ISSN:2321-6212
ISSN:2321-6212
Ismail Kamil*
Department of Materials Science and Engineering, Jimma Institute of Technology, Jimma University, Jimma, Ethiopia
Received: 24-Jun-2022, Manuscript No. JOMS-22-64507; Editor assigned: 27-Jun-2022, PreQC No. JOMS-22-64507(PQ); Reviewed: 11-Jul-2022, QC No. JOMS-22-64507; Revised: 18-Jul-2022, Manuscript No. JOMS-22-64507(R); Published: 25-Jul-2022, DOI: 10.4172/2321-6212.10.6.005.
Visit for more related articles at Research & Reviews: Journal of Material Sciences
This review article provides an overview the way of improving electrical conductivity of scrap melted aluminium conductor by using different types pre-melt treatments in cable industry. This pre-melt treatment only by itself is not the way of achieving high purity aluminium from scraps but by combining with in-melt treatment which is using different fluxing chemicals and gases we can achieve high purity aluminium conductor product from aluminium scrap production. In Africa there are a numbers of cable industries but their usage of secondary material is very low due to the lack of product quality. One reason for this problem is lack of the steps in secondary materials treatment. Scrap melting and purification of the product controlled by energy consideration of melt process. From several metals aluminium has highest difficulty to achieve desired purity from its secondary source during melting. But there are different kinds of solution to decrease the negative impact of impurity in aluminium scrap melted products which can be applicable in pre-melt and in-melt stages. Pre-melt treatment is critical to achieve desired purity with the combination of in-melt treatment once high impurity enters in melting process it makes difficult to purification and we can make ease in-melt treatment by using this pre-melt treatment. Due to it’s widely usage the availability of aluminium scarps is high enough and this secondary material usage has enormous benefit in economic and environmental perspective. However, the presence of deleterious impurities in recycled aluminium product are the main drawback especially for electrical application since electrical conductivity property highly depends on purities of the materials. There are different kind of solutions to deal with
undesired impurity which can be apply before melting process and during melting process. This work gives an overview about removing undesired impurities from aluminium scraps before melting process specifically for electrical conductivity application.
Scraps; Conductivity; Pre-melt treatment; Resistivity; Impurity
Aluminium is mostly and widely used non-ferrous metals on the earth. Aluminium and aluminium alloy uses for foil, beverage cans, cooking, food processing, architectural, electrical application, and structure for boats, aircraft and for other types of transportation. Aluminium has highest corrosion resistance because of naturally occurring surface oxide films (Al2O3) [1]. That is why aluminium found in large area of application. On equal weight and cost basis, aluminium is a better electrical conductor than copper. Its high thermal conductivity leads to applications such as radiators and ± cooking utensils. Its low density is important for different applications. Wrought aluminium alloys display a good combination of strength and ductility. Aluminium alloys are among the easiest of all metals to form and machine. The precipitation hardening alloys can be formed in a relatively soft state and then heat treated to much higher strength levels after forming operations are complete. In addition, aluminium and its alloys are not toxic and are among the easiest to recycle of any of structural materials [1]. The ease with which aluminium may be fabricated into any form is one of its important assets. Aluminium can be cast any methods and aluminium can be rolled rod; it can be stranded into cable with and required size and type. There is almost no limit to the different profile in which the metal can be extruded [2]. During aluminium industry development time primary producers wanted to increase new aluminium sales, minimize the price, and developing the aluminium alloys and production process that were essential to be more competitive with available materials. And there was no interest by them to recycle aluminium scrap that leads to the development of independent secondary industry. But now due to the industry grew both primary and secondary aluminium industries acquiring and using aluminium scraps because of the availability of different scrap treatment technologies and secondary aluminium producer started producing advanced end products and it reduced the original distinctive difference between primary and secondary aluminium industries. Even they are started producing electrical conductor cable from secondary material for power transmission which is highly sensitive for existence of fractions impurities. Similar development occurred in technology arena and secondary producers were originally low capital salvage operators. In Ethiopia from all over headed electrical conductor around 90% made from aluminium metals because of its weight and high electrical conductivity combination. Aluminium conductor is best choice for overhead power transmission in all over world.
