1,2,3,4,5,6,7,8,9]
The pharmacokinetics of Baclofen is administered orally and is primarily excreted and changed in the urine with a half life 3-4hrs.80% is excreted unchanged in urine within 72hrs
Precautions
A physician should be consulted before taking Baclofen with certain non-prescription medications. Patients should avoid alcohol and CNS depressants (medications that make one drowsy or tired, such as sleep medications, and some pain medications), while taking Baclofen. Baclofen can exacerbate the side effects of alcohol and some other medications.
Baclofen may not be suitable for persons with a history of liver or kidney disease, depressed renal function, mental illness, anemia, high blood pressure, angina (chest pain), or irregular heartbeats and other heart problems.
Therapeutic Uses
Baclofen is a derivative of gamma-aminobutyric acid (GABA). It is primarily used to treat spasticity and is under investigation for the treatment of alcoholism. baclofen retains its therapeutic anti-spasmodic effects even after many years of continued use. Baclofen can also be used to treat hiccups [10,11,12,13,14,15,16,17,18].
The specifications of HPLC instrument used are as follows. A gradient high pressure liquid chromatograph (Jasco, RI 2031 Plus) pumps Jasco PU 2080 Plus, IR detector column oven (Jasco), system controller (Jasco), a disposable guard column LC-18 (Jasco)TM , LC-18, 2 cm, supelco, inc., Bellefonte, and a Reverse Phase C-18 (phenyl) Column (25 cm x 4.6 mm i.d., particle size 5 μm) was used The HPLC system was equipped with the software, Borwin (Jasco) [3].
Drug sample
Standard Baclofen was obtained as a gift sample from Akums drugs and Pharmaceutical ltd.,Ranipur, Hariwar.
Marketed formulations
Baclof (10 mg tablets) from Sun pharma Pvt. Ltd., Mumbai, was procured from local market. In the present investigation, an gradient high pressure liquid chromatograph (Jasco HPLC ) with one LC-10 AT VP pump and UV/Visible detector (JASCO) SGE-11A VP, CTO-10 AS VP column oven (JASCO), system controller (JASCO), a disposable guard column LC-18 (Jasco)TM LC-18, 2 cm, and a Reverse Phase C-18 Column (25 cm x4.6 mm i.d., particle size 5 μm) was used. The HPLC system was equipped with the software class, BORWIN (JASCO).
A rapid and sensitive RP-HPLC Method with UV/Visible detection (272nm) for routine analysis of Baclofen in Bulk and in Pharmaceutical formulation wasdeveloped. Chromatography was performed with mobile phase containing a mixture of Methanol: Water: Acetic Acid (100:100:1) and flow rate 1.0 m-l min. In the range of 10-50 μg/ml, the linearity of Baclofen shows a correlation co-efficient of 0.9993.The proposed method was validated by determining sensitivity, accuracy, precision, robustness, stability, specificity, selectivity, and system suitability parameters.
Assay of Baclofen in Tablets
Twenty tablets were weighed and finely powdered. An accurately weighed quantity of the powder equivalent to 100 mg of Baclofen was transferred to 50 ml volumetric flask containing 25 ml of water (100 volume) and Glacial acetic acid (1 Volume) the contents of the flask were sonicated for 15 min, to ensure the complete solubility of the drug, then the mixture was made up to 50 ml with methanol.
The resulting solution was thoroughly mixed and filtered through a 0.45 μm membrane filter. From this a working standard solution of 100 μg/ml of strength was prepared. Aliquots of 10, 20, 30, 40 and 50 μg/ml were prepared in 100 ml volumetric Flasks & diluted with methanol. This each concentration (20 μl) was injected six times into the column. The mean values of peak areas were calculated and the drug content in the tablets was quantified.
Linearity and range
By using the working standard, aliquots of 1800,1900,2000,2100, and 2200 ppm were prepared with methanol three dilutions of each of the above mentioned concentrations were prepared separately and from these three dilutions, 20 μl of each concentration was injected into the HPLC system and their chromatograms were recorded. Peak areas were recorded for all the peaks and a standard calibration curve of peak area against concentration was plotted. and result shown in
Accuracy
The procedure for the preparation of solutions for Accuracy determination at 80%, 100% and 120% level were prepared in the same manner as explained in method A But in this method mobile phase is Methanol: Water: Glacial acetic acid (100:100:0.5 v/v) was used as solvent. The solutions were filtered through 0.45 μm membrane filter paper and then they were subjected to analysis by RP-HPLC method under the same chromatographic conditions as described above. At each level, three determinations were performed. The results obtained were compared with expected results and were statistically validated.
Precision
The precision of the assay was determined in terms of intra and inter-day variation in the peak area for a set of drug solution (20 μg/ml) assayed three times on the same day and on three different days. The intra and inter day variation in the peak ratio of the drug solution was calculated in terms of co-efficient of variation (CV) and obtained by multiplying the ratio of standard deviation to the mean with 100(CV=SD/MEAN X 100) .
Specificity
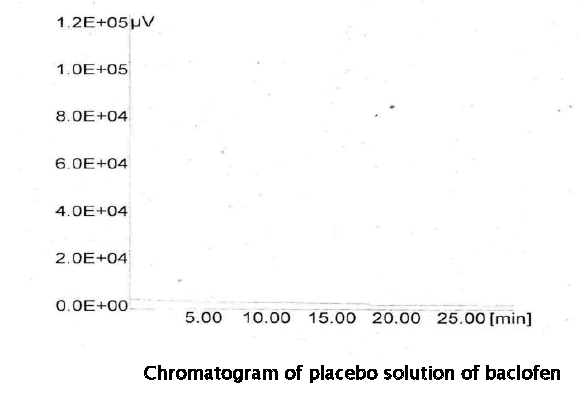
Placebo solution (prepared similarly as the sample solution using placebo for tablet) and the sample solution was prepared and the analyzed by the proposed method. There was no interference from any of the excipients at the retention time of the baclofen peak. The peak purity plot indicate that the baclofen peak is homogeneous and has no co-eluting peaks indicating specificity of the method.
