e-ISSN: 2321-6182 p-ISSN: 2347-2332
e-ISSN: 2321-6182 p-ISSN: 2347-2332
KN Sunil Kumar1*, Suchitra Narayan Prabhu1, B Ravishankar1, Sahana2, B Yashovarma3
1SDM Centre for Research in Ayurveda and Allied Sciences, Laxminarayana Nagar, Kuthpadi, Udupi–574 118, India
2Mangalore University Post Graduate Centre, Cauvery Campus, Madikeri, Kodagu-571 201, India
3SDM College (Autonomous), Ujire, Belthangadi Taluk, Dakshina Kannada-574 240, India
Received date: 17/06/2015 Accepted date: 28/06/2015 Published date: 05/07/2015
Visit for more related articles at Research & Reviews: Journal of Pharmacognosy and Phytochemistry
Background: Aerva lanata (Linn.) Juss. ex Schult. - Amaranthaceae is an herbaceous weed found throughout tropical India. The plant is known as pashanabheda/ gorakshaganja in Ayurvedic system of medicine. The roots are diuretic and used as cure for urinary troubles including stones. In the present manuscript, experimental evidences regarding antiurolithiatic activity of roots of A. lanata are reported in support of its use in lithiasis.
Objective: In the current study it attempted to evaluate the effectiveness of roots of Aerva lanata on renal calculus formation In vitro.
Methods: Investigation of antiurolithic activity was carried out by simultaneous flow static model (S.S.M). Dissolution of precipitate of calcium oxalate and calcium phosphate was determined by (S.S.M).
Results: One per cent aqueous extract preparation of A. lanata root was good at controlling calcium phosphate crystallization. Five per cent aqueous extract was good on calcium oxalate crystallization though it controls both type of crystal formation.
Conclusions: This attempt would provide encouragement for further research on novel drug development for prevention and treatment of urolithiasis.
Pashanabheda, Calcium oxalate and phosphate nucleation, oxaluria
Aerva lanata (Linn.) Juss. ex Schult. - Amaranthaceae is an erect to prostate herb, hoary- tomentose, dioecious, found throughout tropical India. It is a common weed in fields and waste places, and ascends up to an altitude of 900 m in the hills. It has a tap root which is cylindrical, branched, 7 to 12 cm long, 2 to 8 mm thick, straight or slightly twisted with fibrous lateral roots, pale yellowish brown externally, whitish internally, and with camphoraceous odor [1]. The plant is known as pashanabheda and gorakshaganja in Ayurvedic system of medicine. The whole plant is used in diabetes, for arresting bleeding in pregnancy [2], as demulcent and in lithiasis. Other properties ascribed for the plant are for uterus clearance after delivery [3]. The plant is also efficacious as diuretic [4] and dissolve kidney and gall bladder stones [5,6].
The plant was reported to contain sitosteryl palmitate [7]; hentriacontane, β- sitosterol, its D-glucoside, α-amyrin, and betulin [8]. Glycosides like kaempferol-3-galactoside and kaempferol-3-rhamnogalactoside along with alkaloids, saponins, sugars (fructose, rhamnose, galactose and sucrose) and minerals (Aftaq et al.,) [9] are also reported.
The root has diuretic, to cure diarrhoea, in urinary troubles [10] and as antirheumatic [11]. In the present manuscript, phytochemical study and experimental evidences regarding antiurolithiatic activity of roots A. lanata are reported, the result being in support of its use in lithiasis.
Collection of plant material and extraction
Root of Aerva lanata was collected from SDM College of Ayurveda campus, Kuthpady, Udupi. The plant was identified and authenticated by referring to Flora of Udupi [12] with voucher specimen number 609/15050901 was deposited at Pharmacognosy department of SDM Centre for Research in Ayurveda and Allied Sciences, Kuthpadi, Udupi. Shade dried plant material was stored in air tight container at 250°C for further study. Cold percolation method was followed for preparation of aqueous extract and phytochemical studies. The plant material weighing about 100 g was soaked with 2 litres of water and ethanol in a percolator for 24 hrs followed by filtration, the filtrates were poured individually to a pre-weighed china dish and concentrated to dryness over water bath and the dried residue was made moisture free in a desiccators. The extract thus prepared was used for phytochemical tests, HPTLC and In-vitro antiurolithiatic activity by simultaneous flow static model (S.S.M) [13].