Electrical conductivity and effects of impurity
Electrical conductivity of the material mainly depends on purities of the material and electrical resistance of the materials. The physical, mechanical, electrical, and chemical properties of aluminium metals and aluminium alloys depend upon composition and microstructure. The addition of selected elements to pure aluminium greatly enhances its properties and usefulness. But the addition of any elements decreases electrical conductivity of aluminium.
One of the most important characteristics of solid material is the ease with which it transmits an electric current. Ohm’s law relates the current I or time rate of passage to the applied voltage V.

where R is resistance of the material through which current is passage. V is applied potential (voltage) and I is current strength in ampere. But resistance depends on the cross section, purity and length of a uniform conductor [3].
The resistivity of pure aluminium is 2.64 × 10-8 âÃâæ m at 20°C and its electrical conductivity is 66% in International Annealed Copper Standard (IACS) at 20°C. The density of aluminium is 2.7 g/cm3. Electrical resistance is a property of all materials by a measure of which it opposes electrical current [1].
Resistance calculation from specific resistivity given as
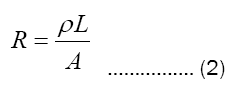
where ρ is specific resistivity, L length of the conductor and A is area of the conductor [3].
Resistivity is the resistance of a conductor whose length and area of cross section=1 this fundamental property of all metal’s resistivity increases with temperature. For pure metal resistivity at a given temperature is constant. Matthiessen’s rule is concerned with electron mobility which in turn decides electrical conductivity and resistivity of metals [4]. Electrical conductivity of a metal can be well explained in the light of free electron model of metals. Metal contains free electron but those free electrons are confined within the metal unless an electrical field is applied. Then free electron travel through the metal and connected conductor. Electron mobility is hampered if electron undergoes collision with impurities. That is why presence of impurities lowers the conductivity of a metal. As a matter of facts electrons are scattered by impurities atoms and as a result electrical conductivity of a metal is decreased. Conversely resistivity increases. Electrons are also scattered by phonons (lattice vibration) as the temperature is raised lattice vibration is also increase resulting greater concentration of phonon. These phonons can scatter free electrons and hence the electrical conductivity is lowered.
The Dc resistivity of metals
The customary approximation used in the discussion of the resistivity of metal is Mathiessen’s rule. Matthiessen’s rule-the statement that the electrical resistivity ρ of a metal can be written as
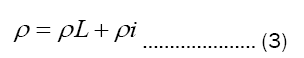
where ρL is due to scattering of conduction electron by lattice vibrations and ρi to scattering by impurities and imperfections. If the impurity concentration is small, ρi is temperature independent.
Impurity level of aluminium is directly related to the electrical resistivity (and conductivity) of aluminium. The resistivity of a pure metal is increased by impurities due to the lattice distortion that affects electron scattering that is partially responsible for the electrical conducting property of the metal. Matthiessen’s rule shows the dependency of electron mobility of electron scattering contributions from impurities and lattice phonon. Electron mobility defines electrical conductivity and resistivity.
Aluminium scrap recycling
Aluminium can be produced from primary and secondary sources. Primary source means that the production of aluminium from its ores that are found in earth crust by using different physical and chemical process and secondary source mean aluminium production from different types of aluminium used scrape or in other word recycling. Primary production needs high energy amount for production such that for one kilogram aluminium production it requires around 186 MJ energy for secondary source production the energy need is very low with compare with that of primary source and it is around for one kilogram aluminium production only 10-20 MJ energy required. However, the existence of impurity in secondary sources or aluminium scraps long-term compositional barrier [1,5]. A number of studies suggests that the existence of unwanted impurities is growing problem. Some of common elemental impurities of aluminium scrapes are magnesium, silicon, nickel, lead, zinc, copper, iron, vanadium, and manganese. Scrap by its self can be divided in two types which are new scrap and old scrap. Composed of leftover aluminium generated during production called new scrap and aluminium that recovered from recycled consumer product is called old aluminium scraps including the finished beverage cans, used aluminium conductors, from car body, home used items and etc [6,7].
Aluminium can be recycled without loss of its quality and profitable ways for several times and it helps to control carbon dioxide emission from weight reduction of different vehicles. Environmental and industrial perspective it involves reducing the consumption of resources, minimizing the use of energy, materials and water [5].