Robustness
As defined by the ICH, the robustness of an analytical procedure describes to its capability to remain unaffected by small and deliberate variations in method parameters. Robustness was performed by small variation in the chromatographic conditions and found to be unaffected by small variations like ± 0.2 ml/min in flow rate of mobile phase, wavelength ±2nm and PH ± 1.0 results are reported.
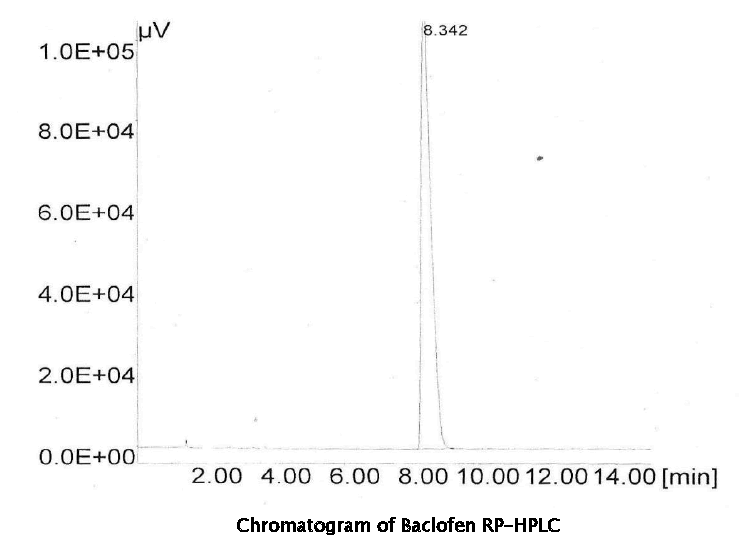
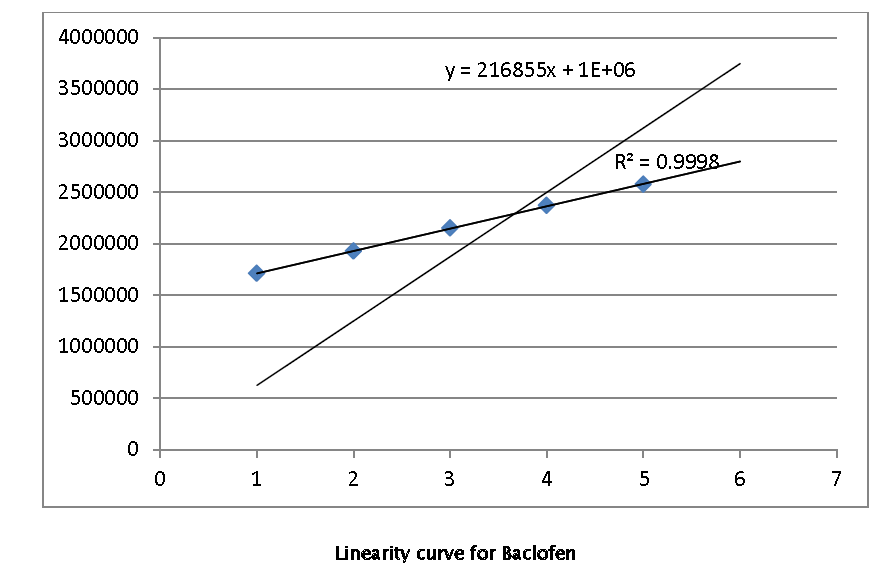
The objective of this study was to develop a rapid and sensitive HPLC method for the analysis of Baclofen in bulk drug and in its tablet formulations using SGE analytical C-18 column with UV- detection. In this method the conditions were optimized to obtain elution of Baclofen Initially, various mobile phase compositions were tried to elute Baclofen. Mobile phase and flow rate selection was based on peak parameters (height, tailing factor, theoretical plates, capacity or asymmetry), run time, resolution. The system with Methanol: Water: Acetic Acid (100:100:1) The run time was set at 15 min and the retention time for Baclofen was found to be 8.08 min as shown in Fig. The sample solution was injected 6 times and the retention times were found to be the same. The average of 6 such determinations of peak areas are shown. When the concentrations of Baclofen and its respective peak areas were subjected to regression analysis, a good linear relationship (r2= 0.999) was observed between the concentration of Baclofen and their respective peak areas in the range 10-50 μg/ml. The regression of Baclofen was found to be Y = 21685X + 1E+06 where ‘Y’ is the peak area and ‘X’ is the concentration of Baclofen reported.
The regression equation was used to estimate the amount of Baclofen, in tablet formulations and in validation study (precision and accuracy). A known amount of the drug solution (80, 100 and 120 %) was added to the powder sample of the tablet formulation and subjected to the estimation of the drug for the recovery studies. There was a high recovery of Baclofen (100.03%,100.18%, and 100.05%) indicating that the proposed procedure for the determination of Baclofen in the tablet formulation is accurate
Robustness of the proposed method was determined by analysis of sample by changes in different parameter like flow rate, detection and Column temperature using similar operational and environmental conditions, the tailing factor was found to be less than2 and is reported.The proposed method was validated in accordance with ICH parameters and applied for analysis of the same in marketed formulations.
The method is precise and high percentage of recovery of as shown in table shows the method is accurate. The HPLC method is more sensitive, precise and accurate and can be adopted in routine analysis of drug formulation compared to the methods. The accuracy and reproducibility of the proposed method was statistically validated by recovery studies.