Physico-chemical examination
The powdered plant material was standardized as per Pharmacopoeial methods [14].
Phytochemical screening
Total ethanol extract was tested for the presence of different phytoconstituents like alkaloid, steroid, flavonoid, tannin, glycoside etc. [15].
Sample preparation: Dried plant root powder (1 g) was extracted with 10 mL of ethnol (90%) and filtered. The filtrates was made up to 10 mL and used for application (Figure 1).
Development and documentation: Three, six and nine micro litres of the sample were applied on aluminium plate pre-coated with silica gel 60 F254 of 0.2 mm thickness (Merck, Germany) using CAMAG LINOMAT 5 applicator. The plate was developed in CAMAG glass twin trough chamber previously saturated with mobile phase toluene : ethyl acetate: formic acid (7.0:1.0:0.1). The plate was derivatized using vanillin- sulphuric acid (VS), and heated at 105 °C till the spots appeared [16,17]. The developed plates were visualized in CAMAG visualizing chamber and scanned in CAMAG Scanner 4 under 254, 366, 540 (pre-derivatisation) with the help of CAMAG WinCATS software. Rf values and densitograms were recorded.
Antiurolithiatic activity In-vitro: Inhibition of calcium oxalate and calcium phosphate mineralization by aqueous extract of Aerva lanata was measured by simultaneous flow static model (S.S.M.). Inhibition of calcium oxalate and calcium phosphate mineralization, procedures were carried out in two sets; one served as blank set and other as experimental set, in blank set, 0.1 M sodium oxalate (10 ml), and 0.1 M calcium acetate (10 ml) were taken in two separate burettes where as in experimental set aqueous extract of the plant i.e. in the concentration of 1.0%, 5.0% (10 ml) were taken in the third burette. In both sets of experiments, chemicals were allowed to fall simultaneously, slowly and at steady pace into a 250 ml beaker. After 30 minutes, the mixtures were kept in hot water bath for 10 minutes, cooled to room temperature and collected into a pre-weighed centrifuge tube. Centrifugation of mixture was done at 3000 rpm for 15 minutes. Supernatant fluid was discarded and precipitate was obtained. All tubes with precipitate were dried in a hot air oven at 1200°C, cooled to room temperature and weighed. Similar process was repeated using 0.1 M sodium phosphate (25 ml) and 0.1 M calcium acetate (25 ml) for inhibition of calcium phosphate mineralization. The percentage inhibition of mineralization was calculated by the following formula [13].
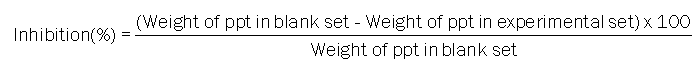
Number of medicinal plants such as Kanghi (Abuliton indicum), Chaya (Aerva lanta), Bishkapa or punarnava (Boerhaavia diffusa), Ajuba (Bryophyllum pinnatum), Gokhuru (Tribulus terrestis), Makka (Zea mays) etc shows antiurolithiatic activity.
Standardization was carried out as it is an important aspect in maintaining and assessing quality and safety of the crude drug. The herb was found to have no other parts as foreign matter. Only roots of the sample were selected for the study. Loss on drying reveals the moisture content, the sample has 5.09% of moisture; total ash is the indication of total inorganic content, 8.99% ash was detected in the sample; acid insoluble ash is the acid insoluble part of total ash, mainly silica, the sample showed 0.40% acid insoluble ash; water soluble ash is the water soluble part of total ash indicating inorganic content without water insoluble inorganic salts like silica, 4.68% was water soluble; water and alcohol soluble extractive is indicative of percentage active constituents soluble in water and ethanol, the values were 15.73 and 3.69% respectively. These physico – chemical standards would indicate the purity and authenticity of the leaves of A. lanata (Table 1).
Preliminary phytochemical screening revealed the presence of carbohydrates/glycosides, phenols, steroids, tannins. The phytochemical constituents present in the extract can be held responsible for different medicinal activities of the plant (Table 2).