Scrap preparation: Due to widely usage of aluminium metals for different application the availability of aluminium scrap is high. Now a day the processing of aluminium scrap becomes more attractive. It has complex chemical composition-based aluminium and on the base of their previous application their purity is differ. Magnesium, zinc, silicon, iron, copper, etc. are the main and common impurities of aluminium scraps. So, one should consider during scrap recycling for what purpose is recycling is doing. For electrical conductor production the scrap background must be high purity because the maximum purity of aluminium required for electrical application and electrical conductivity is highly depends on material purity. During scrap collection for conductor production the scrap may be previous used conductor, household scraps, and high purity scraps. Using automotive scraps may create difficulty to achieve high purity product because of their high alloying element content Figure 1.
Pre-melt treatment
Magnetic separation: Magnetic separation uses to separate aluminium scrap from ferrous metals components. The purpose of magnetic separation for removing magnetic metals such as iron, nickel, their alloy, and if any are present. There are different types of magnetic separators available for magnetic separation and the most common magnetic separator are drum magnets and overhead belt magnets. Drum magnet is stationary featured magnet around which drum rotates and nonmagnetic scraps are fall off the drum and are collected on conveyor running below magnetic materials sticks to the drum and continues to do so until the drum rotates past the magnetic field that is generated. After it falls and is removed by separate conveyor. In second types of magnetic separator which is belt magnet a conveyor belt and with the scrap is fed near another conveyor belt NbFeB magnets. When the scrap approaches to this magnet the scraps which has magnetic property attracted to the magnet and pulled to another conveyor belt and the nonmagnetic scraps are falls into collection bin. This technology uses to only to separate aluminium scrap from magnetic materials inclusion but not others impurities such as plastics, glass, rubber, etc Figure 2[6,8].
Air separation
Metal scraps often contain high level of low-density impurities. Most of the time waste materials or scraps are found with waste paper and plastics. When the fraction of this kind of waste exists in raw materials air separation method is essential since the waste materials are with low density [6]. And air separator technology has different mechanisms of working such as wind fitting, air knives, elutriation, winnowing, air column, etc. their names refer their mechanisms of working. To pull light-weight materials that are present in scrap conveyor belt system is often used. Plastic, foam and rubber are commonly found as light-weight inclusions and they can be easily treated by this kind of method. Vertical air separator system uses air pushing the recycled materials upwards and heavy scraps are collected at the bottom and by pushing other materials to various feeds [9].
Eddy current separation
This separation is based on inducing eddy current in metals and most of the time it uses to sort aluminium household. Based on faradays low electromotive forces are generated by a time variable magnetic field and the electromotive forces are perpendicular to magnetic field. The induced electromotive forces generate current when the magnetic field is within conductive materials [8]. The speed of rotor controls the magnetic field and generated eddy current(i) can be given by the following formula.
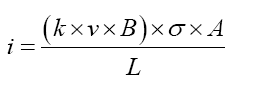
where Lis thickness, A is cross-sectional area, σ is conductivity, B is magnetic field flux density, ν is the frequency of oscillation, and (k × v × B) is potential difference across the material [9]. To improve the separation process between non-ferrous metals electromagnetic system is another and mere recent application of eddy current. To find small metallic objects on the ground induction separation uses with the same principle Figure 3[6].
The extension of eddy current separation technology takes advantage of the fact that the materials with different conductivity will produce different eddy currents and will therefore throw different distances (Tables 1 and 2).
| Elements | Electrical conductivity (× 10^6 âÃâæ cm)-1 | Elements | Electrical conductivity (× 10^6 âÃâæ cm)-1 |
|---|---|---|---|
| Al | 0.67 | Fe | 0.093 |
| Mn | 0.006 | Zn | 0.166 |
| Sb | 0.028 | Mg | 0.226 |
| Pb | 0.048 | Cu | 0.596 |
| Cr | 0.077 | Ag | 0.63 |
| Fe | 0.093 | ||
| Ni | 0.143 |
Table 1. Electrical conductivity of several metals [9].
| Material | Density (Kg/dm3) | Material | Density (Kg/dm3) |
|---|---|---|---|
| Aluminium | 2.6-2.9 | Polystyrene | 1.0-1.1 |
| Lead | 10.7-11.3 | Polypropylene | 0.9 |
| Copper | 7.5-9.0 | Rubber | 0.8-0.9 |
| Stainless steel | 7.5-7.7 | Wood | 0.4-0.8 |
| Brass and bronze | 5.0-7.0 | Polymeric foam | 0.01-0.6 |
| Zinc | 5.2-7.2 | Magnesium | 1.7-1.9 |
| Polyvinyl chloride | 1.4 |
Table 2. densities of aluminium and contaminate materials.
Heavy media separation
By using scraps density difference the heavy media separation or sink float separation uses to separate non-ferrous materials. There are three steps in density-based separation the first step is specific gravity with 1 or water based and uses to separate non-metallic fraction such as foams, wood, plastics and so on. The second step is 2.5 specific gravity bath and it uses to separate magnesium and plastics with higher density. In order controlling the bath magnetite or ferrosilicon powder are used [10]. The third separation step is based on specific gravity with 3.5 and it uses to separate cast and wrought aluminium metals from other heavier metals like copper, lead, and zinc Figure 4 [11].
Hand sorting
In this process by using hand scraps are separated from their inclusions. Mostly this process uses to separate household scraps. Hand sorting technique requires large number of labors. Hand sorting technique is more comfortable in Africa and for developing world. Because the availability of labor with fair cost is high [12,13]. By using visual recognition hand sorters distinguish desirable item in a feed stream and also by using their color it can be easily separate metallic materials with non-metallic materials and also other metals such as brass, copper, and lead. And shape also one important separation way that uses to separate tableware which it made from stainless steel, cans that are made from aluminium or steel and plastic cartons are made from high density polyethylene. Painted and plated metals are difficult to sort by using hands.
Color sorting
By using the advantage of their color difference color sorting technique uses to separate different types of scraps such as copper, zinc, stainless, and brass from aluminium scrap components. When metals are sorted by using hand sorting technique color sorting is basic applicable technique [14,15]. Based on specific color ranges color sorting technique can be automated by using computer image analyzing process.
Hot crush
Thermal-mechanical based of scrap separation is called hot crush. And it uses to separate wrought aluminium from cast aluminium by using their difference between the solidus temperatures. Wrought aluminium component has higher melting temperature than that of cast aluminium alloy component and holding or soaking the both scrap at temperature below the eutectic will result in a softening of the cast aluminium scrap components along their grain boundaries. After that mechanical crushing or grinding causes the alloy to separate from wrought aluminium scrap components. Currently this way is one of best scrap separation technique in aluminium cable industry [16,17].
In energy conservation and environment protection perspective aluminium industries are leading advocate and apricate of global sustainability by use of recycled materials. Without any loss of its potential property aluminium can be recycled with energy and emission saving. And also, aluminium can be recycled several times without significant degradation. Due to wide usage of aluminium metal in different application area the availability of aluminium scrap is high and for electrical conductor industry the use of this scrap has restriction due to the inverse relation of impurity and electrical conductivity. Used aluminium conductor cable, cans and other high purity-based scrap are main and valuable components of municipal scrap stream. Aluminium recycling requires low capital and lower energy than that of primary production. Most cable industry in Africa uses downgrading and dilution system for aluminium recycling because of difficulties in refining process [18-20]. There are more technologies which is not covered here are in development stages. To solve aluminium cable industry problems which they are facing regarding to accumulation of unwanted element in their scrap melted conductor cable product requires understanding of how scrap recycling parameters can be influenced by impurity and their characterization, scrap materials end life flow and evaluations of economical production method that affected by technological change.
Aluminium recycling process innovation should have target on maximizing the aluminium scrap value and increasing the capacity of melting different types of aluminium scraps and new processing integrated design should be found from the starting to modify comminution process and to improve energy consumption. Per-melt treatment that are described in this review article and that are found in developing process phase are key to increase the final quality of recycled aluminium conductor and reducing refining problem which is in-melt treatment. Aluminium scraps their color, shapes, density and usage history are use to support to the type of sorting decision. In Ethiopia and in Africa as well as other developing countries using pre-melt treatment such as hand sorting, air separation, magnetic separation, etc. will gives high advantages for cable industries by saving energy and economy and by protecting the environment.