HPTLC finger printing profiles of Aerva lanata under 254 nm showed the presence of 3 spots (all in green) at Rf of 0.43, 0.55 and 0.62. Under 366 nm there were 7 prominent spots (Fluoroscent) at Rf of 0.13, 0.43, 0.55, 0.62, 0.66, 0.85, 0.92 and when scanned under white light 540 nm, 3 spots were present at Rf 0.43, 0.55, 0.62, following post derivatisation with vanillin sulphuric acid spots( in different colors) were evident at Rf 0.15, 0.38, 0.43, 0.55, 0.57, 0.64, 0.85. Among these the spots were common at Rf of 0.43, 0.55 and 0.62 (except at post derivatisation) at different color intensities (Table 3 and Figure 2).
Rf values by densitometric scan of Aerva lanata showed 18 spots at 254 nm, 10 spots at 366 nm, and 5 spots at 540 nm respectively (Table 4 and Figure 3).
Urolithiasis is formation of one or more calculi in any location within the urinary tract; it is one of those oldest diseases known to mankind [18]. Cause is multifactorial and is related to dietary life style habits or practices [19]. It is common in age group of 20-40 in both sex but men are more prone than women [20]. Lithiasis is categorized into 2 types depending on its anatomical position; when formed in bladder, ureter or any part of urinary tract rather in kidneys as urolithiasis and on the other hand termed as nephorolithiasis when formed in kidneys [21]. Calcification from urine super saturation results in precipitation and subsequent crystallization depends on urinary pH, ionic strength, solute concentration and complexation/ Chelation [22] and is a multistep process which results with formation and ends in retention; crystal nucleation, crystal aggregation, crystal growth and its retention [23]. The precursor for oxalate crystal is oxalic acid/ oxalates biosynthesized from ascorbic acid, glycolates and glyoxylates. So for prevention of stone formation as precautionary measure the oxalate rich food should be avoided, such as spinach, nuts like almonds, wheat bran, strawberries, ascorbic acid rich foods, certain calcium supplements should be avoided as it has tendency to bind to oxalates inducing oxaluria, hypocalcaemia and increase risk of stone formation [24]. It is advisable to ingest large amount of fluid like water and barley and restrict potassium and protein intake [25].
A stone mainly comprises of calcium oxalates [26] and trace amount of calcium phosphates and calcium carbonates [27]. Stones bigger than 5 mm which fails to pass through requires medical intervention, involving extracorporeal shock wave lithotripsy (ESWL), Ureteroscopy (USP), percutaneous nephrolithotom y(PNL) [28] but unfortunately propensity of reoccurrence is not altered and is still about 50% [29]. ESWL has side effect like renal damage, it also results in induced hypertension or renal impairment [30]. In fact there is still no treatment for the prevention of reoccurrence of stones.
As there is a urgent need for the prevention of urinary caliculi formation the investigation carried out in-vitro for calcium oxalate and calcium phosphate mineralization satisfies the need for its potential activity. One % aq. extract of Aerva lanata was effective in controlling calcium phosphate mineralization where as 5% aq. extract was found to inhibit calcium oxalate and calcium phosphate mineralization. One % aq. extract was effective in controlling calcium phosphate mineralization to an extent of 68.22% in comparison with calcium oxalate mineralization restricting it to only 36.44%.
On the contrary 5% aq. extract was effective in controlling both types of crystal mineralization; however it had an upper hand in controlling calcium oxalate than calcium phosphate. Five %aq. extract of A. lanata showed maximum activity in inhibiting calcium oxalate and calcium phosphate mineralization to percentage of 68.53% and 58.05% respectively (Table 5).
The extract had an appreciable potential in controlling calcium oxalate and calcium phosphate mineralization, this property of the extract supports for the claim that chances of crystal formation is reduced thereby preventing its aggregation and subsequent retention in the urinary tract.
The present investigation provides useful information on antiurolithiatic activity of aq. extract of Aerva lanata in controlling calcium oxalate and calcium phosphate mineralization thus this study has high significance in the prevention of urolithiasis. Further studies should be done to understand pharmacological action and its possible mechanism through elaborate preclinical experimentation and clinical trials in preventing urolithiasis in susceptible population.
Authors are grateful to revered President, Dr. D. Veerendra Heggade, SDM Educational Society for the encouragement.